Mortality Risk Similar with Tocilizumab, Rituximab in RA-ILD
•March 11, 2026
0
Why It Matters
Demonstrating comparable mortality between tocilizumab and rituximab broadens viable treatment choices for RA‑ILD, a condition with scarce high‑quality evidence. The results also underscore the urgency for prospective trials to personalize therapy based on patient characteristics.
Key Takeaways
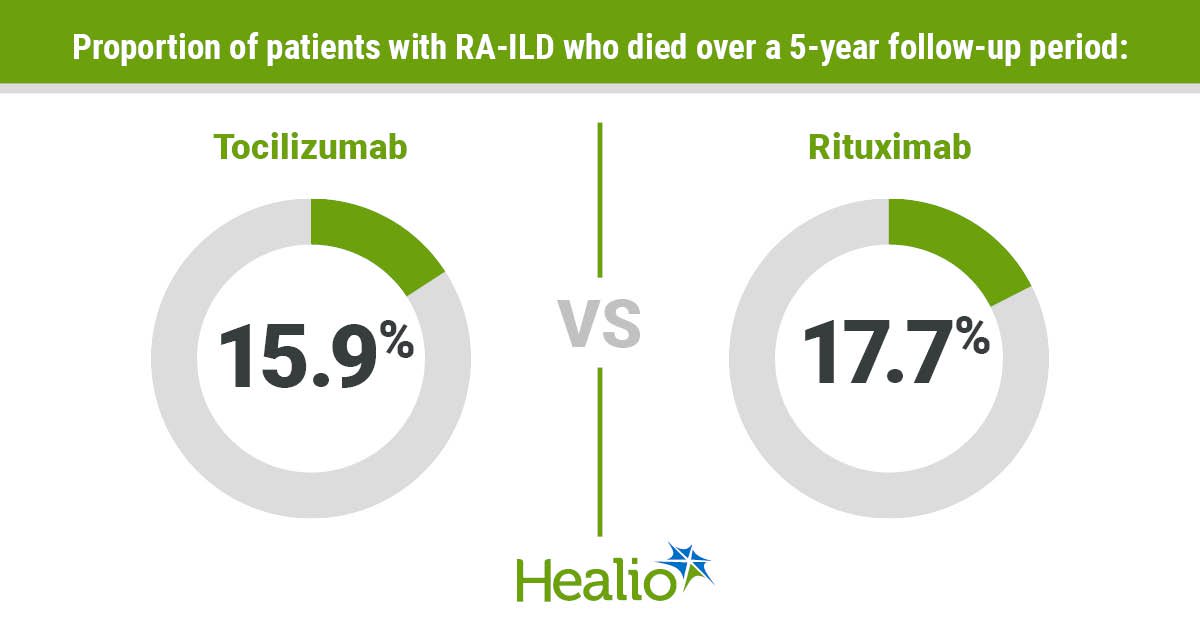
- •Mortality similar for tocilizumab and rituximab in RA‑ILD
- •Hospitalization and ventilation risks also comparable across treatments
- •Elevated inflammatory markers favor rituximab over tocilizumab
- •Women had lower hospitalization; men higher ventilation with tocilizumab
- •Retrospective design limits causality; severity metrics unavailable
Pulse Analysis
Rheumatoid arthritis‑associated interstitial lung disease remains a therapeutic gray zone, with clinicians balancing disease‑modifying antirheumatic drugs against the risk of pulmonary deterioration. Biologic agents such as tocilizumab, an IL‑6 inhibitor, and rituximab, a B‑cell depleting antibody, have emerged as potential options, yet robust comparative data have been lacking. The scarcity of randomized controlled trials—only the TRAIL1 study to date—has left treatment guidelines reliant on observational evidence and expert opinion, prompting the need for large‑scale real‑world analyses.
The recent TriNetX‑based emulated target trial leveraged propensity‑score matching to align 1,194 patients per arm on demographics, comorbidities, and medication use, providing a rare head‑to‑head view of long‑term outcomes. Primary findings revealed equivalent five‑year all‑cause mortality, hospitalization, mechanical ventilation, and serious infection rates between tocilizumab and rituximab. Notably, women experienced a modest reduction in hospitalization with tocilizumab, whereas men faced a higher ventilation risk, and patients with heightened inflammatory markers showed increased mortality on tocilizumab. These nuances suggest that sex and baseline inflammation may inform agent selection, even as overall efficacy appears comparable.
Despite its size, the study’s retrospective nature imposes critical constraints. Absence of pulmonary function test data, ILD pattern classification, and granular RA disease activity hampers risk adjustment, and over half of the rituximab cohort was excluded during matching, raising selection bias concerns. Consequently, clinicians should interpret the results as hypothesis‑generating rather than definitive. Ongoing prospective trials and registries will be essential to validate these observations, refine risk stratification, and ultimately shape guideline updates for managing RA‑ILD with biologic therapies.
Mortality risk similar with tocilizumab, rituximab in RA-ILD
0
Comments
Want to join the conversation?
Loading comments...