Today's Pharma Pulse

Neolaia unveils new CD38 inhibitor series in preclinical data
Neolaia announced the synthesis of a novel series of CD38 inhibitors, marking a significant step in its preclinical development pipeline. In parallel, Incyte disclosed its own next‑generation TYK2 and JAK1 inhibitor candidates, highlighting broader activity in kinase‑targeted drug discovery.
Also developing:
By the numbers: Asahi Kasei acquires German biotech Aicuris for $920.7M

SynbioTech's L. Plantarum FS4722 Emerges as a Potential Preventive Approach for Hyperuricemia
SynbioTech announced that its probiotic Lactiplantibacillus plantarum FS4722 markedly lowers serum uric acid in mouse models, matching the efficacy of conventional uric‑lowering drugs while showing no kidney toxicity. The strain works through a multi‑mechanism gut‑liver‑kidney axis: it reduces intestinal purine absorption, downregulates hepatic uric‑acid synthesis, and boosts renal and intestinal excretion, alongside rebalancing the gut microbiome. Results were published in *Microbiome* and presented at ACLAB 15 to an audience from 17 countries. SynbioTech, with 60 patents and a global supply network, positions FS4722 as a preventive probiotic for hyperuricemia.

Anti-Amyloid Drugs May Not Work Against Alzheimer's but if so, What Will?
A Cochrane systematic review of 17 clinical trials involving over 20,000 patients with early Alzheimer’s found that anti‑amyloid drugs produce little to no clinically meaningful improvement in cognition or daily function. The analysis also highlighted an increased risk of brain...

Natural GLP-1 Discovery Hidden in Joints Could Revolutionize Arthritis Treatment
A study published in The Lancet Rheumatology found trace amounts of the body’s natural GLP‑1 hormone in the synovial fluid of patients with rheumatoid arthritis and spondyloarthritis. Researchers compared joint‑fluid and blood samples from the INART biobank and observed a...

Noxopharm SOF-SKN Data Shows Prolonged Skin Retention with Minimal Systemic Absorption
Australian biotech Noxopharm announced preclinical pharmacokinetic results for its SOF‑SKN candidate, showing a skin half‑life of roughly 3.5 days with negligible systemic exposure. The active moiety, SOF‑16, demonstrated consistent dermal concentrations across normal and disease‑like skin and remained confined to...

FDA Clears Merck's First Once‑Daily Oral HIV Pill
The U.S. Food and Drug Administration approved Merck's once‑daily oral HIV pill, the first daily regimen from a major pharmaceutical firm in recent years. The decision positions Merck to compete more directly in the HIV market and could reshape treatment...

FDA Issues Draft Guidance to Fast‑Track Ultra‑Rare, Individualized Therapies
The U.S. Food and Drug Administration released a draft guidance on Feb. 23, 2026 outlining a “plausible mechanism framework” for individualized therapies targeting ultra‑rare genetic diseases. The roadmap aims to replace traditional randomized trials with mechanistic data, accelerating approval pathways for treatments...

If a Bird Flu Pandemic Starts, We May Have an mRNA Vaccine Ready
A phase‑III trial of Moderna’s mRNA‑1018 vaccine against the H5N1 bird‑flu clade 2.3.4.4b has launched in the UK and the US, enrolling about 4,000 volunteers. The study will measure immune response rather than disease prevention because human transmission remains limited. mRNA...

Biology’s Data Gap: Making Lab Results AI-Ready
Biology generates enormous amounts of data. Almost none of it is ready for AI. #SynBioBeta2026 is May 4-7th in San Jose, California, you can learn more about the conference and get your tickets here: https://t.co/8abYWJ1GbK Most experimental infrastructure was built for individual...

Blocking PGAM‑Chk1 Kills Senescent Cells, Eases Lung Fibrosis
Researchers found that senescent cells depend on a PGAM–Chk1 interaction to boost glycolysis and survive, and blocking this interaction selectively kills these cells and reduces disease effects like lung fibrosis. https://t.co/VTsHTkBcad

STAT+: From Revolution Medicines, More Strong Data on KRAS Drug and a Glimpse of a ‘Novel Class’ Beyond It
Revolution Medicines presented new clinical data on its next‑generation KRAS inhibitor daraxonrasib at the AACR meeting, highlighting strong first‑line activity and synergistic effects in combination regimens for advanced pancreatic cancer. The company also unveiled pre‑clinical results for RM‑055, a candidate...

