Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Psychedelics in Medicine: Curiosity & Caution
In this episode, Dr. Ryan Cole and addiction specialist Dr. Molly Rutherford discuss the surge of interest in psychedelic‑assisted therapies for conditions like addiction, PTSD, and depression, highlighting the recent presidential executive order that accelerates FDA review and funding for these drugs. They explore why patients—especially those stuck on medication‑assisted treatment or facing high relapse rates—are turning to alternatives such as ibogaine, psilocybin, and ketamine, and share personal and clinical anecdotes illustrating both promise and pitfalls. Dr. Rutherford emphasizes the growing acceptance among physicians, the need for rigorous, supervised treatment settings, and the importance of balancing hype with compassionate, evidence‑based care. The conversation underscores the evolving regulatory landscape and the critical role of patient education and provider training.

FDA Approves First‑In‑Human Trial of Motif Neurotech’s Depression Brain Implant
The U.S. Food and Drug Administration has granted Motif Neurotech permission to begin a first‑in‑human study of its miniature brain‑computer interface designed for treatment‑resistant depression. The Houston‑based startup says the device, the size of a blueberry, can be implanted in...
Meta‑Analysis Shows GLP‑1 Drugs Cut Cardiovascular Events by 13% Over Three Years
Researchers at Anglia Ruskin University analyzed data from more than 90,000 patients and found GLP‑1 receptor agonists lower major adverse cardiovascular events by roughly 13% over an average three‑year follow‑up. The benefit appears across diabetic and non‑diabetic high‑risk groups, reinforcing...

FDA ODAC Backs AstraZeneca’s Truqap Combo for PTEN‑deficient Prostate Cancer
The FDA’s Oncologic Drugs Advisory Committee voted 7‑1 to recommend AstraZeneca’s Truqap (capivasertib) combined with abiraterone and androgen deprivation therapy for PTEN‑deficient metastatic hormone‑sensitive prostate cancer. The recommendation follows the CAPItello‑281 Phase III trial, which showed a 19% reduction in radiographic...

Esperion to Be Bought by ARCHIMED in $1.1 B Deal, Shares Surge 56%
Esperion Therapeutics agreed to be acquired by funds managed by ARCHIMED for up to $1.1 billion. Shareholders will receive $3.16 in cash per share—a 58% premium—plus contingent milestone payments that could add $100 million. The announcement sent the stock up 56% in...
FDA Grants Expanded Access to Revolution Medicines' Pancreatic Cancer Pill Daraxonrasib
The U.S. Food and Drug Administration approved an expanded‑access program for Revolution Medicines' experimental pancreatic cancer drug daraxonrasib, allowing eligible patients to receive the pill before formal approval. The move follows trial data that doubled median survival versus chemotherapy and...
Development Analysis and Strategic Insights From a 505(b)(2) Reformulation Product: Enzalutamide
Astellas used the 505(b)(2) pathway to reformulate enzalutamide (XTANDI®) from a 40 mg lipid‑filled soft capsule into 40 mg and 80 mg film‑coated tablets, aiming to cut pill burden. Five biopharmaceutic studies demonstrated comparable AUC between the two dosage forms, while the tablet...

'Build AI that Can Accurately Represent the Full Complexity of Biology': Mark Zuckerberg Wants to Cure All Diseases but Needs...
Meta billionaire Mark Zuckerberg is channeling $500 million into Biohub’s Virtual Biology Initiative to amass massive cellular datasets for AI modeling. The funding splits into $100 million for global data collection and $400 million for advanced imaging and engineering tools. Partnerships with the...

Study Links Troponin‑I Phosphorylation in Obesity to Heart Failure, Reversible with Weight Loss
A new study published in Science identified heightened phosphorylation of the protein troponin‑I as a molecular switch that weakens heart muscle in people with severe obesity and HFpEF. Patients who lost more than 2 kg/m² BMI using GLP‑1 agonists showed restored...

Semaglutide Cuts Heavy Alcohol Drinking in Obese Patients, Lancet Trial Shows
A randomized, double‑blind trial led by Copenhagen University Hospital shows semaglutide, the GLP‑1 agonist behind Wegovy and Ozempic, significantly reduces heavy drinking episodes in patients with obesity and alcohol use disorder. The findings, published in The Lancet, could expand treatment...

