Dermal Filler Recently Approved by FDA Could Skew Breast Imaging Results, Experts Warn
Why It Matters
If the filler interferes with mammography, early breast cancer detection could be compromised, affecting patient outcomes and prompting tighter regulatory oversight for aesthetic products near diagnostic zones.
Key Takeaways
- •Radiesse approved for décolleté use in adults 22+.
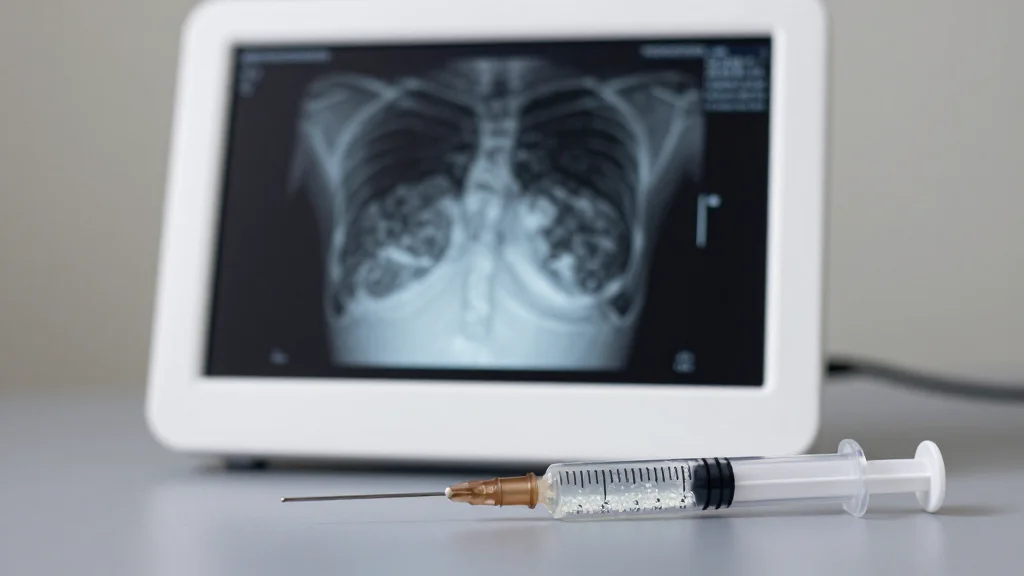
- •Hydroxyapatite microspheres may appear on mammograms.
- •FDA requires post‑market imaging study of 30 patients.
- •Merz disputes imaging concerns, cites no evidence.
- •Patients advised to disclose filler before breast exams.
Pulse Analysis
The recent FDA clearance of Radiesse for the upper‑chest (décolleté) marks a notable expansion for a filler already popular in facial rejuvenation. Hydroxyapatite‑based injectables like Radiesse have been praised for durability and natural feel, driving demand among cosmetic practitioners seeking to smooth fine lines on the chest. This approval opens a new revenue stream for Merz Aesthetics, positioning the product alongside other high‑volume aesthetic treatments and signaling regulatory confidence in its safety profile for a broader anatomical area.
However, the very composition that makes Radiesse effective—microscopic calcium‑phosphate spheres—poses a unique challenge for breast imaging. Radiologists warn that these particles can create radiopaque artifacts on mammograms and potentially obscure tiny calcifications that signal early malignancy. The concern is amplified by the filler’s proximity to breast tissue, where even subtle image distortion can affect diagnostic accuracy. Studies on similar fillers have shown mixed results, but the lack of definitive data has prompted clinicians to advise patients to disclose any chest injections before scheduling screening exams.
In response, the FDA has mandated a limited post‑market assessment involving 30 participants, requiring baseline imaging before a series of three filler treatments and follow‑up scans after completion. This precautionary step reflects a growing regulatory focus on the intersection of cosmetic procedures and diagnostic imaging. For providers, the takeaway is clear: integrate thorough patient histories into imaging protocols and consider alternative treatment sites when possible. For patients, transparency about cosmetic procedures can safeguard timely cancer detection, preserving both aesthetic goals and health outcomes.
Dermal filler recently approved by FDA could skew breast imaging results, experts warn
Comments
Want to join the conversation?
Loading comments...