Durvalumab Plus FLOT Improves Resectable Gastric Cancer Outcomes
EFS in MATTERHORN: A Ph3 RCT of durvalumab plus 5-fluorouracil, leucovorin, oxaliplatin & docetaxel chemotherapy (FLOT) in resectable gastric/gastroesophageal junction cancer (GC/GEJC) - @YJanjigianMD et al. #ASCO25 LBA5 https://t.co/lW8KUtXdB3 #NCT04592913 #stcsm #esocsm

CtDNA‑guided Cam
Camizestrant + CDK4/6i for the Tx of emergent ESR1 mutations during 1L endocrine-based therapy & ahead of disease progression in Pts w/ HR+/HER2– advanced breast cancer (ABC): ...ctDNA-guided SERENA-6 trial - Turner et al. #ASCO25 LBA4 https://t.co/9jFfuGd1IS #NCT04964934 #bcsm

STAT+: Kyverna Therapeutics Plans to Submit Cell Therapy for Stiff Person Syndrome for FDA Approval
Kyverna Therapeutics announced that its personalized CAR‑T cell therapy improved mobility and reduced disability in patients with stiff person syndrome, a rare autoimmune neurological disorder. The company plans to file an FDA submission by mid‑2026, which would make it the...

Rusfertide Shows Efficacy in Phase 3 PV Trial
Results from VERIFY, a phase 3, double-blind, placebo (PBO)-controlled study of rusfertide for treatment of polycythemia vera (PV) - @KuykendallMd et al. #ASCO25 LBA3 https://t.co/XID3tkj6Sk #NCT05210790 #mpnsm

Adjuvant Nivolumab Boosts Outcomes in High‑Risk HNSCC
NIVOPOSTOP (GORTEC 2018-01): Ph3 RCT of adjuvant nivolumab added to radio-chemotherapy in Pts w/ resected head & neck squamous cell carcinoma at high risk of relapse - Bourhis et al. #ASCO25 LBA2 https://t.co/Uxh4JhZyDE #NCT03576417 #hncsm #ImmunoOnc

Zymeworks Presents New Phase 1 Data for ZW191, a Folate Receptor Alpha-Targeting ADC at AACR Annual Meeting 2026
Zymeworks unveiled Phase 1 data for ZW191, a folate‑receptor‑α‑targeting antibody‑drug conjugate, at the AACR 2026 meeting. In platinum‑resistant ovarian cancer the drug showed a confirmed objective response rate of 61% at 6.4‑9.6 mg/kg, with a median progression‑free survival of 7.6 months and responses...

Atezolizumab Boosts Adjuvant Therapy for dMMR Colon
RCT of standard chemotherapy +/- atezolizumab as adjuvant therapy for Pts w/ stage III deficient DNA mismatch repair (dMMR) colon cancer (@ALLIANCE_org A021502; ATOMIC) - @FASinicropeMD et al. #ASCO25 LBA1 https://t.co/cGalNPlxMe #NCT02912559 #crcsm

WGS Uncovers KMT2A PTD, Surpassing Old AML Tests
Detection of KMT2A partial tandem duplication (PTD) in AML by whole genome sequencing (WGS): Addressing limitations of traditional techniques in the era of revumenib approval - Huether et al. #ASCO25 Abst 6532 https://t.co/EEsXm7JGdv #AMLsm #leusm #PrecisionMedicine @TempusAI

