Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Amgen Files Update to Tavneos Label as FDA Escalates Push to Withdraw
Amgen filed a supplemental application to revise the prescribing information for its rare‑disease drug Tavneos (avacopan) as the FDA intensifies its effort to withdraw the product. The label update seeks to add new safety warnings and monitoring requirements after the agency flagged concerns about serious infections and renal complications observed in post‑marketing data. Amgen also committed to conduct a post‑approval study to assess long‑term outcomes. The move comes amid ongoing litigation and a potential market impact for the $1.5 billion drug.
Getein Biotech Q1 Profit Rises 18% to $10.7M as Revenue Hits $37.5M
Getein Biotech posted a first‑quarter profit of RMB76.3 million ($10.7 M), up 18% year‑over‑year, and revenue of RMB267.5 million ($37.5 M), a 9.6% increase. The results highlight accelerating demand for molecular diagnostics in China.

GoodRx Partners with Novo Nordisk to Offer Self-Pay Pricing for New Oral Ozempic® Pill
GoodRx has teamed with Novo Nordisk to sell the newly launched oral Ozempic® pill at transparent self‑pay rates, ranging from $149 to $299 per month depending on dosage. The tiered pricing covers the 1.5 mg, 4 mg and 9 mg strengths, letting patients...

Amgen Positions MariTide as Potential ‘Best Monthly’ Obesity Drug
Amgen is positioning its bispecific antibody‑peptide MariTide as the premier monthly or less‑frequent obesity treatment. The company launched a Phase 3 MARITIME‑Switch study to evaluate switching patients from weekly GLP‑1 injections to four‑to‑six monthly doses. Phase 2 data showed up to a...

Finland’s Algorithmiq Takes Top Spot in $50M Q4Bio Challenge with Quantum Drug Simulation Milestone
Finland’s Algorithmiq won the $2 million prize in Wellcome Leap’s $50 million Q4Bio challenge, becoming the first to demonstrate an end‑to‑end quantum‑classical workflow for drug simulation on real hardware. Using up to 100 qubits on IBM’s quantum computer, the team modeled the...

Theradaptive Receives US FDA Approval to Initiate P-III Trial of OsteoAdapt SP for Spinal Fusion Procedures
Theradaptive has secured U.S. FDA approval to launch the pivotal OASIS Phase III trial evaluating its OsteoAdapt SP device as a bone‑graft alternative in transforaminal, anterior, and lateral lumbar interbody fusion (TLIF, ALIF, LLIF) procedures. The trial follows earlier studies involving...

Enzymatic DNA Synthesis Frees Synthetic Biology Design
“You should design for the biology you want to build, not for the process limitations of a supplier that’s been holding the field back.” That line from @Jason_Gammack , CEO of @AnsaBio , gets right to the heart of one...
AstraZeneca’s Breast Cancer Drug Fails to Earn Backing of FDA Advisory Committee
The FDA’s advisory committee voted against recommending AstraZeneca’s oral SERD camizestrant for HR⁺/HER2‑ metastatic breast cancer patients with an ESR1 mutation, citing concerns over the Phase 3 SERENA‑6 trial design. The study switched patients to camizestrant at the point of mutation...

Lilly's Double-Beat Widens the GLP-1 Gap—And a New Pill Could Make It Permanent
Eli Lilly posted a strong Q1 2026, with revenue up 56% YoY to almost $20 billion, driven by $12.8 billion in GLP‑1 sales from Mounjaro and Zepbound. The FDA also approved Foundayo, the first oral GLP‑1 agonist for obesity that can be taken without...

BIO on the American Road Tours Gene Therapy Hub in Ohio
BIO President John F. Crowley toured Ohio on April 28, spotlighting the state’s emerging gene‑therapy hub. Researchers at Nationwide Children’s Hospital have delivered two of the FDA’s first eight approved gene therapies for Duchenne muscular dystrophy and spinal muscular atrophy type 1....
Corcept Ties ALS Drug to Improved 2-Year Survival as Phase 3 Start Date Nears
Corcept Therapeutics reported that its experimental ALS drug dazucorilant reduced the two‑year risk of death by roughly 87% in a Phase 2 extension study, despite missing its primary motor‑function endpoint. The survival benefit was most pronounced at the 300 mg dose, though...

MDMA-Assisted Therapy for Depression: A Promising but Early First Step
A small open‑label proof‑of‑principle study examined MDMA‑assisted therapy in 12 adults with moderate‑to‑severe major depressive disorder. Participants received two MDMA dosing sessions spaced a month apart together with nine psychotherapy sessions. At two months, 75% of participants achieved remission and...