FDA, AstraZeneca and Amgen Launch Real‑Time Clinical Trials Initiative
The U.S. Food and Drug Administration announced a real‑time clinical trials (RTCT) program backed by AstraZeneca and Amgen, allowing safety and efficacy data to flow continuously to regulators. The pilot seeks to trim the 10‑12‑year development cycle by eliminating the...

MHRA Names Former CDC CIO Jason Bonander as New Chief Digital and Technology Officer
The Medicines and Healthcare products Regulatory Agency (MHRA) has appointed Jason Bonander, former CIO of the U.S. CDC, as its chief digital and technology officer. He will steer a five‑year modernization plan that emphasizes data, AI oversight and agile regulation,...

Incyte Secures FDA Nod for Jakafi XR Extended‑Release Tablets Across Three Hematologic Indications
Incyte announced that the FDA approved Jakafi XR (ruxolitinib) extended‑release tablets for adults with intermediate‑ or high‑risk myelofibrosis, polycythemia vera refractory to hydroxyurea, and adults and children 12 + with steroid‑refractory acute or chronic graft‑versus‑host disease. The once‑daily formulation is bioequivalent...
Human Organ Chip Systems Reshape Drug Development
Harvard’s Wyss Institute, led by Dr. Donald Ingber, has spent over a decade perfecting Human Organ Chip systems that mimic organ-level functions in a thumb‑drive‑sized device. Recent FDA and NIH policy shifts endorse these chips as viable alternatives to animal...
KRAS G12D Drug Shrinks Tumors, Delays Progression
Setidegrasib, an investigational therapy targeting KRAS G12D, demonstrated tumor shrinkage and delayed disease progression in early trials for advanced lung and pancreatic cancers, offering a potential new approach for hard-to-treat mutations. oncology

Patent Monopolies Fueled Opioid Crisis, Perdue Falls
With Perdue Pharma biting the dust, it might be a good time for a post-mortem that looks at the government-granted patent monopolies that provided the incentive for pushing opioids https://t.co/bWKnxlYaS2

Could Ozempic Help With Alzheimer’s Disease? Scientists Are Taking a Closer Look
A new Anglia Ruskin University review of 30 preclinical studies suggests GLP‑1 drugs such as Ozempic and Wegovy can lower amyloid‑beta and tau, the hallmark proteins of Alzheimer’s disease. Twenty‑two studies reported reduced amyloid‑beta and nineteen showed decreased tau, with liraglutide...

Malaria Vaccine Team Faced Funding Delays at Every Stage
“The researchers who worked on this malaria vaccine struggled at every step of the process to get faster funding.” ~@salonium

ByteDance's AI-Designed Therapies Spotlight Global Progress
ByteDance’s drug unit presents AI-designed therapies at global conferences Yes this is reality of AI applications, but we are "winning" by slowng Chinese company progress on AI? https://t.co/G3lIeOAddU
Cornell Team Decodes Ketamine’s Antidepressant Pathway, Shows Low‑Dose Cocktail Works in Mice
Weill Cornell Medicine scientists identified a specific set of opioid receptors on prefrontal‑cortex interneurons that mediate ketamine’s rapid antidepressant action. In two papers published in Cell and Science Advances, they also demonstrated that a three‑drug, low‑dose cocktail can reproduce the...
R‑MDDMA Boosts Neuroplasticity and Ant
methylated MDMA analog in animals R-MDDMA "still promoted structural neuroplasticity in cortical neurons, facilitated fear extinction learning, and produced sustained antidepressant-like effects" did not directly activate 5-HT2B receptors https://t.co/w0ltV7VXQL

Mimio’s Fasting Supplement Shows Measurable Metabolic and Cardioprotective Gains
Mimio announced that its fasting‑mimetic supplement delivered significant improvements in metabolic health, hunger regulation and cardioprotective markers in a decentralized clinical trial published in Nature. The study, run with People Science, underscores the potential of supplement‑based biohacking to replicate fasting...
UniQure Advances AMT-130 Gene Therapy Toward UK Approval for Huntington's Disease
uniQure N.V. reported a successful pre‑submission meeting with the UK Medicines and Healthcare products Regulatory Agency for its Huntington's disease gene therapy AMT-130, setting a Q3 2026 filing target. The milestone, coupled with a pending FDA Type B meeting, moves...
Boosting One Protein Helps the Brain Fight Alzheimer’s
Researchers at Baylor College of Medicine discovered that increasing the protein Sox9 in astrocytes enables mouse models of Alzheimer’s disease to clear existing amyloid plaques and retain memory performance. The study, published in Nature Neuroscience, showed that elevated Sox9 enhances...