Guizhou Xinbang Pharma Posts $16.8M Full-Year Profit Rise as Revenue Slides 6.8%
Guizhou Xinbang Pharmaceutical Co., Ltd. announced a full‑year profit of RMB120.285 million ($16.8 million), up from RMB101.381 million a year earlier, while revenue fell 6.8% to RMB5.619 billion ($787 million). The mixed results underscore pressure on Chinese drug makers to balance cost control with growth.
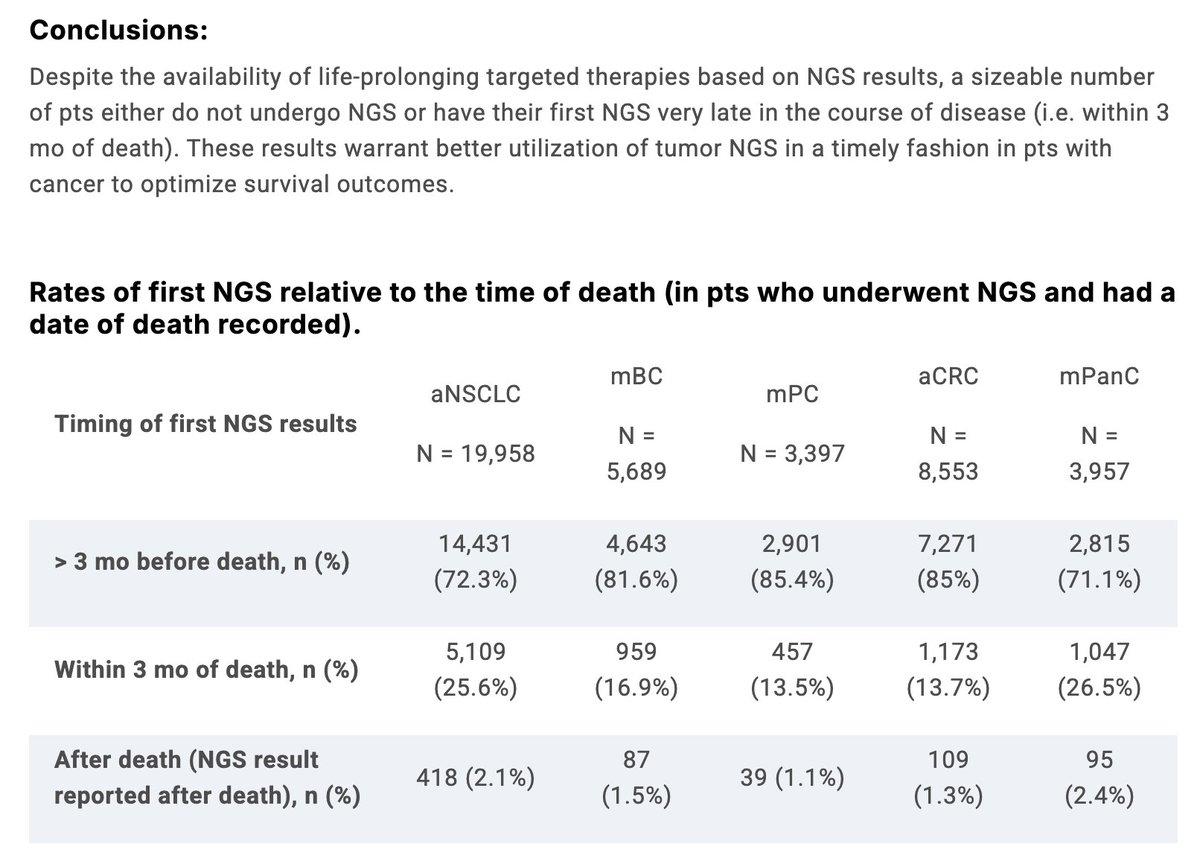
First Tumor NGS Usage Varies Across Top Five Cancers
Utilization and timing of first tumor next-generation sequencing testing (NGS) in Pts w/ 5 most common cancers in the USA - @chadihcmd et al. @huntsmancancer #ASCO25 Abstract 11014 https://t.co/lWefncnTb2 #PrecisionMedicine #hemonc #bcsm #crcsm #lcsm #pancsm #pcsm https://t.co/oIX8QTRD8x
Aging Mechanisms Are Now Engineerable, Says SynBioBeta
Aging isn't just inevitable decline. It's a set of biological systems that are starting to look engineerable. #SynBioBeta2026 is May 4-7th in San Jose, California, you can learn more about the conference and get your tickets here: https://t.co/8abYWJ1GbK DNA damage accumulates. Protein...
STAT+: Insurers Refuse to Join Medicare Pilot Offering Weight Loss Drugs to Seniors at Steep Discount
The Trump administration negotiated a $245‑per‑month price with Eli Lilly and Novo Nordisk for obesity drugs, intending to offer seniors a $50 monthly copay through a Medicare pilot called BALANCE. Insurers refused to join, arguing the program would strain their finances. Consequently,...

World's First Longevity Board Launched to Accelerate Aging Therapies
I am very happy to report that we formed the world's first Longevity Board at the public biotechnology company. Our objective is to discover and develop longevity therapeutics with pipeline-in-a-product potential. GLP-1 therapeutics paved the way and demonstrated the path...

Trump's Order Is a Milestone for Proponents of Using Psychedelics as Medicine
President Trump signed an executive order that mandates federal agencies to speed up research and regulatory approval of psychedelic compounds for mental‑health treatment. The order calls for the DEA to reassess scheduling of substances such as psilocybin and MDMA and...

Lilly's Retatrutide Achieves 28.7% Weight Loss, Higher Dropouts
Lilly’s triple G agonist boasts 28.7% weight loss in Phase III trial Lilly is investigating retatrutide in seven other Phase III trials, which are due to read out in 2026. 👨🏻⚕️“Discontinuation rates due to adverse events were 12.2% and 18.2% with retatrutide...

Zero Relapses Reported in Pre‑Surgery Immunotherapy Trial for Bowel Cancer
University College London and UCL Hospitals reported that none of the 32 stage II/III bowel‑cancer patients in the NEOPRISM‑CRC trial relapsed after a median 33‑month follow‑up. The pre‑operative pembrolizumab regimen shrank tumours in 59% of participants and could cut the need...

Discovery of a Small Molecule HPK1 Inhibitor for Immuno-Oncology
A biotech firm has disclosed a novel small‑molecule inhibitor of hematopoietic progenitor kinase 1 (HPK1) that demonstrates potent immuno‑oncology activity in preclinical models. The compound achieves sub‑micromolar potency, oral bioavailability, and drives up to 70% tumor regression when combined with...

Debiopharm Secures FDA Fast Track for Lunresertib/Zedoresertib in Platinum‑Resistant Ovarian Cancer
Debiopharm announced that the FDA granted Fast Track designation to its lunresertib‑zedoresertib regimen for adult patients with CCNE1‑amplified or FBXW7/PPP2R1A‑mutated platinum‑resistant ovarian cancer. The move follows Phase I data presented at AACR 2026, positioning the combo as a potential first‑in‑class...

