Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Atsena Advances XLRS and LCA1 Gene Therapies to Pivotal Phase 3 Trials
Atsena announced that its XLRS (ATSN-201) and LCA1 (ATSN-101) gene‑therapy programs are entering pivotal Phase 3 studies. The move follows 12‑month foveal schisis closure in 7 of 9 treated eyes and a sustained 20‑decibel improvement in dark‑adapted vision across 15 LCA1 patients, positioning the company for a 2028 BLA filing and a possible priority‑review voucher.

DOJ, DEA Reschedule Medical Cannabis Products
The U.S. Justice Department and DEA have reclassified FDA‑approved and state‑licensed medical cannabis products from Schedule I to Schedule III under the Controlled Substances Act. The change follows a 2025 Trump executive order and a 2024 DEA proposal that attracted 43,000 public...

Pharmaceutical Executive Daily: GSK Enters Collaboration to Support Launch of Bepirovirsen
GlaxoSmithKline (GSK) has signed a 5.5‑year exclusive partnership with Chia Tai Tianqing Pharmaceutical Group to launch bepirovirsen, a chronic hepatitis B candidate, across more than 5,000 Chinese hospitals. The drug is currently in priority regulatory review in mainland China, while GSK...

Spotlight On: Biosimilar Litigations - May 2026
The May 2026 Venable LLP update delineates which disputes qualify as biosimilar litigations. It includes lawsuits between biosimilar applicants or manufacturers and reference‑product sponsors, as well as conflicts among biosimilar firms themselves. It expressly excludes disputes solely between reference‑product sponsors, university‑sponsor...
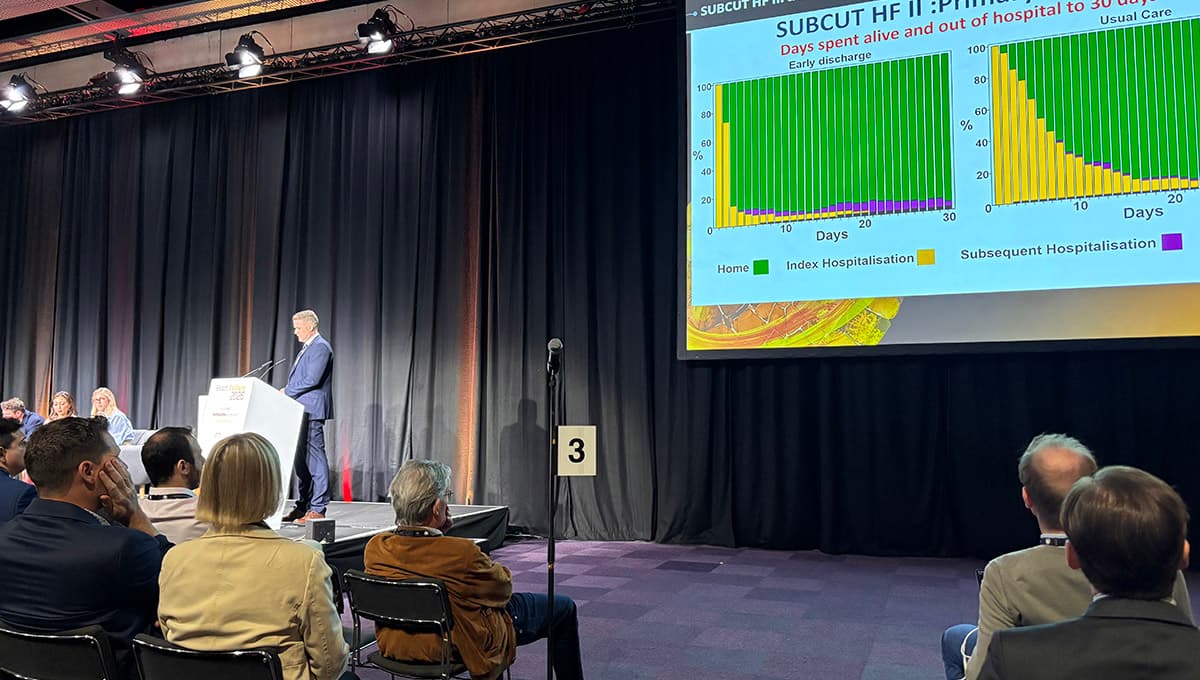
Subcutaneous Furosemide Aids With Earlier Discharge: SUBCUT II HF
A randomized SUBCUT II HF trial in 22 UK hospitals showed that subcutaneous furosemide delivered via a minipump enables safe early discharge for heart‑failure patients. The early‑discharge arm reduced average hospital stay from 11.0 to 5.6 days and added four additional days...