Boosting NAD+ May Restore Sleep in Dementia
NAD+‒circadian rhythm coupling in dementia "Pharmacological and lifestyle-based strategies targeting NAD+ restoration are outlined as potential approaches to improve sleep and circadian rhythm integrity..." https://t.co/qQXLHS8nou https://t.co/te8QGUftC4

Your Next Dog May Live Longer
Longevity biotech Loyal, founded by Celine Halioua, has received FDA clearance to market a daily pill that improves insulin sensitivity and could extend dogs’ lifespans. The agency deemed the drug likely effective based on a small study of about 50...
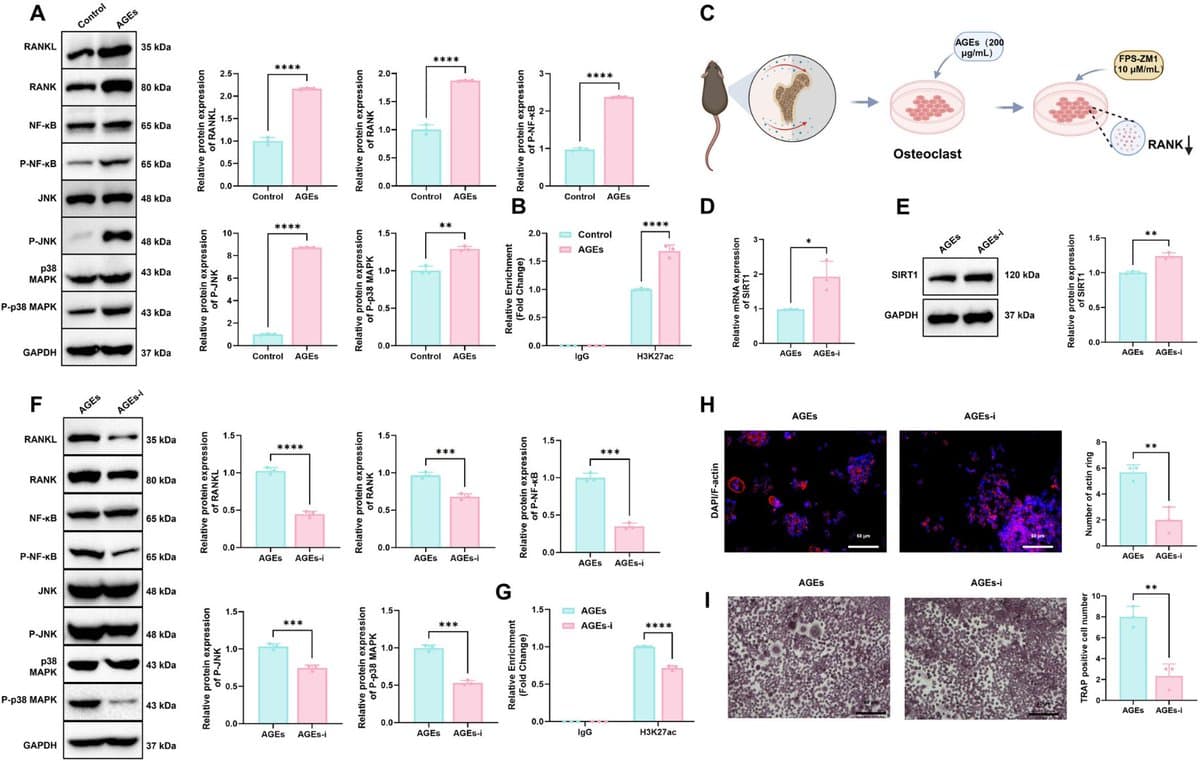
AGEs Trigger SIRT1 Loss, Accelerating Osteoarthritis via RANKL
SIRT1 Downregulation by Advanced Glycation End Products Activates RANKL-Dependent Osteoclast Signaling and Drives Chondrocyte Senescence During Osteoarthritis Development "Targeting this mechanism may offer new therapeutic opportunities for delaying age-related OA progression." https://t.co/HB2nmyZ43u

5th Circuit Halts Mail‑Order Abortion Pill, Mandating In‑Person Dispensing
A three‑judge panel of the 5th U.S. Circuit Court of Appeals ordered that the abortion pill mifepristone can no longer be mailed, requiring in‑person distribution at clinics. The decision overturns FDA guidance, expands state‑level abortion restrictions and is expected to...