Scaling Bio 008: Serif Biomedicines' Jake Rubens on Turning Modified DNA Into a New Class of Medicine
In this episode, Jake Rubens of Serif Biomedicines explains how the company is turning DNA into a new class of medicines by using chemically modified DNA and a protein co‑factor to overcome DNA’s historic immunogenicity and delivery challenges. He contrasts...
LLMs Beat JEPA World Models on Sparse Biological Data
Sci-JEPA's and Biology: I'm testing & training LeCunn style JEPA's @SciRouterAI vs more classical Dual encoder LLMs (small 35k data sets) for novel molecule targets for disease quick scans. What's interesting is that even though world models have, in theory, powerful...
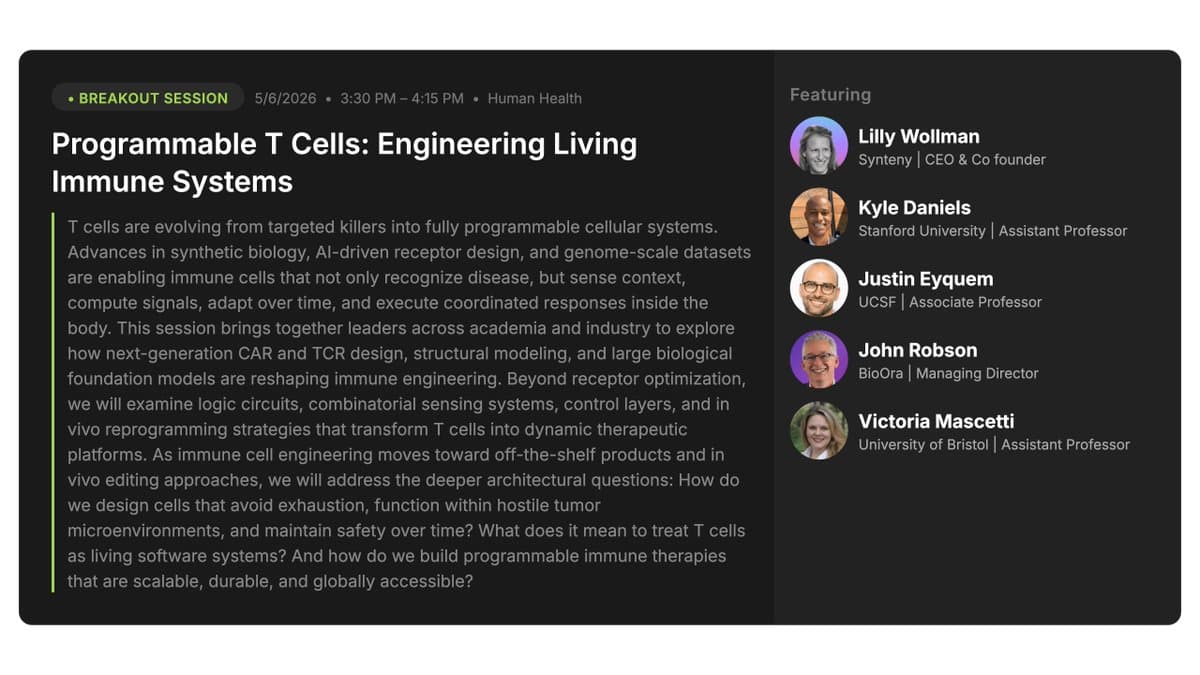
Beyond CAR T: Programmable Immune Cells Shaping Future Therapies
CAR T cells cured patients who had no other options. That was just the opening move. #SynBioBeta2026 is May 4-7th in San Jose, California, you can learn more about the conference and get your tickets here: https://t.co/8abYWJ1GbK The next generation of T...