BIOTECanada Statement on Canada–Mexico Life Sciences Collaboration
BIOTECanada signed two memoranda of understanding with Mexico’s Asociación Mexicana de Laboratorios Farmacéuticos (AMILAF) and La Cámara Nacional de la Industria Farmacéutica (CANIFARMA), marking a new phase of life‑sciences collaboration. The MoUs were part of Mexico’s trade mission to Canada,...

A New Hantavirus Vaccine Is in the Works
Moderna announced that it is co‑developing an mRNA‑based hantavirus vaccine with Korea University’s Vaccine Innovation Center, a partnership that began in 2023. The effort follows a deadly outbreak on a Dutch cruise ship that killed three passengers and highlighted the...

FDA Alerts Health Care Providers and Patients About Increased Risk of New Blood Cancers with Tazverik (Tazemetostat) Use; Sponsor to...
The FDA has announced a voluntary market withdrawal of Tazverik (tazemetostat) after a Phase 1b/3 SYMPHONY‑1 trial revealed a 5.7% incidence of hematologic second primary malignancies (SPMs) in treated patients, compared with none in the control arm. The drug, approved in...

Is Longevity a $1.2 Quadrillion Opportunity?
Peter Diamandis released the 2026 Longevity Metatrend Report, a free 200‑page analysis of the rapidly advancing health‑span sector. The report highlights breakthroughs such as human trials of partial epigenetic reprogramming, AI‑engineered proteins achieving 50‑fold efficacy gains, and the first pig‑organ...

Dave Asprey Says Trump’s Psychedelics Order Could Reshape Men’s Mental‑health Biohacking
Biohacking pioneer Dave Asprey hailed President Donald Trump’s executive order to accelerate FDA review of psychedelic breakthrough therapies, saying it could finally give men a science‑backed way to break through stress and trauma. The move, he argues, shifts the biohacking...

FDA Issues Final Guidance on Post‑Approval Pregnancy Safety Data Collection
The U.S. Food and Drug Administration released final industry guidance on how drug sponsors should collect post‑marketing safety data for pregnant patients. The framework outlines registry design, real‑world evidence methods, and statistical standards, aiming to fill long‑standing data gaps and...

Advancing Aseptic Precision: How Modern Filling Lines Elevate Sterility, Flexibility, and Throughput
Modern pharmaceutical filling lines are adopting isolator‑based, fully automated systems that integrate washing, sterilization, filling, sealing, and environmental control. Automation and AI‑driven analytics are boosting sterility assurance, reducing human intervention, and meeting stricter EU GMP Annex 1 and FDA requirements. These...

Advancing Generic Drug Development: Bioequivalence Challenges for Patient-Centric Oral Formulations - 06/11/2026
The FDA’s Center for Drug Evaluation and Research hosted a two‑day workshop on June 11, 2026 to address bioequivalence challenges in patient‑centric oral generic drugs, including pediatric formulations. Experts discussed development hurdles for chewable tablets, orally disintegrating tablets, granules, suspensions, and sublingual...
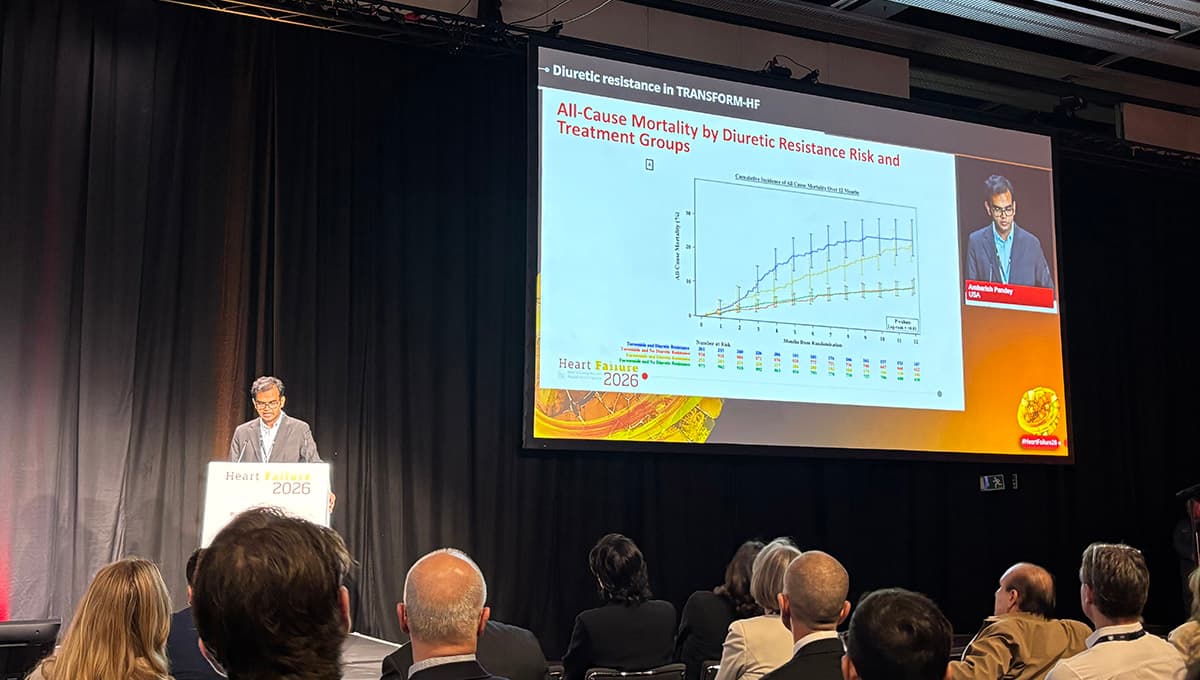
Diuretic Resistance Score Confirms TRANSFORM-HF’s Message
A post‑hoc analysis of the TRANSFORM‑HF trial applied the BAN‑ADHF risk score to identify patients at high risk of diuretic resistance. About 21% of the 2,421 participants scored above 12, indicating a markedly worse prognosis with 79% higher mortality and...