Interim Data Hint Weaker PFS; Selloff Caps Losses
Leerink's Daina Graybosch on $SMMT "... In our base case scenario of the company’s likely statistical design choices and enrollment timing, not hitting statistical significance at this interim suggests a degradation in PFS Hazard Ratio (HR) of ~10 percentage points...

FDA Approves First Non‑antipsychotic for Dementia Agitation
⚕️ FDA Approves First Non-Antipsychotic Drug to Treat Agitation Associated with Dementia 🔗https://t.co/cPNtkiSXBs 🌐 #DHPSP #Pharma #Healthcare https://t.co/KsBKc45ekk

Cue Biopharma Licenses Ascendant-221 From Ascendant Health Sciences in a ~$691.5M Deal
Cue Biopharma announced an exclusive license with Ascendant Health Sciences for Ascendant-221, an anti‑IgE monoclonal antibody aimed at allergic diseases. The agreement grants Cue worldwide rights, except Greater China, in exchange for a $15 million upfront payment and up to $676.5 million...

Approaches to Reducing Toxicity and Side Effects in Cell and Gene Therapy
Cell and gene therapies are expanding rapidly, with the market projected to exceed $9 billion in 2025 and grow over 15% annually through 2035. Safety remains a hurdle, prompting multiple strategies to curb cytokine release syndrome (CRS) and related toxicities. Companies...

FDA's ODAC Delivers One Loss, One Win for AstraZeneca
The FDA’s Oncologic Drugs Advisory Committee (ODAC) voted 6‑3 against AstraZeneca’s oral SERD camizestrant in combination with CDK‑4/6 inhibitors for first‑line HR‑positive, HER2‑negative breast cancer with ESR1 mutations, citing a lack of overall survival benefit despite a 56% progression‑free survival...
Gene Editing at Scale, Clinic Seeks Generalizable Therapies
Integrated DNA Technologies helped deliver a CRISPR therapy that rescued baby KJ Muldoon from a fatal urea‑cycle disorder, proving gene editing can correct a single disease‑causing mutation. The success highlights the field’s next hurdle: scaling personalized edits for disorders with...

Smarter AAVs Drive Gene Therapy’s Next Chapter
Gene therapy’s growth is hampered by AAV manufacturing bottlenecks, safety concerns, and high costs, prompting a wave of innovations across bioprocessing, analytics, and vector design. Companies like Thermo Fisher, PackGene, Catalent, and Asimov are deploying design‑space modeling, high‑throughput purification, and...

What’s Next in the Evolution of Standards for Biologics Development
The United States Pharmacopeia (USP) is redefining how documentary standards support biologics, moving from product‑specific monographs toward a hybrid model that blends platform‑based chapters, emerging standards, and analytical reference materials. This shift addresses the growing complexity of monoclonal antibodies, ADCs,...

Space-Efficient Cryogenic Sample Storage
Azenta Life Sciences introduced the CryoArc™ Pico Automated Storage System, a compact LN‑2 cryogenic platform that maintains samples at –190 °C. Designed for biobanking, clinical research, and cell‑gene therapy labs, the system integrates barcode‑driven software for sample tracking, chain‑of‑custody, and CFR 21...

Amgen Posts $5.6 B Q1 Revenue, Raises 2026 Guidance Amid Pipeline Gains
Amgen announced first‑quarter 2026 revenue of $5.6 billion, up 24% year‑over‑year, and raised its full‑year revenue outlook to $37.1‑$38.5 billion. The biotech giant highlighted strong growth in its lipid‑lowering and rare‑disease portfolios and noted progress on the Meritide program and a new...

Seaport Therapeutics Upsizes IPO, Raises $255 Million at $18 per Share
Boston‑based Seaport Therapeutics priced an upsized IPO of 14.16 million shares at $18 each, pulling in about $255 million in gross proceeds and valuing the company near $912 million. The offering, underwritten by a consortium led by Goldman Sachs and JPMorgan, marks a...

The Cost of Inefficiencies in Clinical Trials
Clinical trials are hampered by fragmented technology, leading to roughly 45% higher costs compared with unified platforms. Integration fees, duplicate data entry, and managing multiple vendors add significant expense and delay timelines. Unified data‑management solutions such as CRScube claim 50‑60%...

