Today's Pharma Pulse
CMS to launch Medicare GLP‑1 Bridge program with $50 monthly copay for obesity drugs
The Centers for Medicare & Medicaid Services will start a short‑term demonstration on July 1 2026 offering eligible Part D beneficiaries a flat $50 monthly copay for three branded GLP‑1 obesity treatments. The program runs through December 31 2027 and aims to gather utilization data ahead of potential broader policy decisions.
Kura Oncology Reports 67% Response Rate for Darlifarnib Combo in Pancreatic Cancer at ASCO
Kura Oncology announced a 67% confirmed objective response rate for its darlifarnib and adagrasib combination in KRAS G12C‑mutated pancreatic ductal adenocarcinoma, based on Phase 1a data presented at ASCO 2026. The early signal, derived from 26 evaluable patients, could reshape treatment strategies for a cancer with historically poor outcomes.

Are GLP-1 Medications Transforming Chronic Disease?
She Med’s video argues that GLP‑1 medications are a clinical breakthrough for chronic disease, especially for women with conditions like PCOS, rather than a cosmetic weight‑loss tool. The company cites its large real‑world study showing participants experience reduced inflammation, regular menstrual...

Noxopharm CEO Discusses Sofra Cancer Platform
Noxopharm CEO Olivier Lasker outlined the company’s Sofra platform, an RNA‑based drug suite designed to modulate immune‑receptor activity. The technology, built on a seven‑year discovery of endogenous immune regulation, aims to either dampen or amplify inflammation, depending on the disease...

Veozah & Cancer Risk: What Menopausal Women with Hot Flashes Should Know | Felice Gersh, MD
Dr. Felice Gersh discusses Vioza (generic fezolinitant), a newly approved neurokinin‑3 receptor antagonist marketed for moderate‑to‑severe menopausal vasomotor symptoms. While clinical trials such as Skylight and Moonlight demonstrated efficacy, recent peer‑reviewed research raises alarms about a potential link between the...
Imugene CEO Leslie Chong Highlights Promising Blood Cancer Data
Imugene CEO Leslie Chong unveiled results from Cohort 2, a basket trial targeting blood‑cancer patients who have never received an autologous CAR‑T therapy. The study spans chronic lymphocytic leukemia, marginal‑zone lymphoma, Waldenström macroglobulinemia and follicular lymphoma, and early data indicate objective...
ADA: Obesity Partners Zealand and Roche Aim for Weight Loss ‘Sweet Spot’ with Amylin Agonist
Roche and its partner Zealand reported that their amylin analog petrelintide produced a 9% weight reduction after 42 weeks in the Phase 2 ZUPREME‑1 trial, falling short of analyst expectations for 12% loss and behind Lilly’s eloralintide. The companies argue that...

Correlation Between Prognostic Nutritional Index and Oral Mucositis in Patients with Lung Adenocarcinoma Treated with Almonertinib: A Multicenter Prospective Study
A multicenter prospective study of 537 Chinese lung adenocarcinoma patients treated with the third‑generation EGFR‑TKI almonertinib found that low baseline prognostic nutritional index (PNI) significantly increased the risk of oral mucositis (OM). Each one‑unit rise in PNI reduced OM odds...
H. Lundbeck A/S (HLBBF) Discusses PROCEED Trial Results and Data Presented at AHS on Bokunebart for Headache Treatment Transcript
Lundbeck presented Phase 3 PROCEED trial data for its migraine therapy bokunebart (formerly Lu AG09222) at the American Headache Society meeting. The study met its primary endpoint, showing a roughly 45% reduction in monthly migraine days versus placebo, and demonstrated a...
MAIA Therapeutics Targets 100 Patients in Phase 3 NSCLC Trial of Ateganosine
MAIA Therapeutics announced that its Phase 3 THIO-104 trial of ateganosine in third‑line non‑small cell lung cancer has dosed 29 patients across 34 sites and will aim for 100 enrollments by year‑end. The FDA granted Fast Track designation, positioning the...
FDA Accepts Pharming's Joenja Resubmission, Paving Way for Pediatric APDS Treatment
The U.S. FDA has accepted Pharming Group's supplemental New Drug Application to expand Joenja (leniolisib) to children aged 4 to 11 with activated PI3K‑δ syndrome. The agency set a PDUFA target decision date for fall 2026, moving the therapy closer...

Bio-Techne, Refeyn Partner on Workflow for Bispecific Antibody, Biosimilar Characterization
Bio‑Techne and Refeyn have launched an integrated workflow that pairs Bio‑Techne’s MauriceFlex icIEF fractionation system with Refeyn’s TwoMP mass‑photometry platform. The solution enables researchers to correlate charge heterogeneity with molecular weight and aggregation at single‑molecule resolution in about four hours...

Study Data Convince Lundbeck to Push New Migraine Drug Forward
Denmark‑based Lundbeck reported Phase 2 data for its experimental migraine preventive, bocunebart, which targets the PACAP pathway. In the intravenous arm, patients experienced a 4.24‑day reduction in monthly migraine days versus a 2.86‑day drop with placebo, and the broader Phase 2 program...