European Regulators Approve First‑In‑Human GLP‑1 Gene Therapy Trial for Fractyl Health
Fractyl Health received European regulatory approval to begin the first human trial of its GLP‑1 gene therapy, a novel approach that could replace chronic GLP‑1 injections. The clearance marks the inaugural clinical test of this gene‑editing strategy in Europe and...

In the Clinic for May 11, 2026
BioWorld’s May 11, 2026 “In the Clinic” roundup aggregates the day’s most relevant biopharma and med‑tech snapshots, special reports, and infographics. The page links to data‑driven analyses covering topics from mRNA vaccine research and China’s GLP‑1 landscape to pulsed‑field ablation for atrial...

Endometriosis Inspires Re-Examination of Known Targets at the Inaugural HERS Meeting
The inaugural Hormone Endometriosis Research Society (HERS) meeting used endometriosis as a lens to revisit established drug targets, revealing fresh therapeutic angles. Researchers presented data linking progesterone‑receptor modulators, anti‑inflammatory pathways, and the emerging biomarker GDF15 to disease regression. Genetic profiling...

New Home for Novo's Parkinson's Cell Therapy; GSK's Deal to Sell Drug in China
Novo Nordisk has transferred its early‑stage Parkinson's disease cell therapy to a specialized biotech partner, allowing the pharma giant to offload development risk while retaining royalty rights. GSK secured a distribution agreement to launch its flagship drug in China, opening...

CEL-SCI Enters Strategic Agreement with Amarox for the Registration, Commercialization, and Distribution of Multikine in Saudi Arabia
CEL‑SCI Corp. has signed a strategic partnership with Saudi firm Amarox to register, commercialize, and distribute its immunotherapy Multikine in Saudi Arabia, with a 50/50 revenue‑sharing model. Amarox will act as the local regulatory representative, seeking the Saudi Food and...
Avenacy Announces Launch of Ready-to-Use Tranexamic Acid in 0.7% Sodium Chloride Injection in the U.S. Market
Avenacy, a specialty injectable drug company, has launched a ready‑to‑use Tranexamic Acid 0.7% Sodium Chloride Injection in the United States. The product mirrors the active ingredient of Pfizer’s CYKLOKAPRON® and is indicated for short‑term use in hemophilia patients undergoing tooth...

