Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Merck Announces First Dose in Phase 3 Study with Enpatoran for Lupus Patients with Active Skin Manifestations
Merck announced the first patient has been dosed in its global Phase 3 ELOWEN program, testing the oral TLR7/8 inhibitor enpatoran in lupus patients with active skin manifestations. The double‑blind, placebo‑controlled studies—ELOWEN‑1 and ELOWEN‑2—will enroll roughly 200 participants each across 266 sites in 26 countries, with the primary endpoint measuring change in the CLASI‑A skin score. Enpatoran previously showed clinically meaningful skin improvements in Phase 2, positioning it as a potential first‑in‑class therapy for cutaneous lupus. Merck reported 2025 sales of €21.1 billion (about $23 billion).

FDA Proposes Excluding Novo, Lilly Weight Loss Drugs From Bulk Compounding List in Win for the Companies
The FDA has proposed removing the active ingredients of Novo Nordisk’s semaglutide and liraglutide and Eli Lilly’s tirzepatide from the bulk‑compounding list used by 503B outsourcing facilities. If the rule is finalized, these high‑demand obesity and diabetes drugs could only be...

Irisin Hormone Reverses Obesity and Insulin Resistance
Irisin, a hormone released by muscle during exercise, reverses obesity and insulin resistance in mice -- without cutting food intake or causing muscle loss. As a medical school professor, I find this striking. We have spent a decade asking how to...

CGT Global Appoints Charlotte Ivancic to Board of Advisors, Strengthening Strategic Leadership in Cell and Gene Therapy Policy and Market...
CGT Global announced the appointment of Charlotte Ivancic to its Board of Advisors, bringing over 25 years of federal health policy and legislative experience. Ivancic, a partner at FGS Global and former senior aide to Senate Majority Leader Bill Frist,...

What We Know (and Don’t Know) About Peptide Safety
Peptide safety is neither automatically assured by mimicking endogenous hormones nor inherently hazardous because research is incomplete. The risk profile depends on the specific peptide, the depth of clinical evidence, product purity, and real‑world usage conditions. Early‑phase trials provide maximum...

GLP-1s May Prevent Incident AF, Series of Studies Shows
Observational analyses presented at Heart Rhythm 2026 suggest GLP‑1 receptor agonists, including tirzepatide, lower the risk of incident atrial fibrillation by 33%‑47% across diverse patient groups. The benefit appears consistent in individuals with or without diabetes, obesity, and chronic kidney...

Gene Therapy Soon as Routine as Everyday Surgery
Eric Kelsic envisions the future of gene therapy will be viewed in the “the same way that many of us have had some form of surgery at certain points in our life.” As therapies become safer and more effective, the decision...

Why Protocol Complexity Keeps Increasing – and How to Fix It
Protocol complexity in late‑stage trials is spiraling, with Phase III studies now averaging about 6 million data points—a rise of roughly 11% each year. The growth is fueled by scientific advances, global regulatory demands, and the ease of capturing extra data via...
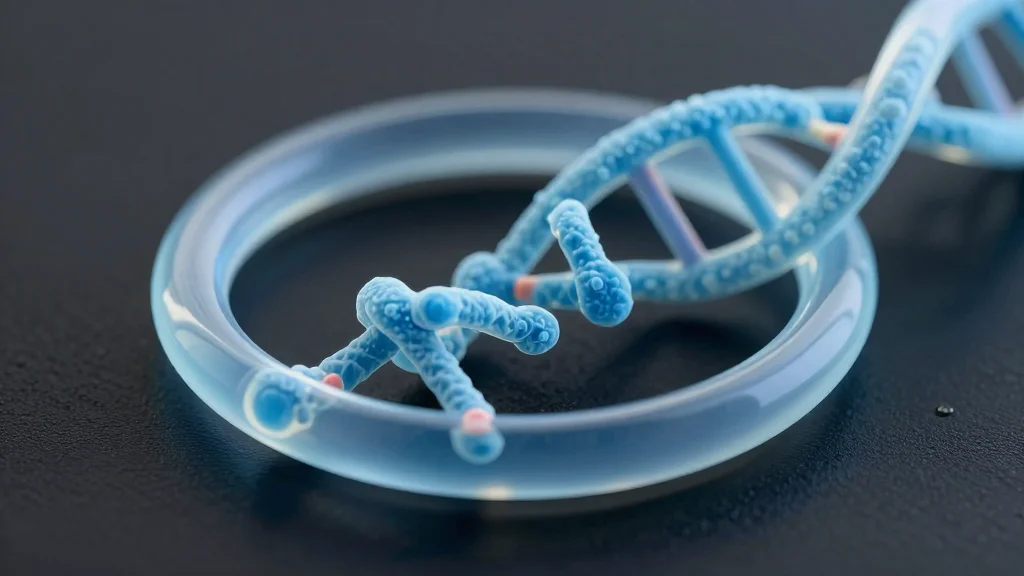
DNA Nano‑Ring Precisely Captures and Orients Membrane Proteins
A new DNA nano-ring platform enables precise capture and orientation of individual membrane proteins, advancing the study of these vital cellular gatekeepers and opening new possibilities in medicine, imaging, and synthetic biology. nanotechnology

