No-Option CLTI Patients Report Better QoL After Blood Flow Diversion Therapy: PROMISE III
Why It Matters
TADV provides a viable limb‑salvage option for patients previously deemed untreatable, lowering amputation risk while delivering measurable quality‑of‑life gains, which could reshape CLTI care pathways and reimbursement models.
Key Takeaways
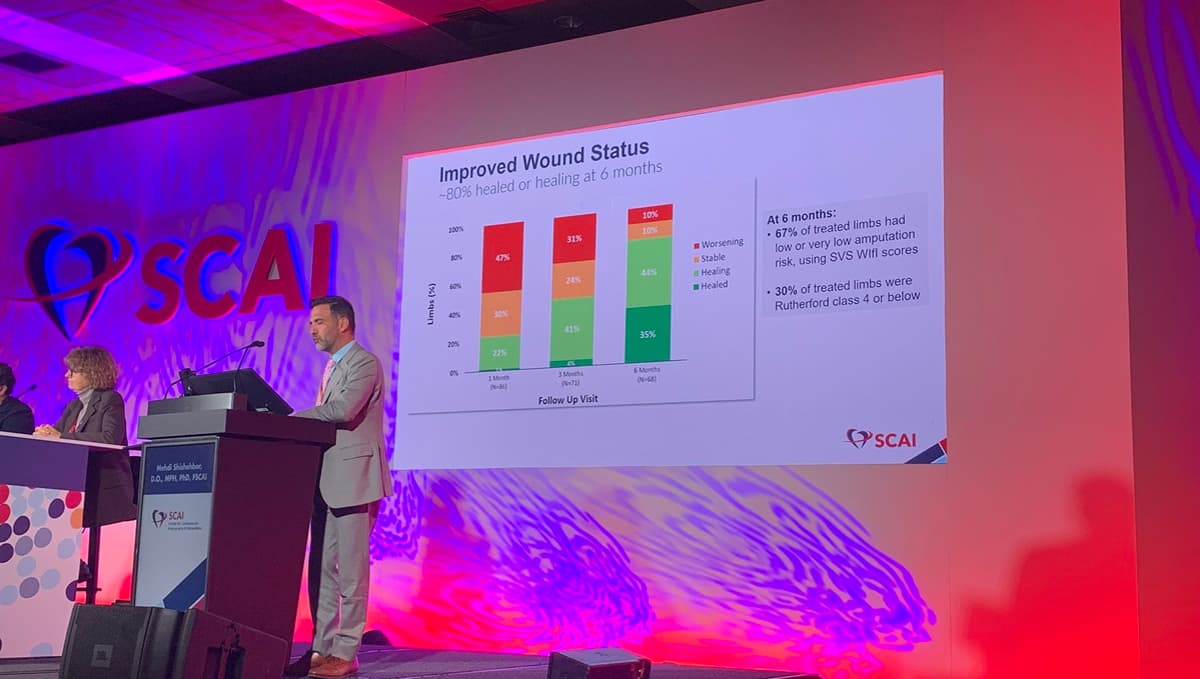
- •80% of patients had healed or healing wounds at 6 months
- •81% survived six months without major amputation (amputation‑free survival)
- •Median pain score dropped from 6 to 2 after TADV
- •Reintervention rate fell to ~16%, down from two‑thirds in PROMISE II
Pulse Analysis
Chronic limb‑threatening ischemia has long been a therapeutic dead‑end for patients who exhaust conventional revascularization options. The LimFlow system creates a controlled arteriovenous fistula, diverting arterial flow into the foot’s venous network and bypassing occluded tibial arteries. After FDA clearance in 2023, the technology entered broader clinical evaluation, and PROMISE III provides the first robust, multicenter data set confirming its safety and efficacy in a high‑risk cohort with a median age of 71.
The trial enrolled 100 no‑option CLTI patients across 24 U.S. sites, most of whom carried diabetes, hypertension, and chronic kidney disease. Six‑month outcomes reveal a striking 80% wound‑healing or healing rate and an 81% amputation‑free survival figure, far surpassing historical benchmarks. Pain, a primary driver of disability, fell dramatically, while patients reported fewer sleep disruptions and greater participation in social activities. Notably, the need for secondary limb interventions dropped to roughly 16%, indicating that procedural refinements and better patient selection are translating into real‑world efficiency gains.
For health systems, the data present both an opportunity and a challenge. While the clinical benefits are clear, the upfront cost of the LimFlow device and the specialized training required for plantar vein access limit rapid diffusion. Ongoing discussions with CMS aim to secure reimbursement pathways, which could make the technology financially viable for larger limb‑salvage programs. As more evidence accumulates, TADV may become a standard of care for the most vulnerable CLTI patients, driving both improved outcomes and potential cost savings through reduced amputations and associated long‑term care.
No-Option CLTI Patients Report Better QoL After Blood Flow Diversion Therapy: PROMISE III
Comments
Want to join the conversation?
Loading comments...