Inhibrx Says Combo Therapy Shrank More Tumors than Merck's Keytruda Alone
San Diego‑based biotech Inhibrx reported that its experimental antibody INBRX‑106, when paired with Merck’s immunotherapy Keytruda, produced a higher rate of tumor shrinkage than Keytruda alone in patients with metastatic head and neck squamous cell carcinoma. The early‑stage study enrolled...
Therorna to Showcase Clinical-Ready Circular RNA in Vivo CAR-T and CircRNA Pipeline at the 2026 American Society of Gene &...
Therorna Inc. will present three posters at the 2026 ASGCT meeting, highlighting its lead in‑vivo CAR‑T candidate TI‑0032, a circRNA‑encoded CD19×CD3 T‑cell engager, and the HPV16 therapeutic vaccine TI‑0093. TI‑0032 has just entered a first‑in‑human investigator‑initiated trial for refractory autoimmune...
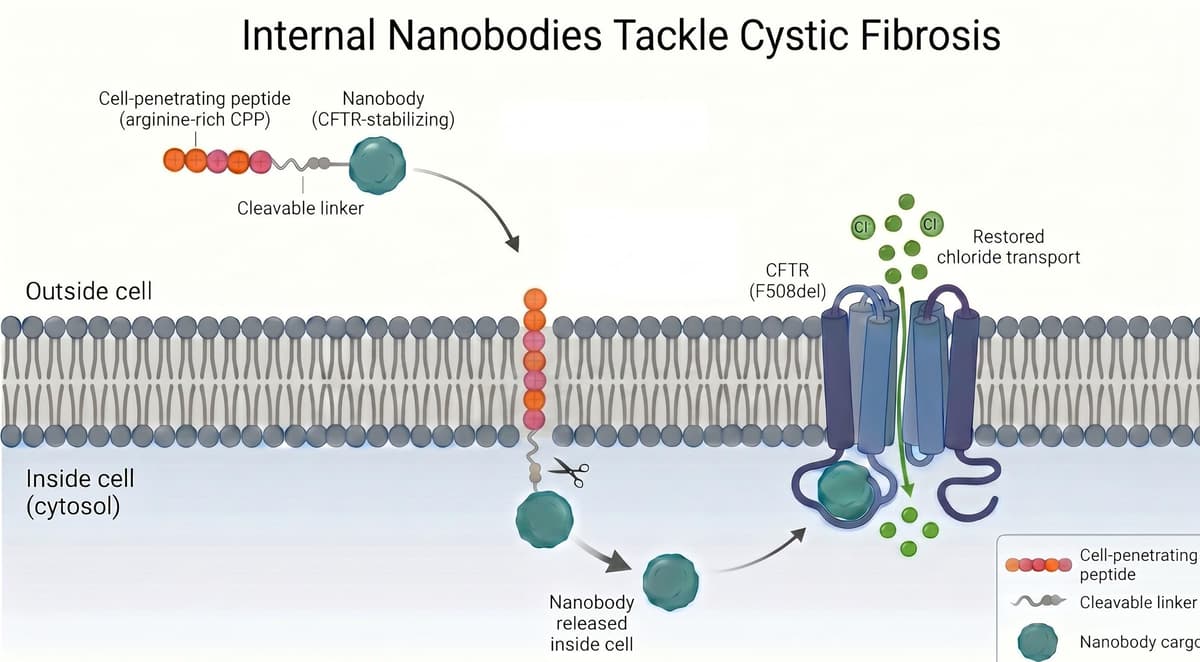
Internal Nanobodies Tackle Cystic Fibrosis
Researchers have engineered a cell‑penetrating nanobody that enters airway cells and stabilizes the misfolded CFTR protein responsible for cystic fibrosis. By fusing the nanobody to a ten‑arginine peptide, the hybrid molecule crosses the cell membrane and restores up to 90%...

Delos Capital and AP Biosciences Launch Collaboration to Create New Biotech Companies
Delos Capital and AP Biosciences have announced a strategic collaboration to launch and incubate new biotechnology companies focused on next‑generation antibodies. Through its Delos Foundry, Delos will lead therapeutic ideation, financing, and strategic development, while APBio supplies its proprietary antibody...

