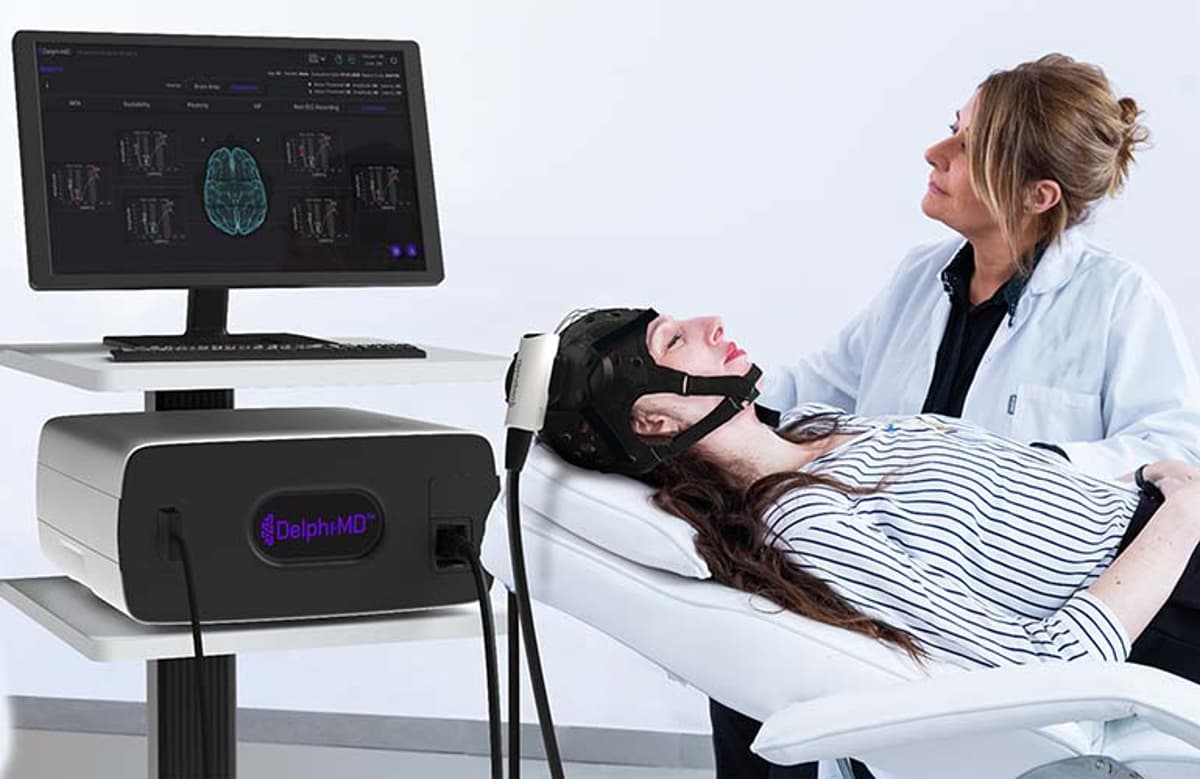
This Startup Developed a First-of-Its-Kind Brain Function Assessment Device
Why It Matters
By providing objective, bedside neuro‑functional data, Delphi‑MD could reshape diagnostic pathways and treatment monitoring across neurology and psychiatry, accelerating personalized care and reducing reliance on subjective assessments.
Key Takeaways
- •First FDA de novo TMS‑EEG combined device.
- •Real‑time brain function reports via proprietary algorithms.
- •Operable by non‑expert staff; quick setup.
- •Breakthrough designations for stroke, dementia, hydrocephalus.
- •Normative database currently ages 50‑75, expansion planned.
Pulse Analysis
The convergence of transcranial magnetic stimulation and electroencephalography has long been a research curiosity, but QuantalX’s Delphi‑MD marks the first commercial system to integrate these modalities into a single, FDA‑cleared platform. By eliminating the technical barriers that previously confined TMS and EEG to specialist labs, the device democratizes access to functional neuro‑imaging. Its automated artifact‑reduction pipeline and real‑time quality monitoring ensure data fidelity, while a user‑friendly probe and cap enable clinicians to obtain reliable measurements in minutes, positioning Delphi‑MD as a practical point‑of‑care tool.
Clinically, the device’s ability to benchmark patient responses against a multi‑center normative database opens new diagnostic avenues. Early‑stage FDA Breakthrough Device designations highlight its potential for stroke and dementia risk stratification, as well as predicting outcomes for ventriculoperitoneal shunt procedures in normal‑pressure hydrocephalus. These indications suggest Delphi‑MD could complement existing imaging modalities, offering functional insights where structural scans fall short. Neurologists and psychiatrists may soon rely on its objective metrics to differentiate Parkinson’s disease from Lewy‑body dementia, assess traumatic brain injury, and monitor disease progression, thereby enhancing treatment precision.
Looking ahead, QuantalX plans to broaden its normative database beyond the current 50‑75 age range and to accumulate condition‑specific datasets. This expansion will enable more granular, indication‑focused reporting, fostering a data‑driven approach to brain health. As hospitals and outpatient clinics adopt the technology, the market could see a shift toward continuous, personalized neuro‑functional monitoring, similar to how wearable cardiac devices transformed cardiology. The convergence of robust analytics, regulatory endorsement, and scalable usability positions Delphi‑MD to become a cornerstone of next‑generation neurological care.
This startup developed a first-of-its-kind brain function assessment device
Comments
Want to join the conversation?
Loading comments...