The BioPharm Brief: AI, Immunology, and Regulatory Momentum
AstraZeneca announced consistent Phase III data showing its IL‑33 biologic cuts COPD exacerbations, reinforcing the cytokine as a therapeutic target. Boehringer Ingelheim disclosed a broadened AI program that will be embedded across early discovery and development stages to speed target identification. The...
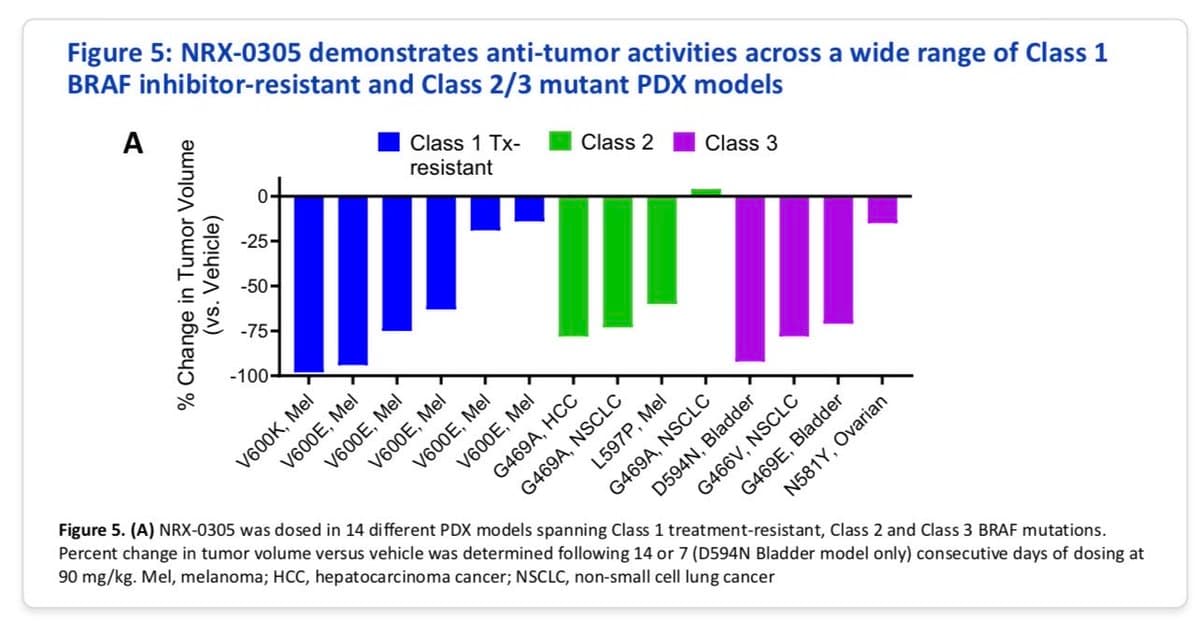
Nurix's NRX-0305 Shows
New degrader from Nurix looks interesting: NRX-0305 demonstrated antitumour activity across a wide range of Class 1 BRAF inhibitor-resistant and Class 2/3 mutant PDX models #aacr26 https://t.co/0UGF5Od2Cp

First Personalized CAR‑T Shows Mobility Gains in Stiff Person Syndrome
$KYTX Kyverna Therapeutics cell therapy improves mobility in patients with stiff person syndrome. If cleared, the medicine would be first personalized CAR-T therapy for an autoimmune disease https://t.co/B28Zp9IA2O

Business Watch: Inovyn Sells Off Italian Chlor-Alkali Assets; Lilly to Buy Kelonia
Inovyn, the Ineos Inovyn unit, agreed to sell its Italian chlor‑alkali sites at Rosignano and Tavazzano to Esseco Industrial, with the transaction slated for completion by year‑end and financial terms undisclosed. The sale lets Inovyn concentrate on its PVC and...

Daraxonrasib Monotherapy
Eileen O’Reilly’s daraxonrasib monoTx data in 1L PDAC - poster below to save y’all fighting through rows of people to get a view #aacr26 https://t.co/z4yW2apCBf
Avacta Unveils FAP‑cleavable Dual TOPO‑Ii/DDR‑i Payload
Intriguing #AACR26 poster from Avacta w/ CSO Francis Wilson presenting on their dual payload tech. The idea is to release TOPO-Ii + DDRi via FAP cleavage https://t.co/UMeB8y8KaC

CMS’ Chris Klomp, Stephanie Carlton Talk Affordability, Prior Authorization, Drug Pricing
CMS Deputy Administrator Chris Klomp and Chief of Staff Stephanie Carlton outlined the agency’s recent policy agenda, emphasizing four pillars—fraud reduction, affordability, health‑initiative programs, and AI‑driven streamlining. They highlighted a voluntary agreement to overhaul prior‑authorization processes and a 2025 most‑favored‑nation drug‑pricing pledge...
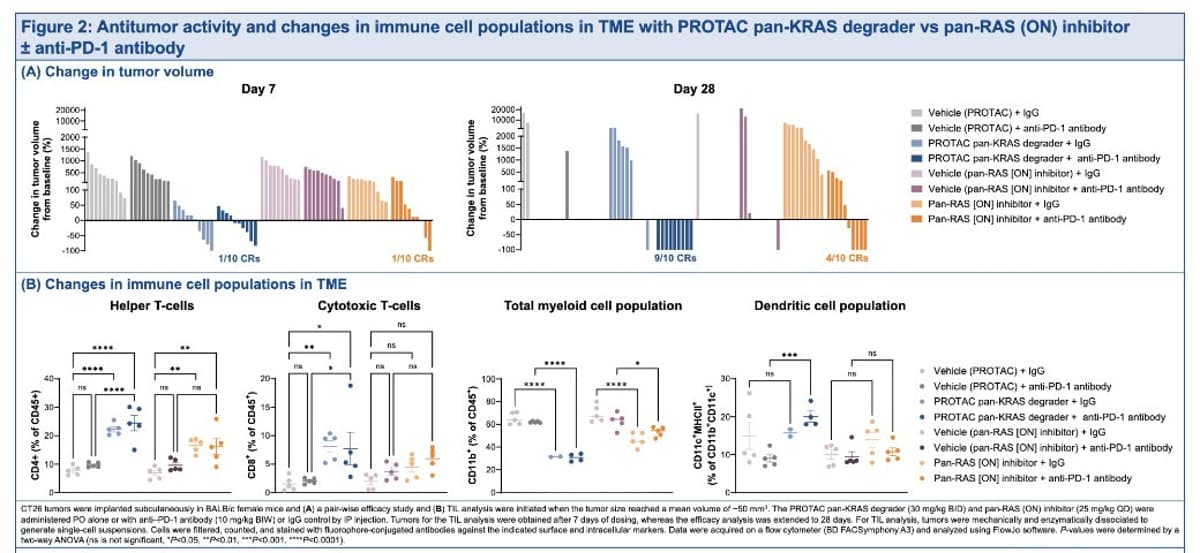
ARVN KRAS G12D Preclinical Shows Weak Tumor Inhibition
In the battle of the inhibitors vs glues vs degraders, this one (KRAS G12D preclinical) from ARVN doesn't look too encouraging with little TGI #aacr26 https://t.co/Ng9UWTQcsv