How MFN Impacts Drug Development and Launch Planning
President Trump’s Most Favored Nation (MFN) drug‑pricing proposals, still pending congressional approval, would cap Medicaid, Medicare Part B and Part D prices at the lowest GDP‑adjusted price among a list of 20 reference nations. The rule would force pharmaceutical companies to reassess...
Ocrevus Slows Disability Progression in Advanced PPMS, Trial Finds
A Phase 3 ORATORIO‑HAND trial involving more than 1,000 adults with primary progressive multiple sclerosis (PPMS) showed that Ocrevus (ocrelizumab) significantly slows disability progression. Over a median follow‑up of nearly three years, the drug reduced the risk of confirmed disability...
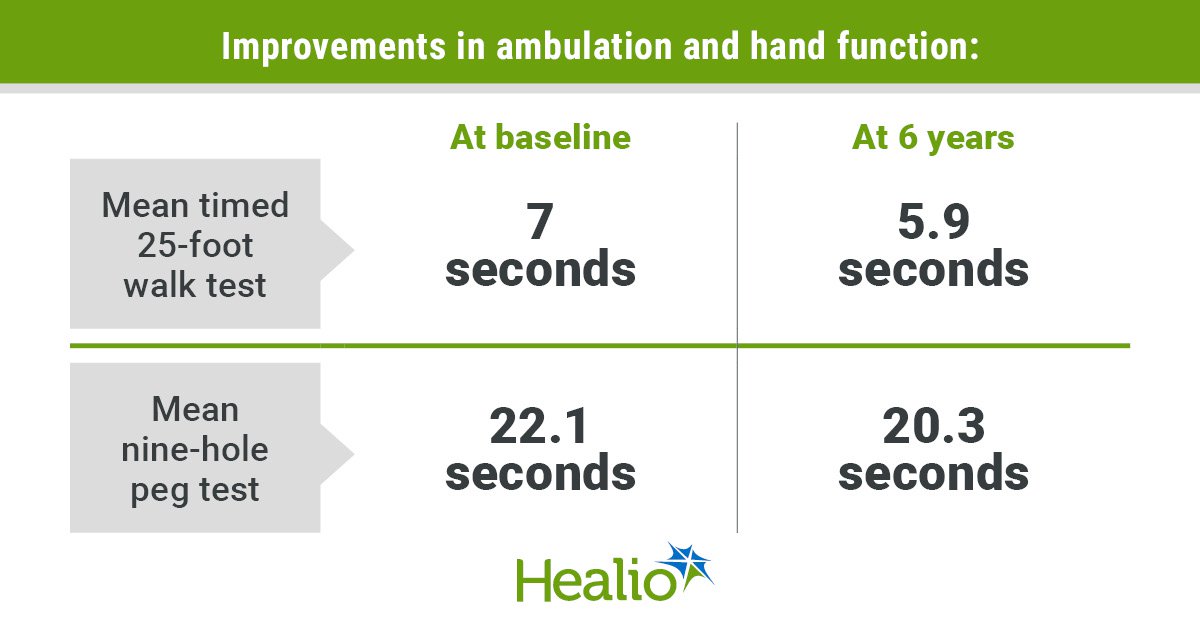
Ocrelizumab Preserves Ambulation, Hand Function in MS
A six‑year analysis of the ENSEMBLE trial shows that early‑stage relapsing‑remitting multiple sclerosis patients treated with ocrelizumab largely maintained functional ability. 86.1% preserved normal ambulation and 93% kept normal hand dexterity throughout the study, while 34% of those with baseline...

Braveheart Bio's Hengrui-Licensed Cardiac Drug Scores Second Clinical Win
Braveheart Bio announced that its heart‑muscle therapy, licensed from China’s Jiangsu Hengrui Pharmaceuticals, achieved its primary endpoint in a mid‑stage (Phase 2) trial for heart failure with reduced ejection fraction. The study reported an 8% absolute improvement in left‑ventricular ejection fraction...

Drug Repurposing
The FDA has issued a request for public comment on drug repurposing, seeking input from patients, clinicians, and researchers on priority disease areas and candidate drugs. The initiative builds on existing programs such as Project Renewal, which updates oncology drug...

The Next Frontier for Hantavirus: Finding Vaccines and Treatments
A hantavirus outbreak on a cruise ship has killed three people and sickened several others, marking the first documented person‑to‑person transmission of the disease. The strain’s heightened transmissibility has revived interest in vaccine and therapeutic candidates that were previously shelved...

Late Line RCC: Where Darlifarnib Fits and Why LITESPARK-012 Matters
At the International Kidney Cancer Symposium, Kura presented phase 1 data showing its next‑generation farnesyl transferase inhibitor darlifarnib combined with cabozantinib achieved a 44% objective response rate in clear cell renal cell carcinoma patients previously treated with cabozantinib. The cohort was...

Fractyl Secures Dutch CTA for First GLP‑1 Gene Therapy Trial
Fractyl Health today announced that it has received Clinical Trial Application (CTA) authorization in the Netherlands to initiate the Phase 1/2 first-in-human study of RJVA-001 - $GUTS first clinical candidate from its Rejuva 🧵👇 GLP-1 Gene Therapy platform and the...

Noncovalent Fragments vs WRN
Researchers at Merck and Proteros reported a noncovalent fragment‑based campaign against the Werner syndrome helicase (WRN), a synthetic‑lethal cancer target. Using a 1,020‑compound fluorine‑fragment library screened by 19F‑NMR and a separate 500‑compound SPR screen, they identified seven primary hits, three...

