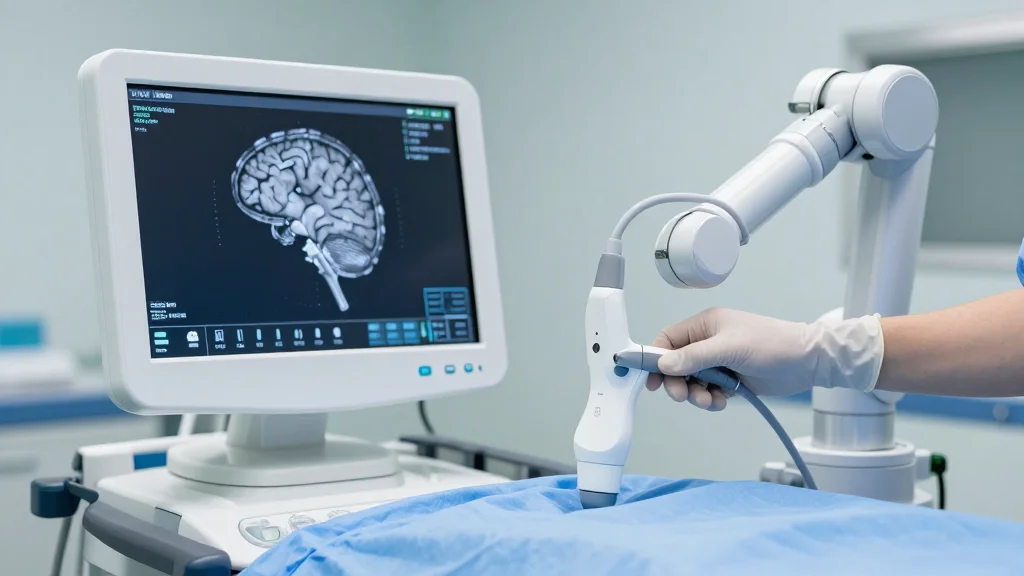
Medtronic Wins FDA Clearance for Robot in Cranial, ENT Surgeries
Why It Matters
The clearance expands Medtronic’s robotic portfolio, opening revenue streams in high‑margin neurosurgery and ENT markets. It also raises the competitive bar for image‑guided surgical robotics, potentially reshaping operating‑room workflows.
Key Takeaways
- •FDA grants 510(k) clearance for cranial and ENT uses
- •Stealth AXiS integrates AI tractography and real‑time ultrasound
- •System targets hospital and ambulatory surgery centers
- •Medtronic aims to capture share of $15B market
- •Surgeons report improved decision‑making and anatomical detail
Pulse Analysis
The FDA’s recent 510(k) clearance for Medtronic’s Stealth AXiS system marks a pivotal moment for image‑guided robotic surgery. While the company first introduced the platform for spine procedures, the new approvals for cranial and ENT applications unlock a broader patient base and reinforce the trend toward modular, AI‑driven operating‑room tools. By marrying AI‑based tractography with GE HealthCare’s bkActiv ultrasound, Stealth AXiS offers surgeons a richer, real‑time view of complex anatomy, which can translate into shorter procedures and potentially better outcomes.
From a market perspective, Medtronic’s move directly targets the $15 billion cranial and spinal technologies segment, a space dominated by a handful of legacy players. The ability to deploy the same hardware across multiple specialties—spine, brain, sinus and skull‑base surgery—creates economies of scale and positions the company to capture incremental share from hospitals seeking versatile, cost‑effective solutions. Analysts are watching the rollout closely, as successful adoption could accelerate Medtronic’s revenue trajectory and pressure competitors to broaden their own robotic portfolios.
Clinically, the integration of AI‑enabled tractography and real‑time ultrasound addresses a long‑standing challenge: delivering precise, patient‑specific navigation without overwhelming the surgical team. Surgeons like Dr. Michael Ivan and Prof. Vijay Ramakrishnan highlight how enhanced visualization supports more confident decision‑making in high‑risk procedures. As hospitals increasingly prioritize minimally invasive techniques and outcome‑driven metrics, technologies that improve anatomical fidelity and workflow efficiency—such as Stealth AXiS—are likely to become standard fixtures in modern operating rooms.
Medtronic wins FDA clearance for robot in cranial, ENT surgeries
Comments
Want to join the conversation?
Loading comments...