Optoelectronic Tweezers Integrated with Microfluidics Promise Low‑Power Nanoscale Manipulation
Researchers led by Shuailong Zhang published a technical roadmap that couples optoelectronic tweezers (OETs) with microfluidic platforms, dramatically lowering optical power and thermal risk. The paper outlines materials and architectural solutions that could enable high‑throughput, AI‑driven biomedical assays.

FDA Clears Veppanu, First PROTAC Breast Cancer Pill From Arvinas and Pfizer
The U.S. Food and Drug Administration approved Veppanu, the first proteolysis‑targeting chimera (PROTAC) for advanced estrogen‑receptor positive, HER2‑negative breast cancer. Developed by Arvinas and Pfizer, the pill offers a new treatment option for patients whose tumors carry ESR1 mutations after...

The Company that Built TikTok’s Algorithm Is Now Designing Drugs for Diseases Pharma Called Undruggable
ByteDance’s Anew Labs showcased its first AI‑designed therapy, a small‑molecule inhibitor of IL‑17, at the American Association of Immunologists meeting in Boston. The molecule targets a protein‑protein interaction long deemed undruggable, suggesting generative AI can breach a major pharmaceutical barrier....

Promising New Technique Uses Nanoparticles to Detect Pancreatic Cancer
Scientists at Oregon Health & Science University have unveiled a blood‑based assay that uses an electronic jolt to harvest tumor‑derived nanoparticles, achieving 97% accuracy in detecting pancreatic cancer. The technique, validated in a blinded study of 36 participants, outperforms the...

The Commodification of Sensitive Open Data
The European Union’s European Health Data Space (EHDS) regulation, adopted in March 2025, will make the electronic health records of roughly 450 million residents available for secondary use by March 2029. The framework defaults to inclusion, requiring citizens to opt out and offering...
Precise Spatiotemporal Cardiac Repair and Regeneration
Researchers are advancing spatiotemporal drug delivery systems (DDS) that synchronize therapeutic release with the heart’s natural healing stages after myocardial infarction. These platforms integrate bioelectrical scaffolds, programmable degradation, and cell‑free vesicle carriers to provide phase‑specific immunomodulation, angiogenesis, and antifibrotic treatment....
FDA Approves Pfizer/Arvinas Oral Breast Cancer Drug Veppanu for ESR1‑Mutated Tumors
The U.S. Food and Drug Administration approved Veppanu, an oral antibody‑drug conjugate co‑developed by Pfizer and Arvinas, for adults with advanced estrogen‑receptor positive, HER2‑negative breast cancer carrying ESR1 mutations. The decision follows a late‑stage trial of 624 patients that showed...

FDA Approves Auvelity, First Non‑Antipsychotic for Alzheimer’s Agitation
The U.S. Food and Drug Administration has approved Auvelity (dextromethorphan‑bupropion) for agitation associated with dementia due to Alzheimer’s disease, making it the first non‑antipsychotic therapy for this indication. Axsome Therapeutics says the drug’s NMDA‑antagonist and sigma‑1‑agonist mechanism could address a...
5th Circuit Halts Nationwide Mail Distribution of Abortion Pill, Restores In‑Person Rule
The Fifth U.S. Circuit Court of Appeals issued a unanimous injunction that blocks the FDA’s 2023 rule allowing mifepristone to be mailed nationwide, reinstating the requirement that the drug be dispensed in person. The ruling, driven by Louisiana’s lawsuit, threatens...