STAT+: Pharmalittle: We’re Reading About Medicare and Alzheimer’s Drugs, Estrogen Patch Shortages, and More
Medicare enrollment in the newly approved Alzheimer’s therapies Leqembi and Kisunla is dramatically lower than the federal government expected. The agency now projects minimal spending on the drugs through 2027, a sharp reversal from earlier forecasts of billions in annual...
CSL Ltd Slashes FY2026 Forecast, Flags $5 B Impairments as Shares Dive 20%
Interim CEO Gordon Naylor announced that CSL Ltd now expects FY2026 revenue of about $15.2 billion and underlying net profit of $3.1 billion, down from prior guidance. The company also disclosed roughly $5 billion in additional non‑cash impairments, triggering a 20% share plunge...

QP Essentials: Reducing Risk and Delays in EU and UK Trials
Clinical trial sponsors often encounter delays when shipping supplies to the EU and UK because Qualified Persons must certify each batch. Catalent’s webinar outlines the core QP responsibilities, highlights differences between EU and UK regulations, and offers practical steps such...
FDA Grants Fast Track Designation to Zai Lab’s DLL3-Targeting ADC for epNECs
Zai Lab’s DLL3‑targeting antibody‑drug conjugate zocilurtatug pelitecan received FDA Fast Track designation for extrapulmonary neuroendocrine carcinomas (epNECs). Early‑stage data showed a 38.2% objective response rate in heavily pre‑treated patients, indicating meaningful antitumor activity. The designation promises more frequent FDA interactions,...
Study Compares Two Antibiotics in Treating Severe Hemorrhagic Bacterial Pneumonia
Researchers at Osaka Metropolitan University compared cefiderocol (CFDC) and levofloxacin (LVFX) in a mouse model of severe hemorrhagic pneumonia caused by multidrug‑resistant Stenotrophomonas maltophilia. Both drugs significantly increased survival and reduced bacterial loads in lungs and heart relative to untreated...

Inside CSL’s Strategy to Simplify a Biotech Giant
CSL Limited is entering a strategic simplification phase, announced in August 2025, that will see its vaccine arm Seqirus spun off as a separate ASX‑listed company by the end of 2026 and a restructuring program that could cut up to 15% of...
Novo Nordisk microRNA Drug Fluffs Its Lines in Heart Failure
Novo Nordisk’s microRNA‑targeting drug CDR132L failed to demonstrate a statistically significant improvement in left ventricular end‑systolic volume index in the phase 2 HF‑REVERT trial. The study enrolled 280 post‑myocardial‑infarction patients with an ejection fraction of 45% or lower and elevated NT‑proBNP...

What the Six Hundred Billion Dollar MFN Headline Misses: The Best Price Carveout, the IQVIA Net Price Hole, and the...
The Council of Economic Advisers released a report projecting up to $600 billion in drug‑price savings over ten years, but the headline masks three critical flaws. First, the voluntary MFN agreements rely on the GENEROUS carve‑out that isolates supplemental rebates from...

GLP‑1 Cash‑pay Users Set to Triple by 2030
In its initiation on $HIMS, The J.P. Morgan healthcare team estimates the long term GLP-1 cash pay addressable market to be over 120M individuals in the US, with only ~3M of those currently utilizing compounded or branded GLP-1s (~2.5% penetration)....

Dog Drug Flavor Loved, but Owners Smell Vomit
from the trenches of dog drug development: last week we had to change the flavoring in one of our drugs because, while the dogs LOVE it, to dog owners it smells like ........ vomit

LQDA’s Soaring Market Share Proves Drug Superiority
$LQDA "Market share has gone from 10% to 16% to 23% across past 3 quarters" That doesn't happen against an entrenched competitor unless the drug is truly better.