New GLP‑5 Study Is Only Mouse
A friendly reminder, just because that new Nature study on a quintuple agonist (LOL GET READY FOR "GLP-5" discourse) is out today: It's a study on MICE. MICE TESTING IS NOT HUMAN TESTING. IT'S JUST MICE.

The FDA Real Time Clinical Trial Announcement Quietly Dissolves Phase Gates, Breaks Biotech Capital Markets Plumbing, and Opens a Founder...
The FDA announced on April 28 that real‑time clinical trials (RTCTs) are now live, with AstraZeneca’s phase‑2 lymphoma study and Amgen’s phase‑1b small‑cell lung cancer trial streaming data through Paradigm Health. The agency claims a 20‑40 % reduction in development timelines, but...

Trump's Psychedelic Order Preserves FDA Oversight Balance
My op ed in today's @WashPost - "Trump’s executive order on psychedelics strikes a healthy balance: The president preserved the FDA's role, even as some want the agency's gatekeeping dismantled." https://t.co/9hH7kz107J
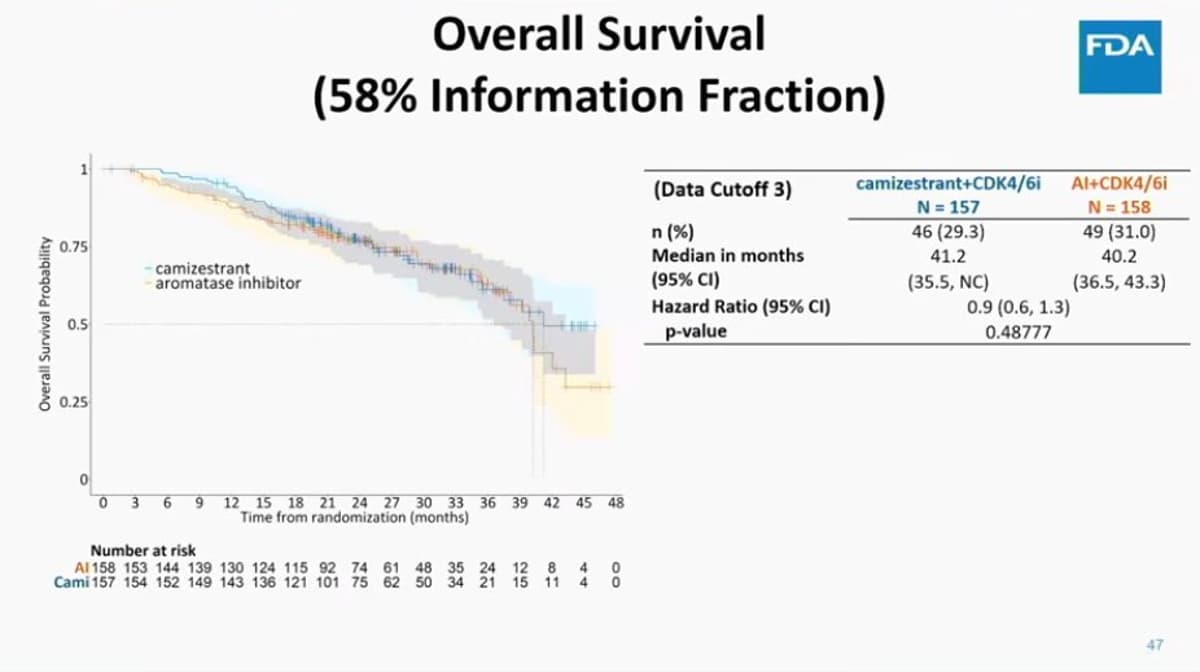
Serena-6 Shows Unexpected Immature OS Curves
$AZN Serena-6 immature OS curves as revealed at the adcom. Not sure we've seen these before. https://t.co/L9oAQxhrh9

Clinical Trial Finds Rapamycin Undermines Exercise Gains in Older Adults
Researchers led by Brad Stanfield reported that a 13‑week, randomized trial of 40 sedentary older adults in New Zealand found rapamycin blunted the physical benefits of a modest home‑exercise program. Participants taking the drug showed weaker strength, more fatigue and...