Monoclonal Antibodies Boost Fight Against Infectious Diseases
This is a great conversation w @Invivyd chairman and @politico today on why monoclonal antibodies will enhance our war against infectious diseases.

Why Managing Potency and Degradation Are Crucial in ADC Cleaning Validation
At INTERPHEX 2026, Paul Lopolito of STERIS highlighted the unique cleaning‑validation challenges posed by antibody‑drug conjugates (ADCs). He explained that ADCs merge highly potent small‑molecule payloads with large‑molecule biologics, creating divergent degradation profiles that demand product‑specific, risk‑based cleaning strategies. Lopolito...

Justin Rebo's KindBio Grows Organs On Demand
The Man Growing Organs On Demand Justin Rebo, KindBio and their fantastic sacks 🔗https://t.co/ed4bZbZV0T @Justin_Rebo https://t.co/nCffjjaqJH
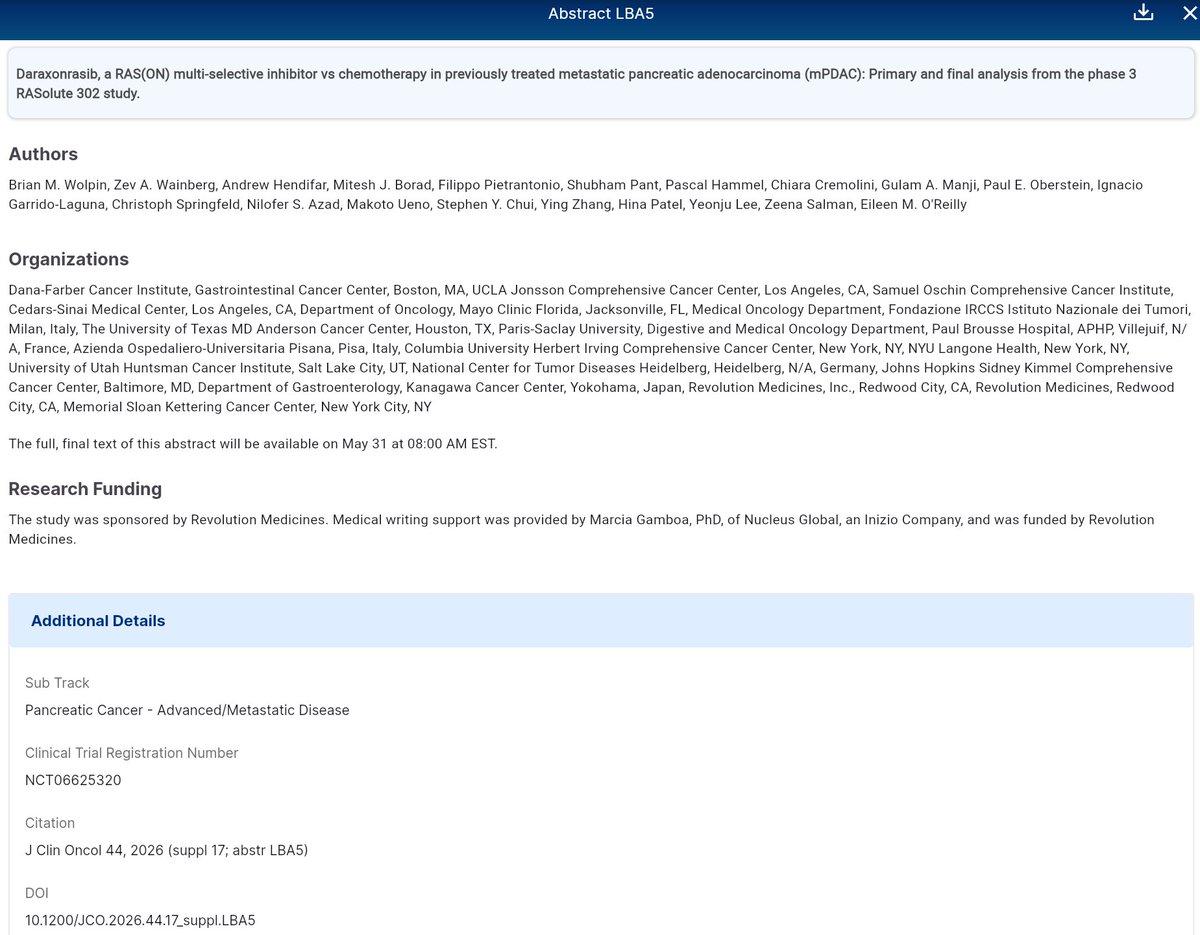
Daraxonrasib Beats Chemo in Pre‑treated Metastatic Pancreatic Cancer
#ASCO26 LBA5 - Wolpin - Daraxonrasib RAS(ON) multi-selective inhibitor vs chemo in prior Tx metastatic pancreatic adenocarcinoma (mPDAC) - Ph3 RASolute 302 [May 31, 2026] https://t.co/oWEeVo2oyq #NCT06625320 #pancsm #caxtx #PrecisionMedicine https://t.co/adz922fg5z

