Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

The BioPharm Brief: Metabolic Phase III Progress, HER2 Oncology Momentum, and US Manufacturing Expansion Drive Industry Scale
Zealand Pharma and Roche are moving petrelintide, an amylin analog, into Phase 3 trials for chronic weight management, with enrollment slated for the second half of 2026 after earlier studies showed double‑digit weight loss and tolerability comparable to placebo. The FDA granted priority review to a HER2‑targeted therapy for gastroesophageal adenocarcinoma, accelerating its path to market based on Phase 3 data showing improved survival. Novartis completed its seventh U.S. manufacturing site, part of a multibillion‑dollar plan to boost domestic capacity for APIs, biologics and advanced therapies, enhancing supply‑chain resilience. Together, these moves illustrate how clinical progress, regulatory acceleration, and manufacturing scale are converging to meet rising global demand for complex biologics.

HYTN Successfully Exports GMP-Manufactured Vape Cartridges
HYTN Innovations announced the export of 1,000 one‑gram live‑resin vape cartridges manufactured under Good Manufacturing Practices to Australia’s regulated medical cannabis market. The shipment represents the company’s first full commercial batch of finished, non‑sterile cannabis‑oil products, following a Health Canada‑approved...

Drug Trials Snapshots: BIMZELX
UCB’s BIMZELX, a dual IL‑17A/F monoclonal antibody, received FDA approval on Oct 17 2023 for adults with moderate‑to‑severe plaque psoriasis. The approval was based on two phase‑3 placebo‑controlled trials that enrolled 839 patients across 13 countries, with a demographic profile of 72%...

Drug Trials Snapshots: AGAMREE
Santhera Pharmaceuticals received FDA approval on October 26, 2023 for AGAMREE (vamorolone), a corticosteroid designed to treat Duchenne muscular dystrophy (DMD) in patients aged two years and older. The approval rests on a multinational, double‑blind, 24‑week trial of 121 male patients that...

Drug Trials Snapshot: TALVEY
TALVEY (talquetamab‑tgvs), a bispecific antibody from Janssen Biotech, received FDA approval on August 10 2023 for adult patients with relapsed or refractory multiple myeloma who have exhausted at least four prior therapies. The pivotal trial enrolled 339 participants across 47 sites, with...

Drug Trials Snapshots: OJJAARA
OJJAARA (momelotinib) received FDA approval on September 15 2023 for adults with myelofibrosis‑related anemia. The approval rests on two pivotal trials—MOMENTUM, which enrolled JAK‑inhibitor‑experienced patients, and SIMPLIFY‑1, which enrolled JAK‑naïve patients. In MOMENTUM, 25% of OJJAARA recipients achieved a ≥50% reduction in...

There Are Two GLP-1 Side Effects Your Doctor Doesn’t Know About, and They Can Affect Your Workouts
A new *Nature Health* study used AI to scan 400,000 Reddit posts, finding that roughly 70,000 users were taking GLP‑1 drugs such as Ozempic, Wegovy, or Zepbound. While nausea and fatigue remain the most common side effects, about 4% of...

Justices Poised to Protect Generics Manufacturers From Liability for Decisions of Pharmacists About Prescribing Their Products
The Supreme Court heard arguments in Hikma Pharmaceuticals USA v. Amarin Pharma, where Amarin seeks to hold generic maker Hikma liable for pharmacists dispensing its generic product in ways that infringe Amarin’s Vascepa patents. The Court appeared skeptical that Hikma’s...

RNA-Built Droplets Create Customizable Organelles Inside Living Cells
UCLA researchers have engineered programmable artificial organelles by assembling RNA nanostars into droplet‑like condensates inside living cells. The RNA sequences encode assembly instructions, allowing precise control over condensate size, composition, and subcellular location. Published in Nature Nanotechnology, the study demonstrates...

Drug Trials Snapshots: ELREXFIO
Pfizer’s ELREXFIO (elranatamab‑bcmm) received accelerated FDA approval on Aug 14 2023 for relapsed/refractory multiple myeloma after at least four prior therapies. The approval rests on a single‑arm trial of 187 patients, with 97 efficacy‑eligible participants showing a 57.7% objective response rate, including...