Zepbound Soars and Lilly Advises Patience on Foundayo
Eli Lilly reported a blockbuster first‑quarter, with revenue jumping 56% to $19.8 billion driven by soaring sales of its obesity drugs Zepbound and Mounjaro. Zepbound alone generated over $4 billion, up about 80% year‑over‑year, while Mounjaro nearly doubled to close to $9 billion. The...

Drug Amount Reporting: FDA Publicly Identifies over 7,700 Noncompliant Companies
On March 31 2026 the FDA released a public list showing that more than 7,700 drug manufacturers failed to submit the required 2024 drug‑amount reports. The list separates 1,254 firms with active drug listings from 6,480 firms with inactive listings, highlighting a...

How Saudi Arabia Is Developing as a Middle East Hub for Clinical Trials
Saudi Arabia is positioning itself as the Middle East’s primary hub for clinical trials, backed by robust state investment and the Vision 2030 Health Sector Transformation Programme. Healthcare spending is forecast to reach $61.05 million in 2026, while pharmaceutical sales are...
New Genetic Discovery Could Spell This Aggressive Cancer’s Downfall
UCLA researchers uncovered a genetic weakness in small cell neuroendocrine carcinoma (SCNC) by creating prostate‑derived organoid models and running genome‑wide CRISPR screens. The screens identified the transcription factor E2F3 as a synthetic‑lethal partner of RB loss, and inhibiting E2F3 halted...
This Treatment Could Reverse Osteoarthritis Joint Damage With a Single Injection
Researchers at the University of Colorado Boulder have secured a $33.5 million ARPA‑H grant to develop a regenerative osteoarthritis therapy that could reverse joint damage with a single injection. The approach uses a controlled‑release particle system to deliver an approved drug...

BMS Brings the Buzz Around Late-Stage Readouts Amid a So-So Q1
Bristol Myers Squibb’s Q1 earnings showed modest 1% revenue growth to $11.5 bn, beating forecasts, while its growth portfolio surged 9% to $6.2 bn. The company highlighted late‑stage readouts for milvexian, a next‑generation anticoagulant, and Cobenfy, a schizophrenia therapy, as potential blockbuster...

Solvent‐Free Preparation of Pharmaceutically Active Diindolylmethane Derivatives by Stable Triple‐Interpenetrated Heterometallic‐Organic Frameworks With Sc‐Pd/Pt Synergistic Sites
Researchers reported a one‑pot synthesis of triple‑interpenetrated heterometallic‑organic frameworks (HMOFs) containing Sc‑Pd or Sc‑Pt synergistic sites. The resulting ScPd‑INA, ScPt‑INA and ScPt‑MCA catalysts achieved up to 99% conversion in solvent‑free Friedel‑Crafts alkylation of diindolylmethane (DIM) precursors. Single‑crystal X‑ray diffraction confirmed...
STAT+: Her Daughter Mila Got a Bespoke Medicine. Now She’s Starting a New Biotech to Make More
Julia Vitarello, whose eight‑year‑old daughter Mila was treated with a tailor‑made gene therapy, announced she is launching a new biotech to scale individualized medicines. Her previous company, EveryONE Medicines, folded after FDA guidance on custom therapies proved insufficient for investors....
State Tele-Buprenorphine Prescribing Policies by Medical Professional Type
A RAND‑led legal mapping of all 50 states and D.C. reveals stark differences in Medicaid policies governing fully virtual buprenorphine prescribing for opioid use disorder. While 32 states permit physicians, nurse practitioners and physician assistants to prescribe via telemedicine, 12...

What Happens When AI Transforms a Specialized Field Overnight?
In 2020 DeepMind released AlphaFold2, an AI that predicts protein structures with laboratory‑grade accuracy, quickly generating models for over 200 million proteins—a 1,500‑fold jump from prior data. The breakthrough earned its creators a 2024 Nobel Prize in Chemistry and sparked a...

LEO Pharma to Acquire Replay for Rare Skin Disease Gene Therapy Platform
LEO Pharma has signed a definitive agreement to acquire gene‑therapy specialist Replay for an upfront cash payment of $50 million, with additional milestone fees and tiered single‑digit royalties. Replay brings a herpes simplex virus (HSV) platform that delivers large therapeutic genes...