Year Two Funding Crisis: Grants Dry, Competition Rises
With federal research funding cuts, the first year of Trump 2.0 was tough. Year 2 is in some ways grimmer, a @statnews survey shows. Bridge funding is drying up, existing grants are ending & competition for fewer new grants is...
Akeso’s ASCO Plenary Raises Questions on Lung Cancer Data
This week's Biotech Scorecard: An #ASCO26 mystery: What does Akeso’s primo plenary spot say about its ivonescimab lung cancer survival data? $SMMT If the news was good, we'd have heard by now, right? Or, is Akeso just being ultra conservative?...

STAT+: Pharmalittle: We’re Reading About a Supreme Court Hearing on ‘Skinny Labels,’ AstraZeneca U.K. Expansion, and More
AstraZeneca announced a $400 million investment to complete the Rosalind Franklin building in Cambridge and launch a new digital‑focused laboratory in Macclesfield, reviving UK R&D expansion after the US‑UK trade deal lifted NHS cost‑effectiveness thresholds. Meanwhile, the U.S. Supreme Court heard...

FDA Grants Priority Review to BeOne’s TEVIMBRA Combo for HER2+ Gastric Cancer
The U.S. Food and Drug Administration has granted priority review to BeOne Medicines’ TEVIMBRA (tislelizumab) combined with ZIIHERA (zanidatamab) and chemotherapy for first‑line treatment of HER2‑positive gastroesophageal adenocarcinoma. The decision follows a Phase 3 trial that showed a median overall survival...

Chiesi to Acquire KalVista for $1.9 B, Adding First Oral HAE Therapy
Chiesi Group announced a $1.9 billion cash deal to buy KalVista Pharmaceuticals, paying $27 per share—a 36% premium—to acquire the FDA‑approved oral hereditary angioedema therapy Ekterly. The transaction, slated to close in the third quarter of 2024, marks Chiesi’s largest acquisition...

Pfizer Unveils Nanoparticle Platform Aimed at Precision Cancer Therapy
Pfizer disclosed a nanotechnology‑based drug‑delivery platform that uses engineered nanoparticles to target tumors with high precision. The initiative, led by Puja Sapra of the Targeted Therapeutics Unit, seeks to reduce collateral damage to healthy cells and could reshape oncology pipelines.
GSK's EXDENSUR Secures FDA Approval with Groundbreaking Twice‑Yearly Dosing for Severe Asthma
GlaxoSmithKline's depemokimab, branded EXDENSUR, received FDA approval on Dec. 16, 2025 as an add‑on therapy for severe eosinophilic asthma. The biologic is administered subcutaneously at 100 mg once every six months, a dosing interval not seen before in this therapeutic class. Approval was...

Avalyn Pharma Prices $300 Million IPO at $18 per Share, Expanding Biotech B2B Financing
Avalyn Pharma announced the pricing of its upsized initial public offering, selling 16.67 million shares at $18 each to raise $300 million. The Nasdaq‑listed biotech will use the proceeds to advance its pulmonary‑fibrosis pipeline and deepen B2B collaborations across the pharmaceutical supply...

Five Companies Pushing Biotech in Scotland
Scotland’s life‑science sector now comprises roughly 750 companies and 46,000 employees, anchored by strong university spin‑outs in Edinburgh, Glasgow and Dundee. Within this ecosystem five biotech firms are drawing attention: Cumulus Oncology’s portfolio‑driven oncology asset creation, EnteroBiotix’s microbiome‑based IBS therapy...

