Survey Indicates Preferences for Needle-Free Epinephrine Options
•March 1, 2026
0
Why It Matters
The clear preference for needle‑free, portable epinephrine signals a shift that could accelerate product innovation and improve reaction times, potentially saving lives. Clinicians and manufacturers must align with these expectations to remain competitive.
Key Takeaways
- •90% prefer needle‑free epinephrine over injectors.
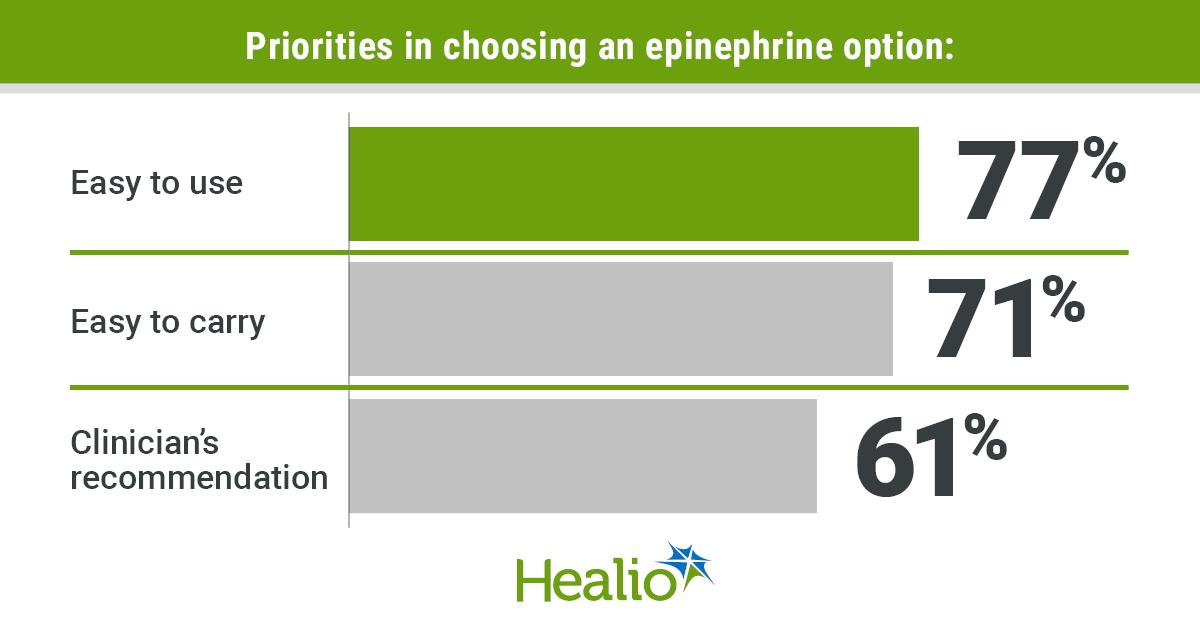
- •89% prioritize small, easy‑to‑carry devices.
- •Needle‑free expected to cut response time by three minutes.
- •87% worry about heat affecting epinephrine stability.
- •Clinician recommendation influences 61% of device choice.
Pulse Analysis
The survey’s results underscore a growing demand for needle‑free epinephrine solutions, a segment that has historically lagged behind traditional autoinjectors. Patients and caregivers cite needle phobia and the inconvenience of bulky devices as barriers to timely administration. By offering a nasal spray or dissolvable film, manufacturers can address both psychological and logistical hurdles, positioning needle‑free formats as the next generation of anaphylaxis treatment.
Speed of delivery is a critical factor in anaphylactic emergencies. The study projects a three‑minute advantage for needle‑free options, shrinking the window between symptom onset and drug action. Faster administration can translate into reduced severity of reactions, lower emergency department visits, and ultimately better patient outcomes. Healthcare providers can leverage these data to counsel patients on device selection, emphasizing that a quicker, more comfortable option may improve adherence to emergency action plans.
Temperature stability emerged as a secondary yet notable concern, with 87% of respondents worried about heat degradation. This highlights an opportunity for formulation scientists to develop epinephrine products with enhanced thermal resilience, expanding usability in varied climates and storage conditions. As insurers and payers scrutinize cost‑effectiveness, the combination of patient preference, improved speed, and robust stability could drive broader reimbursement and market adoption, reshaping the competitive landscape for allergy and immunology therapeutics.
Survey indicates preferences for needle-free epinephrine options
0
Comments
Want to join the conversation?
Loading comments...