Stryker to Acquire Amplitude Vascular Systems, Expanding IVL Portfolio
Participants
Why It Matters
The acquisition broadens Stryker’s product suite in a fast‑growing cardiology niche, potentially accelerating revenue growth and offering clinicians a novel plaque‑modification tool. It also intensifies competition in the IVL market, pressuring incumbents to innovate and potentially speeding FDA clearance timelines.
Key Takeaways
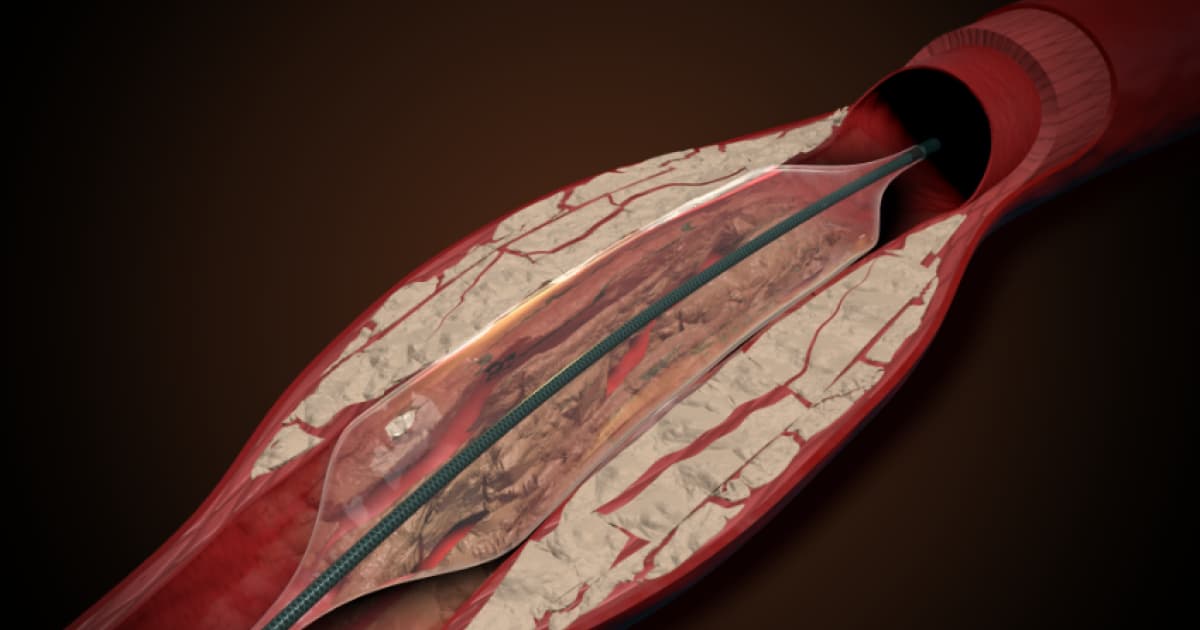
- •Stryker adds Pulse IVL platform to peripheral vascular portfolio
- •AVS's Pulse IVL uses balloon-transmitted pressure waves, no emitter needed
- •Acquisition pending regulatory approval; terms undisclosed
- •IVL market dominated by Shockwave; competitors like Boston Scientific expanding
- •Clinical trials target FDA clearance for Pulse IVL by 2027
Pulse Analysis
Stryker’s entry into intravascular lithotripsy reflects a strategic push to capture a larger share of the peripheral artery disease (PAD) treatment market. By integrating Amplitude Vascular Systems’ Pulse platform, Stryker can offer a catheter‑based solution that avoids the bulkier emitters used by rivals, potentially simplifying procedures and reducing procedural time. This technology aligns with hospitals’ demand for minimally invasive tools that improve patient outcomes while containing costs, a combination that could translate into strong adoption rates once regulatory hurdles are cleared.
The IVL landscape has been dominated by Shockwave Medical, now under Johnson & Johnson, but recent investments from Boston Scientific, Abbott, and Philips signal a crowded field eager for differentiation. Pulse IVL’s unique approach—delivering pulsatile pressure waves through the balloon’s length—offers a distinct clinical advantage, especially in heavily calcified lesions where traditional atherectomy devices struggle. As competitors race to refine their wave‑generation mechanisms, Stryker’s scale and global sales network give it a competitive edge in securing market share and influencing physician preferences.
Regulatory approval remains the pivotal milestone for Pulse IVL. Ongoing trials are designed to demonstrate safety and efficacy comparable to existing IVL systems, with a target FDA clearance around 2027. Successful clearance would not only expand Stryker’s revenue streams but also provide a broader therapeutic arsenal for interventional cardiologists, potentially reducing the need for more invasive surgical bypasses. Investors are likely to watch the clearance timeline closely, as it could catalyze a shift in the peripheral vascular market dynamics and drive future M&A activity among med‑tech firms seeking to bolster their IVL capabilities.
Deal Summary
Stryker announced an agreement to acquire Boston‑based Amplitude Vascular Systems, the maker of the Pulse IVL platform. The terms were undisclosed and the deal remains subject to customary closing conditions. The acquisition broadens Stryker’s peripheral vascular offerings and positions it in the competitive intravascular lithotripsy market.
Comments
Want to join the conversation?
Loading comments...