Pfizer Pushes Elrexfio Into Earlier Multiple Myeloma Line After Phase III Success
Pfizer will ask regulators to approve Elrexfio (elranatamab) as a second‑line treatment for relapsed/refractory multiple myeloma after the MagnetisMM-5 Phase III trial met its primary endpoint. The study, which enrolled 944 patients, demonstrated a statistically significant PFS advantage over J&J's...

Active-Duty US Soldiers to Receive MDMA Therapy for PTSD Next Year
The Department of Defense has approved two MDMA‑assisted therapy trials for active‑duty service members, allocating $4.9 million to each of Walter Reed and an Emory‑UT Health partnership. A total of 186 soldiers with PTSD will receive up to three MDMA doses...

Drug Trials Snapshots: VEOPOZ
VEOPOZ (pozelimab‑bbfg) received FDA approval on August 18, 2023 to treat CD55‑deficient protein‑losing enteropathy (CHAPLE disease) in patients of all ages. The decision was based on a single‑arm trial of ten patients enrolled in Turkey, Thailand and the United States, with a...

Axsome Gets FDA Approval for First Non-Antipsychotic Drug to Treat Agitation Associated with Alzheimer’s Dementia
The FDA has expanded the indication for Axsome Therapeutics’ Auvelity, approving it to treat agitation associated with Alzheimer’s disease dementia. This marks the first non‑antipsychotic medication cleared for this distressing symptom. Auvelity, originally approved in 2022 for major depressive disorder,...
Axsome Hits Record High On Alzheimer's Agitation Approval
Axsome Therapeutics saw its stock surge to a record $207.75 after the FDA approved Auvelity for treating agitation in Alzheimer’s disease patients. The drug, already cleared for major depressive disorder, now gains a second indication, expanding its market reach. This...

FDA Proposal Would Exclude Bulk Compounding of Weight-Loss Drugs Semaglutide, Tirzepatide, and Liraglutide
The FDA announced a proposal to remove semaglutide, tirzepatide and liraglutide from the 503B bulk drug list, concluding there is no clinical need for outsourcing facilities to compound these GLP‑1 weight‑loss agents from bulk substances. Because FDA‑approved versions are already...

Intellia Raises $180 M, Files BLA After First In‑Vivo CRISPR Phase 3 Success
Intellia Therapeutics priced a $180 million public offering of 16.7 million shares and filed a rolling Biologics License Application for its hereditary angioedema candidate lonvo‑z after the drug met primary endpoints in the first global in‑vivo CRISPR Phase 3 trial. The dual milestones...

Drug Trials Snapshots: EXXUA
Fabre‑Kramer Pharmaceuticals received FDA approval for EXXUA (gepirone) on September 22, 2023 as a once‑daily oral antidepressant for adult major depressive disorder. The approval was based on two placebo‑controlled, flexible‑dose Phase III trials that enrolled 456 patients, with 442 forming the...

The Future of Biomanufacturing: Key Highlights From INTERPHEX 2026
INTERPHEX 2026 highlighted a rapid shift toward digital integration and smarter single‑use technologies in biomanufacturing. Panels emphasized automation, continuous processing, and real‑time monitoring as ways to boost scalability while tackling data‑driven control challenges. A second discussion focused on supply‑chain resilience,...

Drug Trials Snapshots: POMBILITI
The FDA approved POMBILITI (cipaglucosidase alfa‑atga) on September 28, 2023 for adult patients with late‑onset Pompe disease (LOPD) weighing ≥ 40 kg who are not responding to existing enzyme replacement therapy. Approval is based on a randomized, double‑blind trial of 123 participants across 61 sites...

Eli Lilly Profits More than Double as Weight-Loss Revenue Soars
Eli Lilly announced net profit more than doubled year‑over‑year, driven by a surge in weight‑loss drug sales. The company’s obesity treatments, chiefly Mounjaro and the newly approved Zepbound, generated roughly $2 billion in revenue this quarter, lifting total sales to about $7.5 billion....

ESOC 2026: What to Expect in Maastricht
The 12th European Stroke Organisation Conference (ESOC) convenes in Maastricht from May 6‑8, drawing roughly 4,200 delegates from 100 nations. Six scientific tracks will showcase the latest clinical trial data on stroke prevention, diagnosis, and treatment, including late‑breaking sessions on pre‑hospital...