BeOne Receives the US FDA Priority Review to Tevimbra Combination for 1L Treatment of HER2+ GEA Cancers
The U.S. FDA granted priority review to BeOne Medicines’ Tevimbra‑Ziihera‑chemotherapy regimen for first‑line treatment of unresectable HER2‑positive gastric, gastro‑esophageal junction and esophageal adenocarcinomas. The decision follows the Phase III HERIZON‑GEA‑01 trial, which enrolled 914 patients and compared the new combos against...

Canada First in G7 to Approve Generic Ozempic
Health Canada has granted approval for the first generic version of semaglutide, the active ingredient in Ozempic, making Canada the first G7 nation to do so. The generic injection, submitted by Dr. Reddy’s Laboratories, is indicated for once‑weekly treatment of adult...

Specialty Drugs Now Consume over Half of Total Drug Spend
Specialty drugs now represent more than half of total prescription spending, despite treating a relatively small patient pool. A 2024 Pharmaceutical Strategies Group survey shows 43% of employers and health‑plan leaders cite managing specialty drug costs as their top priority,...
RAAS Inhibitors Show Benefits in Pediatric CKD Vs. CCBs
A new comparative‑effectiveness study of 2,762 children with chronic kidney disease (CKD) found that renin‑angiotensin‑aldosterone system (RAAS) inhibitors significantly outperformed calcium‑channel blockers (CCBs) in preventing progression to dialysis or transplant. Over a two‑year follow‑up, RAAS users had a 42% lower...

NDMA: A Cancer-Causing Contaminant in Meds and Meat
The FDA has identified the probable carcinogen N‑nitrosodimethylamine (NDMA) in several high‑volume drugs—including valsartan, Zantac (ranitidine) and certain metformin formulations—prompting recalls and market withdrawals. NDMA exposure limits are set at 96 nanograms per day, yet a half‑chicken breast cooked by...
Doubts Cast over 'Wild' Claim that Magnetic Control Can Turn on Genes
Researchers in South Korea announced a magnetically controlled switch that can turn on genes inside cells using an electromagnetic signal, a development touted as a potential breakthrough for non‑invasive therapies. The study appeared in a leading journal but has been...

Trump Administration Move to Reclassify Cannabis Sparks Confusion
The Trump administration issued an order that partially reschedules cannabis, moving products sold under state medical licenses and prospective FDA‑approved cannabis drugs from Schedule I to Schedule III. The change does not affect existing FDA‑approved cannabis pharmaceuticals and leaves the broader recreational...

EXPLAINER: Medicine's Forgotten Biomarker - The Homocysteine Story Your Doctor Missed
In this episode Dr. Robert Lufkin explains why homocysteine, an amino‑acid by‑product of methionine metabolism, is a powerful but overlooked risk factor for heart disease, stroke, and dementia. He reviews the biological mechanisms by which elevated homocysteine damages arterial endothelium...