Biogen's Salanersen Wins FDA Breakthrough Therapy Designation for SMA
Biogen announced that the FDA has granted Breakthrough Therapy Designation to its antisense drug Salanersen for spinal muscular atrophy (SMA). The designation follows Phase 1b data showing motor‑function gains in children who previously received gene therapy. The move accelerates Biotech’s Phase 3...
IMUNON Raises $10 Million to Fund Phase 3 Ovarian Cancer Trial, Bolstering B2B Pharma Pipeline
IMUNON, Inc. announced a $10 million cash financing package that includes preferred stock and secured promissory notes to fund its pivotal Phase 3 OVATION 3 ovarian cancer trial. The capital infusion is aimed at expanding enrollment and positioning the company for deeper B2B...
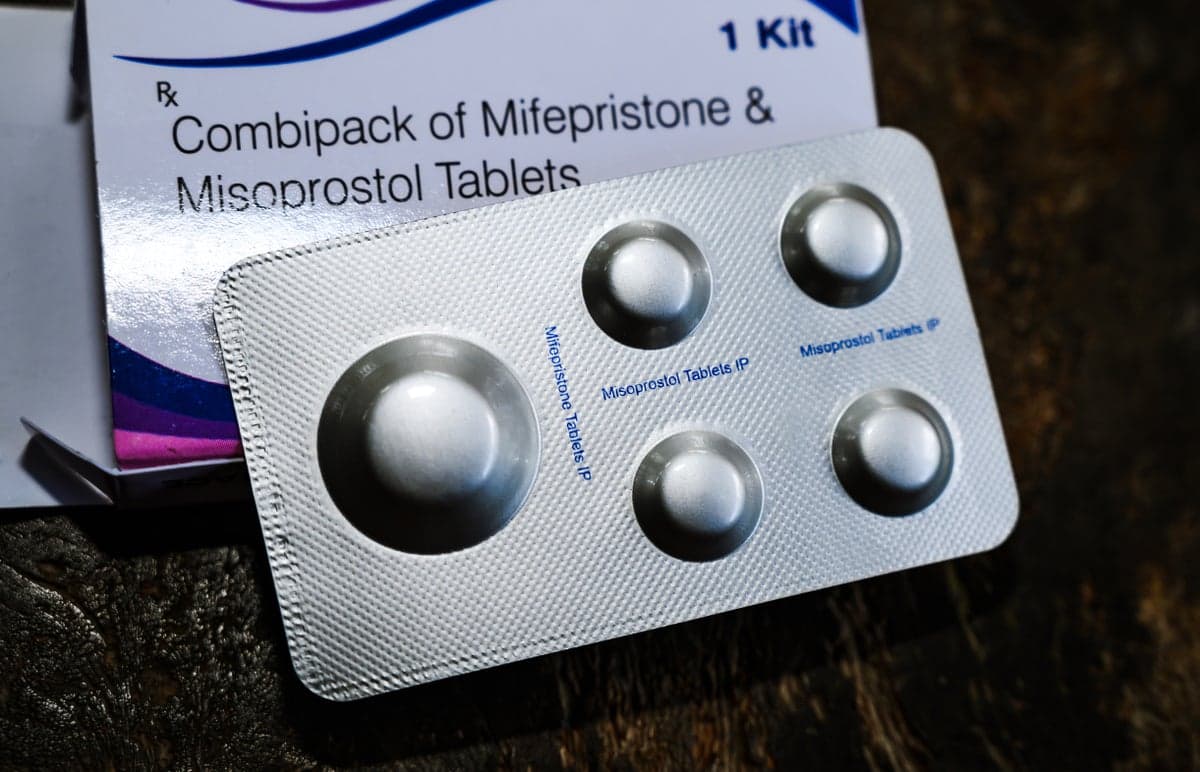
Report: FDA Just Launched a Study on the Abortion Pill
The FDA has opened a six‑month safety review of mifepristone, the abortion pill approved since 2000, amid heightened political scrutiny from the Trump administration. Health experts emphasize that extensive research already confirms the drug’s safety, even when delivered via telehealth....

Mabwell Secures IND Clearance in China for 9MW5211 IBD Antibody
Mabwell announced that China’s National Medical Products Administration approved an IND for its antibody 9MW5211, allowing the first clinical trials of the drug in inflammatory bowel disease on the mainland. The clearance follows a U.S. FDA approval and positions the...

Quoin Secures Japan Orphan Drug Designation for QRX003 Targeting Netherton Syndrome
Quoin Pharmaceuticals announced that Japan's Ministry of Health, Labour and Welfare granted orphan drug designation to its QRX003 therapy for Netherton syndrome, promising up to a decade of market exclusivity. The move adds to QRX003’s U.S. pediatric rare‑disease and fast‑track...
Jade Biosciences Raises $150 Million in Equity Offering at $15 per Share
Jade Biosciences completed a $150 million public equity offering of 10 million shares at $15 each, with an additional 1.5 million‑share over‑allotment option. The proceeds will fund its Phase 2 trial of JADE101 and other R&D initiatives, underscoring strong investor demand for biotech capital.
Lupin Secures FDA Approval for Ranluspec, First Interchangeable Ranibizumab Biosimilar in U.S.
Lupin Limited announced that the U.S. FDA has approved Ranluspec™ (ranibizumab‑hkdz) as an interchangeable biosimilar to Lucentis®, making it the only such product available in both vial and pre‑filled syringe formats. The approval expands affordable treatment options for wet age‑related...