Scientists Build Drug-Carrying DNA Robots to Target Diseases
Scientists have engineered microscopic DNA robots that can carry therapeutic payloads and seek out viruses, acting as nano‑surgeons within the bloodstream. By applying origami‑inspired rigid joints and flexible components, the robots achieve nanometer‑scale precision. Movement is programmed through DNA strand...
Beacon Biosignals Is Mapping the Brain During Sleep
Beacon Biosignals has launched an FDA‑cleared, lightweight EEG headband that records clinical‑grade brain activity while users sleep at home. The device’s machine‑learning platform extracts detailed sleep‑stage metrics and subtle architecture changes, supporting more than 40 global clinical trials for conditions...

Explosion in UK Demand for GLP-1s Risks NHS Capacity Crisis
The Food Foundation’s January survey estimates 8.25 million people in the UK have used or are considering GLP‑1 weight‑loss drugs, representing about 7% of the population. NHS targets to treat 220,000 patients over three years from June 2025 and 3.4 million over 12...
![[Comment] GLP-1 Therapies: An Emerging Approach for Alcohol Reduction?](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/f628ae5758874c46e9810dd10e2c007b.webp)
[Comment] GLP-1 Therapies: An Emerging Approach for Alcohol Reduction?
Alcohol use disorder (AUD) remains one of the world’s most prevalent yet undertreated conditions, with fewer than 2 % of affected Americans receiving an FDA‑approved medication. Recent randomized trials of once‑weekly GLP‑1 receptor agonists, especially semaglutide, have demonstrated statistically significant reductions...

AstraZeneca Certifies 17,000 Staff in AI to Chase $80 Billion Revenue Goal
AstraZeneca has certified more than 17,000 employees in AI competencies, a cornerstone of its plan to hit an $80 billion revenue target by 2030. CFO Aradhana Sarin says the program is already feeding a pipeline of 1,000 AI pilots that could...

Gentler Treatment Improves Survival in Children with Relapsed Leukemia
A UK‑wide trial (UKALL Rel2020) tested a gentler regimen for children and young adults with relapsed acute lymphoblastic leukemia, using reduced‑intensity chemotherapy followed by the targeted immunotherapy blinatumomab. The study enrolled 188 patients across 25 centres and achieved a 92%...

Semaglutide Cuts Heavy Drinking and Cravings in AUD
GLP-1 Drug Semaglutide (Ozempic, Wegovy) Reduced Heavy Drinking & Craving in Adults with Alcohol Use Disorder https://t.co/FYRkjWYhJQ https://t.co/cGtEajk9lp

FDA, up Close and Personal: A Banker’s Recalibration
Former FDA senior advisor Peter Reikes argues that the agency’s perceived slowness stems from its core principles of independence, caution, and process discipline rather than dysfunction. He notes that despite recent leadership turnover and political pressure, a deep bench of...

The CRISPR Medicine That Cured a Child: How Yuri Milner’s Breakthrough Prize Brought Gene Therapy to Hollywood
At the 2026 Breakthrough Prize ceremony, Baby KJ was celebrated after receiving Casgevy, the first FDA‑approved CRISPR gene‑editing therapy for sickle cell disease and beta‑thalassemia. The $3 million prize honored researchers Stuart Orkin, Swee Lay Thein and others whose work on the BCL11A...

EnteroBiotix Raises $25 Million to Launch Largest Microbiome IBS Therapy Study
EnteroBiotix announced a $25 million financing round led by Thairm Bio and the Scottish National Investment Bank to fund the largest microbiome‑based Phase IIb trial in irritable bowel syndrome with constipation (IBS‑C). The capital will support a 300‑patient study in the...

Eli Lilly Inks $2.25 B AI‑driven Drug Discovery Pact with Profluent
Eli Lilly has signed a $2.25 billion agreement with AI‑specialist Profluent to co‑develop a platform for computational drug discovery. The deal, announced today, signals a major capital shift toward AI‑enabled R&D in pharma.