Asembia ASX26: The Strain on the Development of Infrastructure for Sustainable C&G Therapy
Cell and gene therapies promise transformative treatments but remain hampered by steep costs across research, manufacturing, and delivery. At the Asembia ASX26 Summit, Aradigm Health CEO Will Shrank highlighted the fragmented U.S. payment landscape as a core barrier to sustainable...

FDA's Oncology Advisors Vote Against 'New Paradigm' In AstraZeneca Trial
The FDA’s oncology advisory committee voted 6‑3 that AstraZeneca’s late‑stage trial of camizestrant, an oral selective estrogen‑receptor degrader (SERD), did not demonstrate a clinically meaningful benefit. AstraZeneca had positioned camizestrant as a potential first‑in‑class oral therapy for hormone‑receptor‑positive metastatic breast...
Should Regulators Rely More on AI to Accelerate Drug Approvals?
Regulators, led by the FDA, are testing AI tools to speed drug approval processes. The agency’s pilot projects include AI‑driven real‑time monitoring of clinical trials and automated data review. Proponents argue AI can cut analysis time and uncover hidden patterns,...

Novartis Finalizes US Expansion with Seventh Facility to Strengthen End-to-End Drug Manufacturing
Novartas has completed its US expansion plan by adding a seventh facility in Morrisville, North Carolina, dedicated to API production for oral solid‑dose and RNA‑based therapies. The site is part of a $23 billion multi‑year investment to build an end‑to‑end domestic...

FDA Warning Letter Breakdown: A Manufacturer Tells Investigators Its AI Agent Never Said Process Validation Was Required
The FDA’s Center for Drug Evaluation and Research issued a warning letter to a Michigan‑based homeopathic drug manufacturer after an October 2025 inspection uncovered multiple CGMP violations, including unsanitary conditions, missing microbiological testing, and inadequate component verification. The letter’s standout...
Avalyn Pharma Takes a Breath to Raise $300M in IPO Cash for Lung Drug Trials
Avalyn Pharma completed a $300 million IPO, pricing 16.6 million shares at $18 each and listing on Nasdaq under the ticker AVLN. The Boston‑based biotech will use the proceeds to advance inhalable versions of pirfenidone (AP01) and nintedanib (AP02), plus a combination...

Pharma Power Shifts: Controlling Diseases, Not Just Assets
Most pharma pipeline analysis is spreadsheet theater. I used AI to look at the 2020 → 2026 shift. The real question is not: Who has the most assets? It is: Who controls the disease category? GLP-1s are a metabolic land grab. Oncology is becoming a platform stack. Trials...

Pharma Pulse: The Ember Cube 2 and a $1 Trillion Prescription Spending Projection for 2026
Ember LifeSciences unveiled the Ember Cube 2, a modular cold‑chain solution with real‑time monitoring, GPS tracking and cloud visibility, earning a Red Dot “Best of the Best” award. At the Asembia AXS26 summit, policy experts warned that new pricing legislation is compressing post‑launch...

This Gas Provides Rapid Relief For Major & Treatment-Resistant Depression (M)
Recent clinical investigations show that inhaled nitrous oxide can alleviate symptoms of major depressive disorder, including cases resistant to standard therapies, within hours of administration. Across multiple small‑scale trials, roughly 40% of participants achieved remission after a single session, with...

FDA Expedites Review of New HIV Treatment Combo
The FDA has granted priority review to Gilead Sciences’ new single‑tablet HIV regimen that combines bictegravir and lenacapavir. The application is backed by phase 3 ARTISTRY‑1 and ARTISTRY‑2 trials, which showed sustained viral suppression in adults already virologically controlled. If approved,...

FDA Moves to Speed Review Support for Psychedelic-Related Treatments
The FDA announced a new fast‑track mechanism for psychedelic‑based therapies, issuing Commissioner’s National Priority Vouchers to three programs targeting depression and PTSD with psilocybin or methylone. It also granted an IND for DemeRx NB to study noribogaine hydrochloride as a...