Symptom Progression Slowed in Lewy Body Dementia with Zervimesine
Cognition Therapeutics reported that the oral, brain‑penetrant small‑molecule zervimesine slowed symptom progression in a phase 2 SHIMMER trial of 130 adults with mild‑to‑moderate dementia with Lewy bodies (DLB). Over six months, patients receiving 100 mg or 300 mg daily showed 52‑86% slower decline...
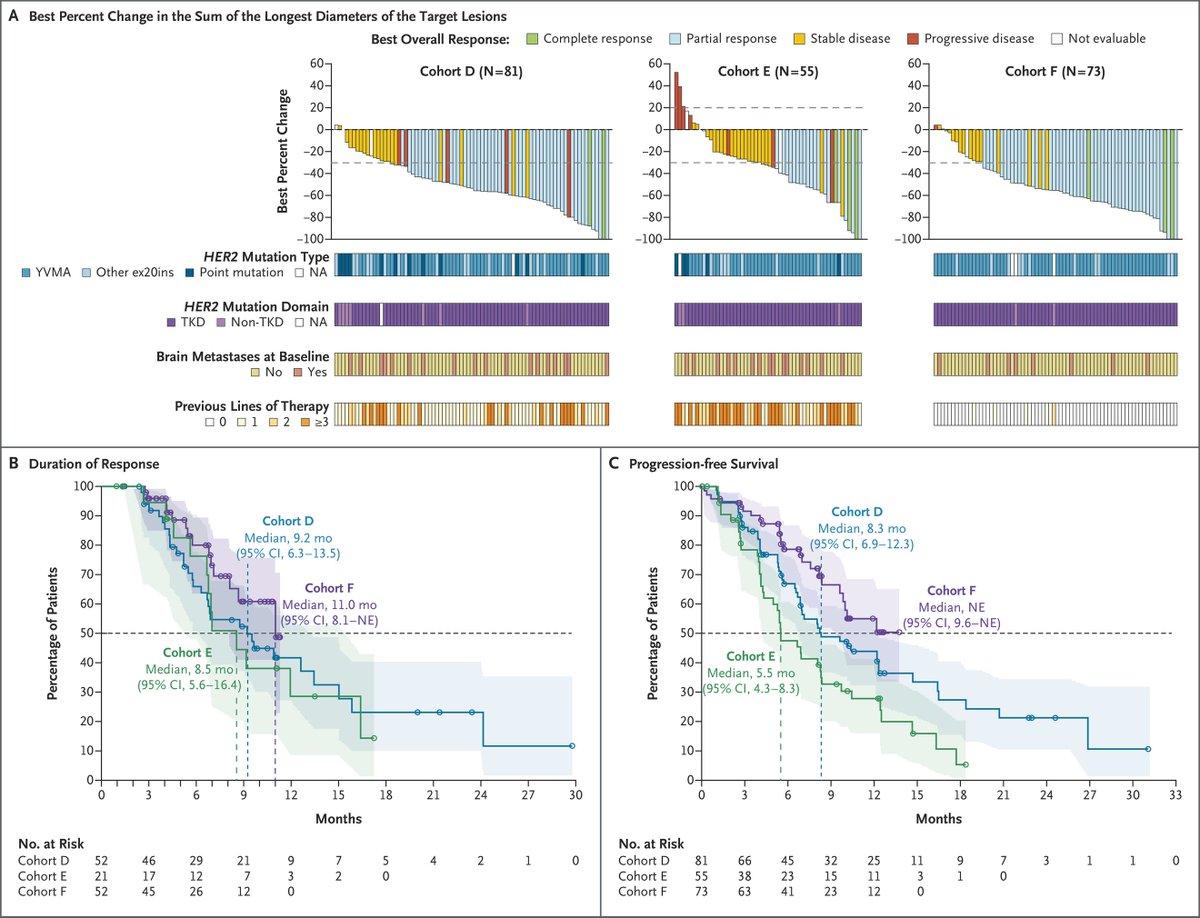
Sevabertinib Shows
EGFR’s Poor Sibling {Editorial} [April 15, 2026] @DCarboneMD @NEJM https://t.co/DjV4gAtKMk #lcsm #PrecisionMedicine RE: #NCT05099172 Sevabertinib in Advanced HER2-Mutant Non–Small-Cell Lung Cancer https://t.co/63kCdZ6JOJ

FDA Slowdown Threatens AZN's SERD Lead as Roche Advances
FDA just pumped the brakes hard on AZN's camizestrant. While the SERD track is crowded and Roche is picking up speed, AZN's innovation may be derailed by regulatory conservatism: https://t.co/9eroTXv5dc https://t.co/rOJwh9iMCz

Laguna Biotherapeutics Reports the US FDA IND Clearance for LGNA-100 to Treat High-Risk Pediatric Leukemias
Laguna Biotherapeutics announced that the U.S. Food and Drug Administration has granted IND clearance for its lead candidate LGNA-100, allowing the company to begin a Phase‑I single ascending dose trial. The study will evaluate safety, tolerability and mechanism of action...

FDA Ends 9-Month Advisory Committee Drought with AstraZeneca Oncology Panels
The U.S. Food and Drug Administration announced it will hold advisory committee meetings for two AstraZeneca oncology applications, breaking a nine‑month gap in drug‑focused panels. Experts say the restart restores a critical venue for independent scientific input and public transparency.

Hemab Therapeutics (COAG) IPO Deck
Hemab Therapeutics, a clinical‑stage biotechnology firm, announced its IPO deck on April 30, 2026. The company focuses on innovative therapies for rare blood‑coagulation disorders, including both bleeding and thrombotic conditions. Its pipeline features two Phase 2 candidates targeting hemophilia and antiphospholipid syndrome, and...

Merck Beats Quarterly Sales Expectations as Keytruda Fuels Growth
Merck reported first‑quarter sales of $8 billion, driven by a 12% increase in Keytruda revenue, surpassing analyst estimates of $7.6 billion. The company posted a loss after taking a $3.62‑per‑share charge related to its acquisition of antiviral developer Cidara Therapeutics. Shares jumped...