We Tried to Get Trump to Back Psychedelic Therapy — Here’s What Happened
The authors drafted a bipartisan policy blueprint to accelerate psychedelic‑based treatments, targeting veterans with PTSD, addiction and depression. After Joe Rogan shared the proposal, it amassed over 1.5 million views on X, prompting meetings with health‑policy influencer Calley Means and a...
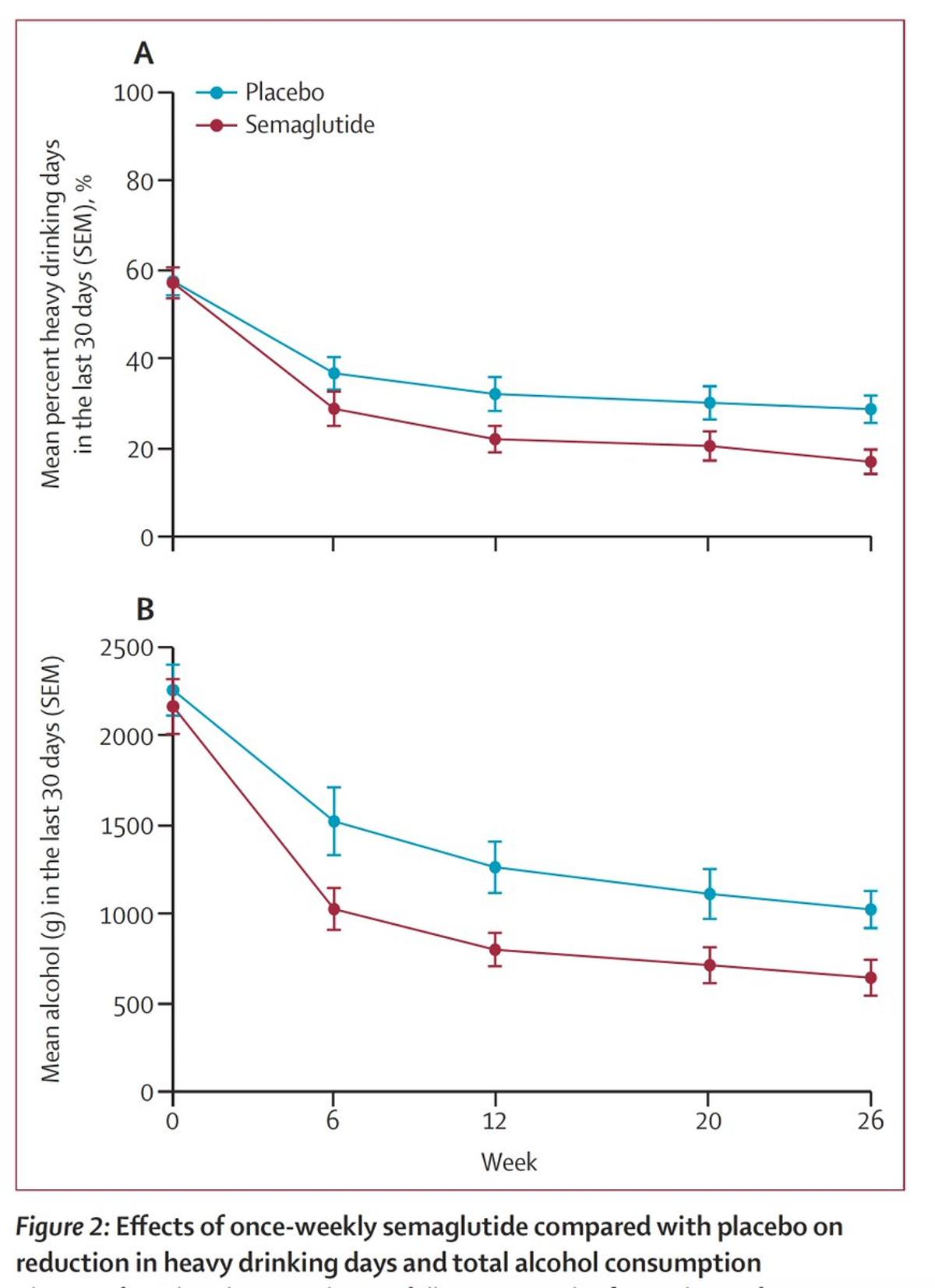
Ozempic Cuts Drinking in Obese AUD Patients
First randomized trial to show Ozempic reduces alcohol consumption in people seeking treatment for alcohol use disorder. Placebo-controlled, double-blind. Participants with BMI >30 kg/m2. https://t.co/M9Pk2gzKHD @TheLancet https://t.co/OT660QIn6N

GLP-1s Reduce Heavy Drinking Days in Patients With Obesity, Alcohol Use Disorder
A randomized, double‑blind trial in Copenhagen found that once‑weekly semaglutide 2.4 mg significantly reduced heavy‑drinking days in patients with alcohol use disorder and obesity. Over 26 weeks, the semaglutide group saw a 41.1‑percentage‑point drop in heavy‑drinking days versus 26.4 points for...
GLP‑1 Drugs Cause Less Muscle Loss than Assumed
Do GLP-1 drugs really cause muscle loss? Turns out perhaps less than what we thought… here’s what you need to know, from: https://t.co/VwrRxnoblW https://t.co/M2RaE1f3EG