FDA Approves Once-Daily Idvynso Tablet for Treating HIV
The FDA has approved Merck’s Idvynso, a once‑daily, two‑drug tablet combining doravirine and islatravir, for HIV‑1‑infected adults who are virologically suppressed. The regimen replaces existing antiretroviral therapy and is tenofovir‑free, targeting patients without prior treatment failure. Approval rests on two...
Federal Appeals Court Temporarily Halts Abortion Pill Access by Mail
A federal appeals court in the Fifth Circuit issued a temporary stay that blocks abortion providers from prescribing mifepristone via telemedicine and mailing the pills to patients. The ruling follows a lawsuit by Louisiana seeking to enforce its near‑total abortion...
Modern Meds Are Precise; Older Drugs Are Broad-Spectrum
Modern drugs tend to be very tightly targeted. Older drugs like metformin or Tylenol cut a wider swath.

Incyte Announces FDA Approval of Jakafi XR™ (Ruxolitinib) Extended-Release Tablets for the Treatment of Myelofibrosis, Polycythemia Vera and Graft-Versus-Host Disease
Incyte announced FDA approval of Jakafi XR, an extended‑release ruxolitinib tablet, for adult intermediate‑ or high‑risk myelofibrosis, hydroxyurea‑intolerant polycythemia vera, and steroid‑refractory acute or chronic graft‑versus‑host disease in patients 12 years and older. The 55 mg once‑daily tablet is bioequivalent to the 25 mg...

224. The Return of the Mifepristone Mess
The Fifth Circuit issued a stay that reinstates the in‑person dispensing requirement for Mifepristone, undoing the FDA’s 2021 and 2023 rules that allowed mail‑order and telehealth prescriptions. The order, grounded in the Administrative Procedure Act, takes immediate nationwide effect, echoing...
Innovative Nanoparticle Technique Advances Early Detection of Pancreatic Cancer
Researchers have unveiled a novel nanoparticle‑based imaging agent that markedly improves early detection of pancreatic cancer. In pre‑clinical trials the probe identified tumors as small as 2 mm, delivering a 30% sensitivity boost over conventional CT scans. The technology earned FDA...

The Biopharm Brief: Microbiome Fast Track, Leo Pharma Gene Therapy Deal, AAV Automation Advances
The FDA has awarded Fast Track designation to a live biotherapeutic aimed at restoring gut microbiota in moderate‑to‑severe ulcerative colitis, accelerating its development path. Leo Pharma is buying Replay’s gene‑therapy program to broaden its rare‑dermatology portfolio, marking a shift toward...

Artificial Intelligence for Drug Development
The FDA’s Center for Drug Evaluation and Research (CDER) is formalizing its AI strategy through a draft 2025 guidance, an AI Council created in 2024, and extensive stakeholder engagement. CDER has reviewed more than 500 AI‑enabled drug submissions since 2016...

External Engagements with FDA for Artificial Intelligence in Drug Development
The FDA has published a comprehensive menu of early‑engagement options for sponsors leveraging artificial intelligence across the drug development lifecycle. Sponsors can request formal meetings for IND or pre‑IND programs, or use targeted programs such as C3TI for trial design,...
Zhejiang Huahai Pharma Posts Q1 Net Profit Rise to $62 M on Robust Generic Sales
Zhejiang Huahai Pharmaceutical Co., Ltd. announced Q1 net profit of RMB444.33 million ($62 million), up from RMB375.01 million a year earlier, driven by strong generic drug sales. Revenue slipped 1.4% to RMB2.335 billion ($327 million), highlighting a shift toward higher‑margin products amid a tightening Chinese...

Katherine Szarama, PhD, Named Acting CBER Director: Who’s Next?
Vinay Prasad stepped down as director of the FDA's Center for Biologics Evaluation and Research (CBER), and Katherine Szarama, PhD, was named acting director. Szarama, a biologist with prior experience as CBER deputy director and stints at CMS, Emerson Collective...

Eli Lilly Q1 2026 EPS $8.55 Beats Forecast, Revenue $19.8B Tops Estimates
Eli Lilly reported first‑quarter 2026 earnings of $8.55 per share and $19.8 billion in revenue, outpacing analyst forecasts by 22.7% and 12.5% respectively. The beat lifted the stock 5.96% in pre‑market trade and prompted an upward revision of full‑year revenue guidance...

Regulatory and IP Considerations for Veterinary Drugs in Canada
The article outlines Canada’s regulatory framework for veterinary drugs, separating them from other animal health products and detailing the data‑protection, patent‑linkage, and supplementary‑protection regimes that apply only to veterinary drugs. It explains submission requirements, labeling rules, priority‑review criteria, and biowaiver...