GLP‑1 Drugs Boost Survival, Cut Recurrence in Obese Breast Cancer Patients
In women with breast cancer and obesity or T2 diabetes, a large propensity matching retrospective analysis reports association of GLP-1 drug therapy with improved survival and reduced risk of recurrence https://t.co/GjimCr97S0
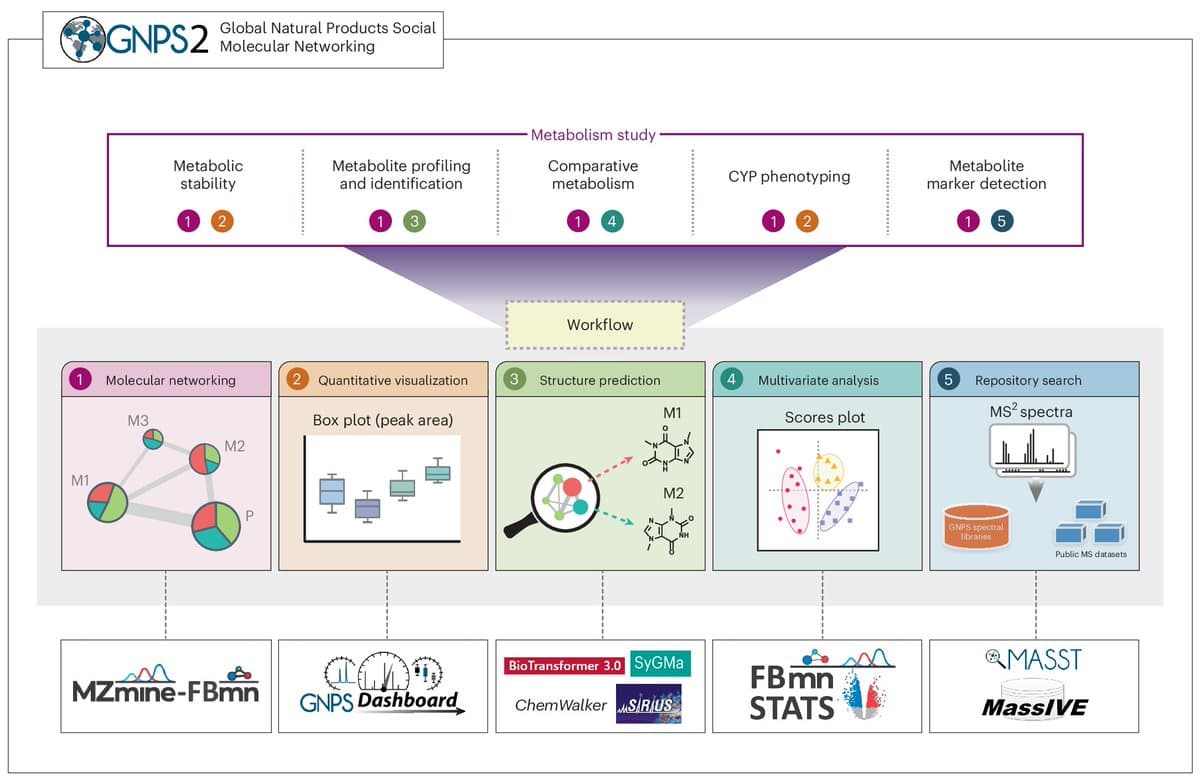
GNPS2 Enables Comprehensive Drug Metabolism Toolkit
Nature Protocols: A versatile toolkit for drug metabolism studies with GNPS2: from drug development to clinical monitoring https://t.co/lEejrO6gXT https://t.co/qlybcRgD9V

Beware Payer Panic; GLP‑1 Drugs Defy Bankruptcy Predictions
This is a reminder: when payers warn a new class of drugs will bankrupt a system, be cautious. We saw this with PCSK9 cholesterol meds, and then with Alzheimer's drugs. Unless of course that class of drugs begins with G and...