Varenicline
Varenicline (Chantix) received FDA approval in 2006 as a partial α4β2 nicotinic receptor agonist, offering a middle‑ground approach between nicotine replacement and bupropion. Its mechanism delivers enough receptor activation to ease cravings while antagonizing nicotine’s rewarding effects. The drug quickly...
IDEAYA Biosciences Files NDA for Darovasertib/Crizotinib Under FDA’s RTOR Pathway
IDEAYA Biosciences announced that the FDA has accepted its New Drug Application for the darovasertib‑crizotinib combo under the Real‑Time Oncology Review (RTOR) program. The submission follows Phase 2/3 OptimUM‑02 data showing a 58% drop in disease‑progression risk and a median progression‑free...

Merck Still Sees ‘Compelling’ Outlook for Terns Leukemia Drug
Merck agreed to acquire Terns Pharmaceuticals for $6.7 billion, paying $53 per share, after updated trial data showed its TERN‑701 leukemia drug achieving a major molecular response (MMR) rate north of 50% at 24 weeks. The data suggested TERN‑701 could outperform...

South Korea Clears Curocell's Limcarto‑Joo, First Home‑Grown CAR‑T Therapy
Curocell's Limcarto‑Joo received marketing approval from South Korea's Ministry of Food and Drug Safety, becoming the country's first domestically developed CAR‑T therapy. The treatment targets adult patients with diffuse large B‑cell lymphoma and primary mediastinal B‑cell lymphoma, marking a shift...

Other News to Note for April 30, 2026
Chinese biotech companies have reached an inflection point, gaining leverage as co‑dealmakers in cross‑border partnerships, according to multinational executives. Roche announced the discovery of new TREM2 agonist compounds aimed at modulating microglial activity for neurodegenerative diseases. Plasticity launched an advanced...

In the Clinic for April 30, 2026
BioWorld’s “In the Clinic” briefing for April 30, 2026 teases upcoming clinical data from Pfizer and PTC, directing readers to a subscription‑only full story. The brief serves as a snapshot of biopharma and med‑tech readouts scheduled for release, though specific trial details...

The Bangui Operation: A Story of Blood, Science and Biomedical Exploitation
In the early 1990s the Pasteur Institute in Bangui ran a covert HIV‑vaccine trial that recruited roughly 3,000 Central African soldiers, extracting over 11,000 blood samples. The research was funded by French institutions and aimed to fast‑track vaccine development at...
STAT+: Five-Way Obesity Drug Is Super Effective — in Mice
Researchers have unveiled a preclinical obesity candidate that combines five distinct mechanisms, delivering dramatic weight loss in mouse models and outperforming current GLP‑1 therapies. The multi‑modal approach targets appetite, metabolism, gut hormones, energy expenditure, and adipose tissue remodeling, achieving up...

Swiss BioTech Startup ALP Bio Raises €1.9 Million to Advance Immune Organoid and AI Platform
Swiss startup ALP Bio announced a €1.9 million (≈$2.1 million) pre‑seed round led by 42CAP to accelerate its immune organoid‑AI platform for antibody development. The platform couples human tonsil organoid readouts with generative AI to predict and reduce immunogenicity risk earlier in...

CMS Extends GENEROUS Model Deadline for Pharma and States
The Centers for Medicare & Medicaid Services has pushed back the application deadline for the GENEROUS Model, a program that lets state Medicaid programs buy drugs at prices comparable to those in other nations. Manufacturers now have until June 11 to...
Top 15 Biopharma R&D Players and Late‑Stage Prospects
My annual report on the top 15 R&D players in biopharma is out. It's a beast at some 9000 words, but if you're interested in the top prospects in the late-stage pipeline, I got a bunch for you. The stakes...

Interim PFS Review Confirms Study Continuation
$SMMT ivo HARMONi-3 update on squamous subgroup interim PFS analysis: "At this early interim PFS analysis reviewed exclusively by the Independent Data Monitoring Committee (iDMC), the iDMC recommended that the study continue as planned."