Xtalks Featured Member: Parminder Singh Bhasin, Senior Director, Head of Analytical Research and Project Management, Intech Biopharm. Ltd
Parminder Singh Bhasin, Senior Director at Intech Biopharm Ltd., has been highlighted as an Xtalks Featured Community Member. With more than 26 years in pharmaceutical R&D, he oversees analytical research, method validation, and project management for complex generics, pMDI, nasal...

Huahui Health and BeOne Medicines Partner for HH160 in a ~$2.02B Deal
Huahui Health and BeOne Medicines have signed a global exclusive option, license and collaboration agreement for HH160, a novel trispecific antibody targeting oncology immunotherapy. Huahui will receive $20 million upfront, $100 million when BeOne exercises its option, and up to $1.9 billion in...

A Combination Treatment Is Claimed to Produce Sizable Life Extension in Aged Mice
Seragon funded a pre‑clinical trial of SRN‑901, a proprietary oral cocktail that blends urolithin A, quercetin, nicotinamide riboside, alpha‑lipoic acid and the company’s SRN‑820. In 18‑month‑old mice on a Western diet, the regimen extended median remaining lifespan by 33% and cut...

Real World Applicability of Ivermectin vs Permethrin Trial for Scabies
A recent cluster‑randomised trial found oral ivermectin more effective than 5% permethrin cream for treating classic scabies under controlled conditions. The study, however, enrolled participants from well‑resourced health centres and excluded severe dermatological cases, raising questions about its relevance to...
Blackstone Life Sciences Puts $400 Million Behind Teva’s Autoimmune Drug Duvakitug
Blackstone Life Sciences has pledged $400 million to Teva Pharmaceutical to advance duvakitug, a monoclonal antibody targeting TL1A in Phase 3 trials. The deal signals private‑equity confidence in Teva’s shift from generics to innovative biologics and could reshape capital flows in the...

Reid Hoffman Backs Manas AI to Accelerate Cancer Drug Discovery with Frontier Models
At a WIRED Health event in London, Reid Hoffman championed Manas AI’s AI-driven platform that aims to shrink cancer drug discovery from a decade to a few years. He urged physicians to treat frontier AI models as a mandatory second...
Regeneron Secures First Gene Therapy Approval, Reports $3.6B Q1
Regeneron announced FDA approval of Otarmeni, the first gene therapy for hearing loss, and reported first‑quarter revenue of $3.6 billion, a 19% year‑over‑year rise. The approval, offered free to U.S. patients, underscores a new therapeutic frontier while blockbuster drugs Dupixent and...

EU’s Joint Clinical Assessment System Still Finding Its Footing
The European Union’s Joint Clinical Assessment (JCA) system, launched in January 2025, saw its first activation with Ipsen’s Ojemda approval, initiating a 30‑day countdown for a centralized clinical report. Designed to replace duplicated health‑technology assessments across member states, the JCA currently...
ADM Research Highlights Microbial Benefits for Psychological Symptoms
A randomized, double‑blind trial examined live and heat‑inactivated probiotic strains in healthy adults with self‑reported mild anxiety. The 12‑week live blend of Bifidobacterium longum CECT 7347 and Lactobacillus rhamnosus CECT 8361 failed to meet primary anxiety endpoints, though it preserved butyrate‑producing bacteria....

Northwestern Chemist Launches Science-Driven Biotech Blog
New biotech blog by a Northwestern medicinal chemist -- debut post is a well-written, science-focused take on $CCCC. Check it out: https://t.co/4Pc5wUUKlO

Seaport Therapeutics (SPTX) IPO Deck
Seaport Therapeutics, a clinical‑stage biotech firm, is preparing an initial public offering to fund its oral drug program targeting major neuropsychiatric conditions such as depression and anxiety. The company’s IPO deck emphasizes a “proven path” in neuroscience, highlighting pre‑clinical data...

The Biotech Bi-Weekly: A Virtual Biology Initiative, a New Discovery Grant and a Protein Supplier to Watch in Cancer Research...
The biotech bi‑weekly highlights a wave of new funding and tools, starting with Biohub’s $500 million five‑year Virtual Biology Initiative to generate global multimodal datasets for predictive biology. Zymo Research launched the Fecal Microbiome Discovery Grant to support early‑stage researchers, while...