Rare Disease Community Gains New Treatment Hope After Five Years
Five years after disaster, a rare disease community gets new chance at treatment Yet another heart-rending story from @Jasonmmast. https://t.co/9vgOKpInL3
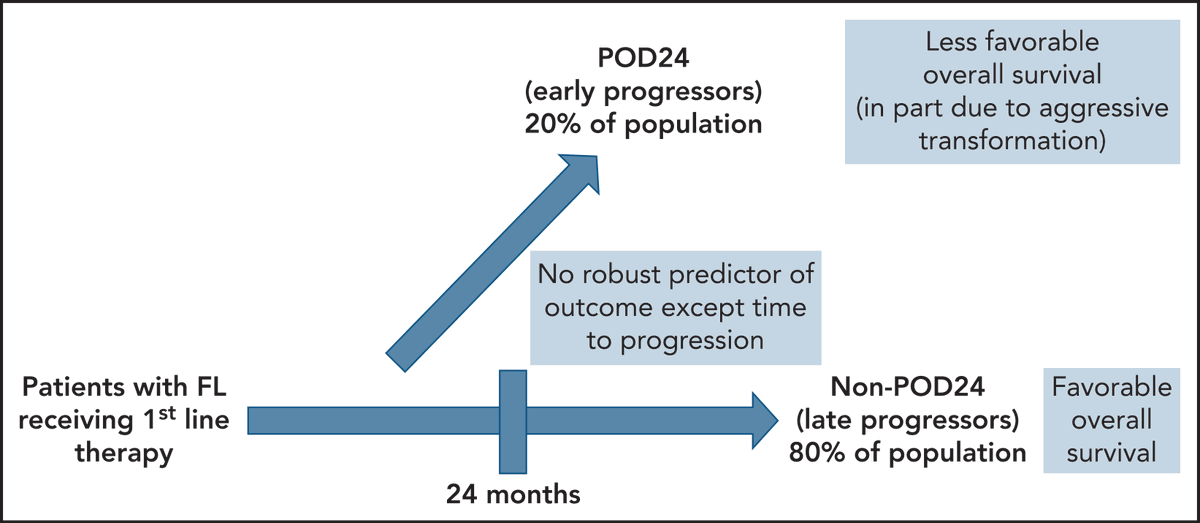
POD24 Signals Urgent Need for Wise Management
POD24 in follicular lymphoma: time to be “wise” [Mar 17, 2022] @JohnPLeonardMD @BloodJournal https://t.co/jLMd14D4Ur #lymsm https://t.co/991dNQe3JH
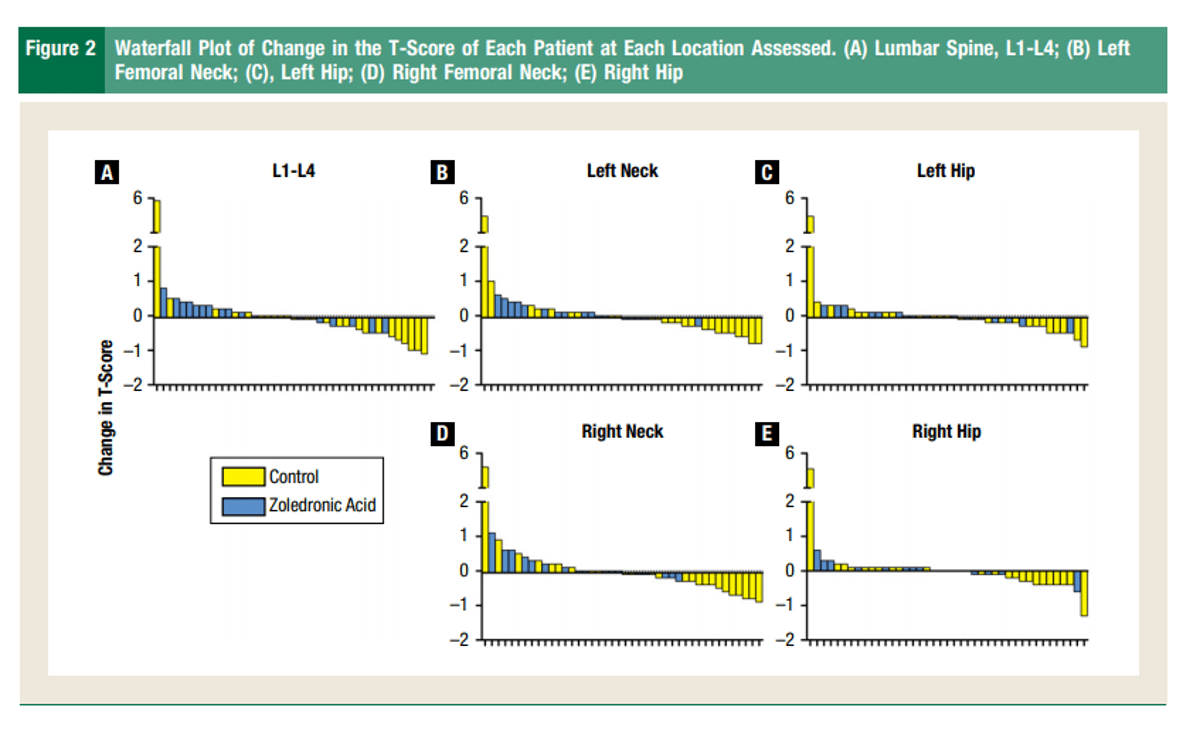
Zoledronic Acid Prevents Bone Loss in Lymphoma Therapy
Zoledronic Acid for Prevention of Bone Loss in Patients Receiving Primary Therapy for Lymphomas: A Prospective, Randomized Controlled Phase III Trial [Dec 29, 2012] @Lymphoma_Doc @mtmdphd et al. CLML https://t.co/iPOYSXEA1M #lymsm #SuppOnc #BoneHealth #NCT00352846 https://t.co/UlaqHyy65d