Zealand Pharma and Roche Advance Petrelintide to Phase 3 for Chronic Weight Management
Zealand Pharma and Roche are moving the amylin analog petrelintide into Phase 3 trials to treat chronic overweight and obesity. The drug achieved up to 10.7% mean weight loss in the Phase 2 ZUPREME‑1 study, with tolerability comparable to placebo. A March 2025...
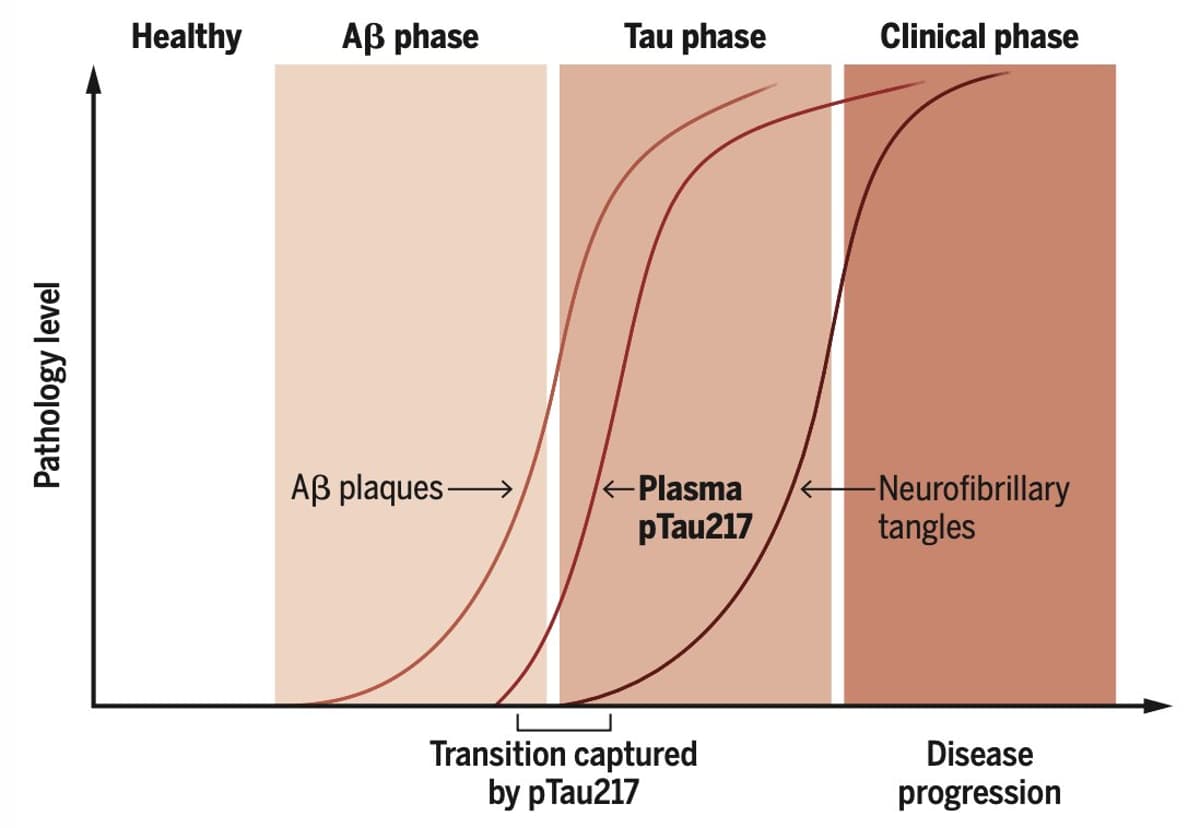
P‑tau217 Blood Test Predicts Alzheimer’s Risk Early
Good summary of p-tau217, the breakthrough blood test to predict risk of Alzheimer's in people well before onset of symptoms @ScienceMagazine https://t.co/qVJtYR4bnz https://t.co/OhjLExTroG
UniQure Pursues UK Approval for Huntington’s Gene Therapy
UniQure, in ‘symbolic’ win, to seek UK approval of Huntington’s gene therapy https://t.co/plxdEw43FD by @Lilah_Alvarado $QURE + 22% #GeneTherapy

Merck Announces First Dose in Phase 3 Study with Enpatoran for Lupus Patients with Active Skin Manifestations
Merck announced the first patient has been dosed in its global Phase 3 ELOWEN program, testing the oral TLR7/8 inhibitor enpatoran in lupus patients with active skin manifestations. The double‑blind, placebo‑controlled studies—ELOWEN‑1 and ELOWEN‑2—will enroll roughly 200 participants each across 266...

Pinetree Therapeutics: Promising Biotech Worth Watching
A biotech you need to keep an eye on... Pinetree Therapeutics $AZN https://t.co/wVt9XMcr0Z (disclosure: SAB member)