FDA Public Meeting on Increasing Access to Nonprescription Drugs - 04/23/2026
The FDA announced a public meeting on April 23, 2026 to discuss expanding access to nonprescription, or over‑the‑counter (OTC), drugs. The agency is soliciting comments through May 8, 2026, with an earlier deadline of Feb 2, 2026 to shape the agenda. Topics include identifying conditions that...
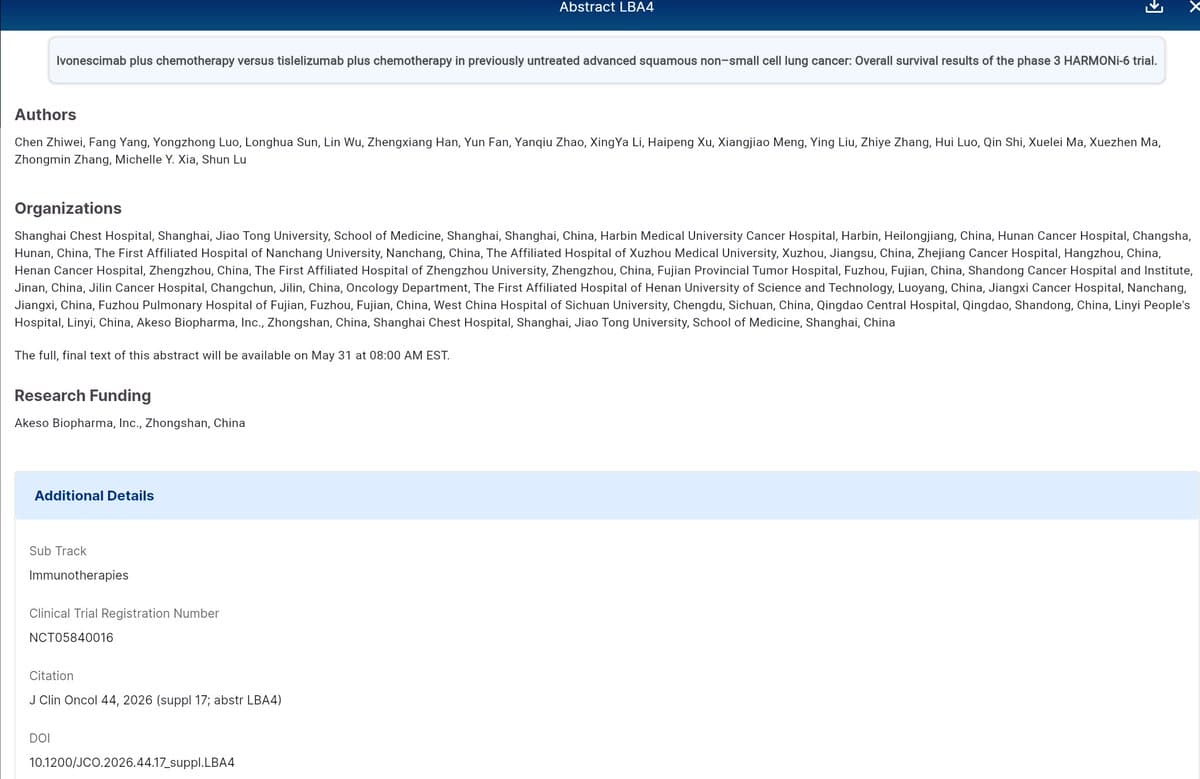
Ivonescimab Plus Chemo Rivals Tislelizumab in First‑line sqNSCLC
#ASCO26 LBA4 - Zhiwei - Ivonescimab + chemo vs tislelizumab + chemo 1L sq NSCLC - Ph3 HARMONi-6 [May 31, 2026] Zhiwei et al. https://t.co/oWEeVo2oyq #NCT05840016 #lcsm #ImmunoOnc https://t.co/NP2qr2gPuY
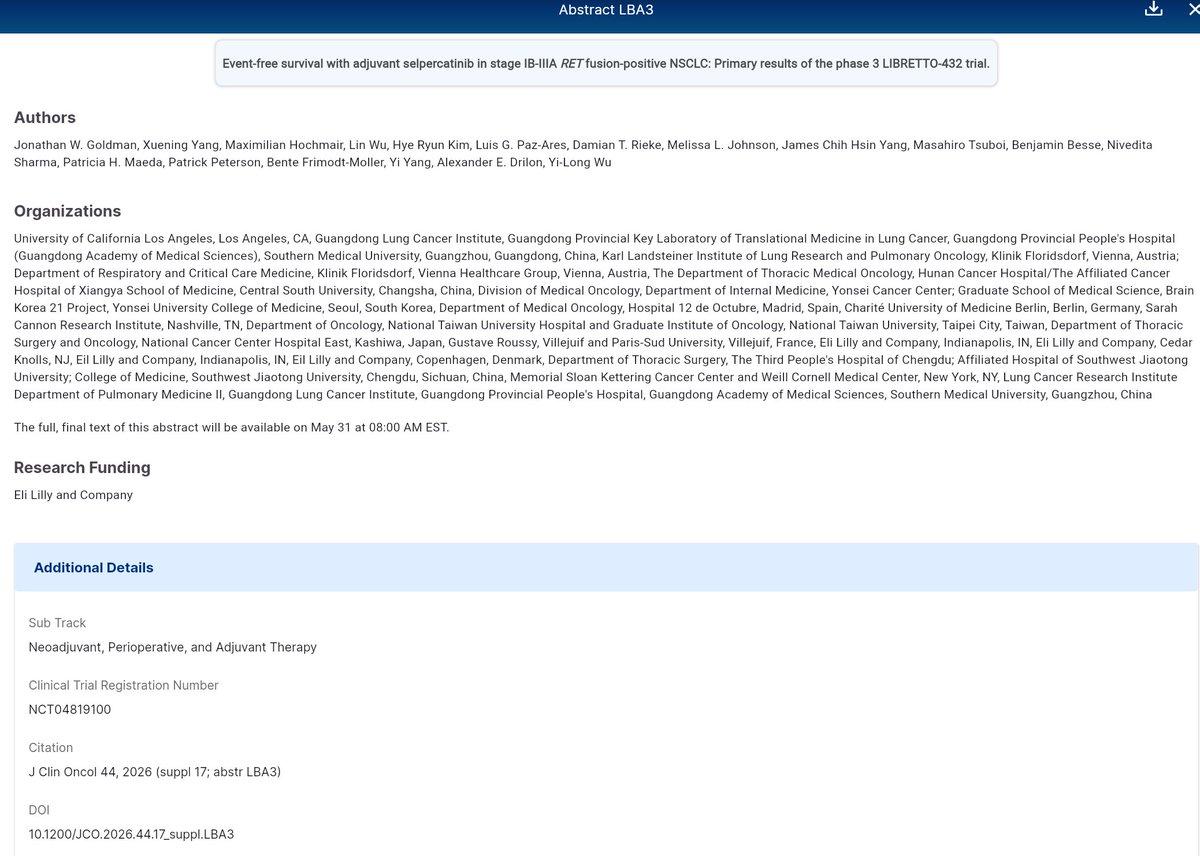
Adjuvant Selpercatinib Shows Promise
#ASCO26 LBA3 - Goldman - EFS w/ adjuvant sepercatinib in stage IB-IIIA RET fusion-positive NSCLC: Ph3 LIRETTO-432 trial https://t.co/oWEeVo2oyq #NCT04819100 #lcsm #PrecisionMedicine https://t.co/HWIS9mg57w

Phase 3 Trial Shows Abemaciclib Benefits Advanced Liposarcoma
#ASCO26 LBA2 - Dickson -SARC041: Ph3 DBRCT abemaciclib vs placebo in Pts w/ adv dedifferentiated liposarcoma [May 31, 2026] https://t.co/oWEeVo2oyq #NCT04967521 #scmsm https://t.co/uD6A9JmBCn

Genomic Profiling Boosts Survival for Advanced Sarcoma Patients
Using Tumor Genomic Profiling to Guide Treatment Selection Extends Survival in Advanced Sarcoma [May 10, 2019] @ASCO Daily News #scmsm #PrecisionMedicine https://t.co/8625ixTvQu

Perioperative
#ASCO26 LBA1 - Taplin - Perioperative APA + ADT - prostate CA [May 31, 2026] https://t.co/oWEeVo2oyq #NCT03767244 #pcsm https://t.co/8o91ntAwL9

Two Decades of Evolving ASCO Clinical Trial Trends
Trends and characteristics of clinical trials presented in ASCO plenary sessions (2011–2025) [] Overheu #ASCO26 Abstract # 11024, Poster Bd # 7 https://t.co/JYI0LY7FbU #ClinicalTrials #ctsm @ASCO