Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech financings
Spanish biotech Ona announced a $86.6 million Series B round, ranking among the largest venture financings in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funds will expand Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical pipeline.
Tilray’s Rescheduling Rally Fizzles as DOJ Plan Stalls, Shares Slip Below $6
Tilray Brands saw its stock plunge from a brief high of $8 to around $6 after the Justice Department announced only a partial, immediate rescheduling of FDA‑approved medical marijuana products. The limited scope of the move and pending administrative hearing have dampened investor enthusiasm for a broader Schedule III reclassification.

RxUtility Launches Personalized AI Companion Mimi™ to Compare Every Drug Price as Affordability Crisis Grows
RxUtility announced the beta launch of Mimi™, an AI‑driven conversational companion that compares real‑time prices for every prescription across U.S. pharmacies. The free‑to‑use service asks users for medication, dosage, insurance and location to surface the lowest‑cost options, including coupons and...

Latus Bio Secures $97 Million Series A to Scale Gene‑Therapy Manufacturing
Latus Bio announced a $97 million Series A financing, including a $43 million extension led by 8VC, to fund its proprietary AAV capsid platform and move two lead programs toward IND filing. The capital raise underscores investor confidence in scalable gene‑therapy solutions...

Latus Bio Raises $97M to Expand Gene Therapy Pipeline
Latus Bio announced a $97 million Series A financing round led by 8VC and DCVC Bio. The funding will accelerate its gene‑therapy pipeline focused on delivering adeno‑associated virus (AAV) capsids that can reach deep brain structures at dramatically lower doses....
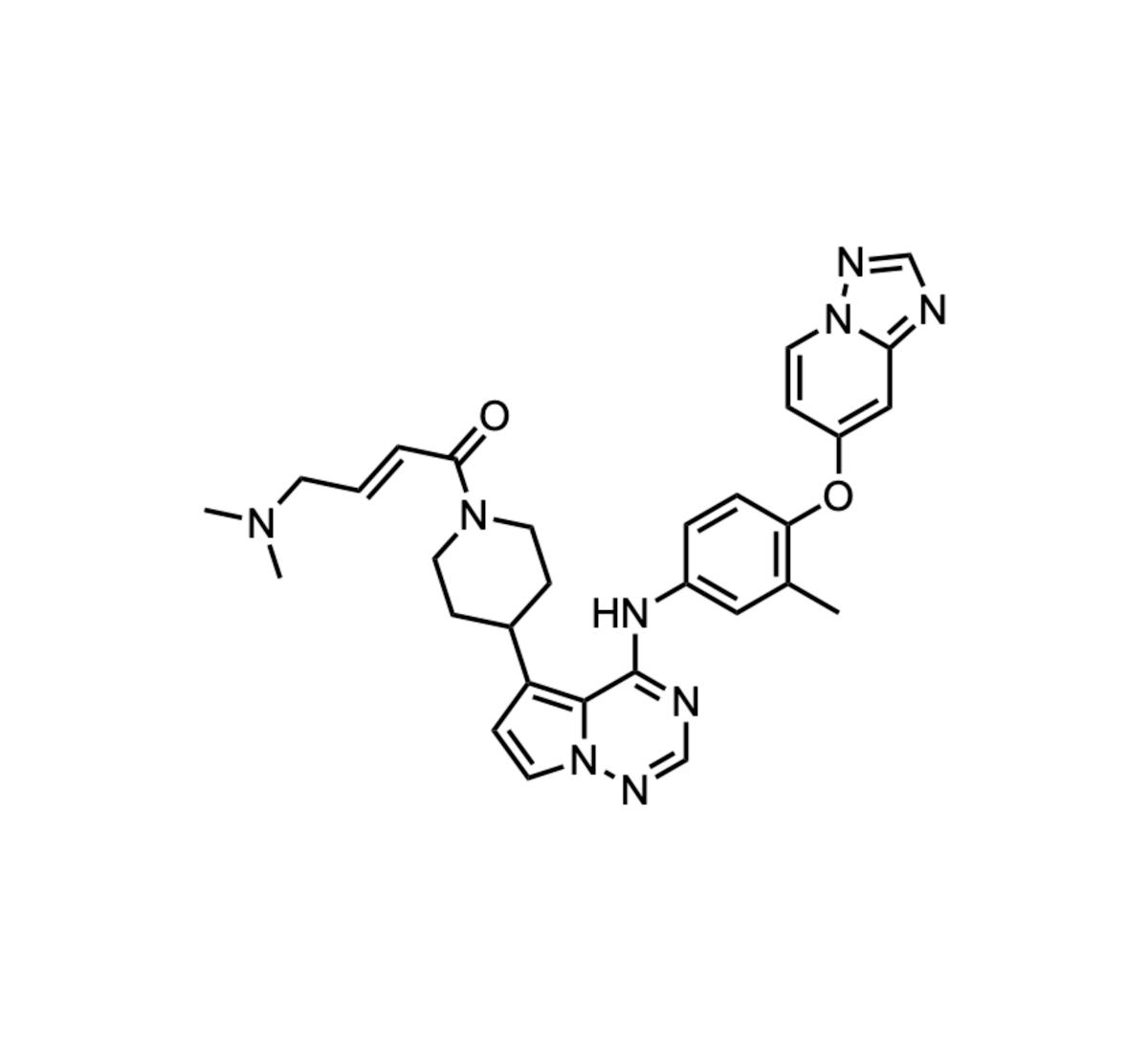
IAM1363
Iambic Therapeutics of San Diego announced the initiation of a Phase 1 clinical trial for an oral covalent inhibitor targeting HER2‑mutant cancers. The molecule, identified through an AI‑guided high‑throughput screening campaign, binds irreversibly to the mutant HER2 kinase domain. Preclinical data...

Asembia AXS26: Why Pharma Can’t Afford to “Go It Alone” Anymore
At the Asembia AXS26 Summit, Syneos Health’s SVP of Market Access Kim Plesnarski warned pharma that rising costs and complex access hurdles demand a shift from legacy patient‑support models to agile, outcome‑focused partnerships. She highlighted that many manufacturers still rely...

STAT+: Pharmalittle: We’re Reading About an Abortion Pill Controversy, Another UCB Deal, and More News
Two drugmakers, Danco Laboratories and GenBioPro, have petitioned the U.S. Supreme Court to restore mail‑order access to the abortion pill mifepristone after a lower court temporarily blocked deliveries. The appeal follows a series of lawsuits challenging the drug’s original 2000...

Choosing the Right CDMO for Long-Term Stability
Biotech firms must scrutinize contract development and manufacturing organizations (CDMOs) before committing to multi‑year projects. Dr. Patrick Meyer of Rentschler Biopharma outlines criteria such as transparent communication, accurate timelines, and a proven commercialization record. Technical expertise and a resilient supply chain...
Gene Therapy’s Evidence Problem—Lessons From Recent FDA Decisions
The FDA recently rejected REGENXBIO’s gene‑therapy candidate RGX‑121, citing an unvalidated biomarker as the primary endpoint and reliance on an external natural‑history control. The decision highlights a broader pattern of mixed regulatory outcomes for advanced therapeutics, with approvals like Sarepta’s...

Supreme Court Faces Emergency Request to Restore Mail‑Order Access to Abortion Pill
The U.S. Court of Appeals for the Fifth Circuit stayed the FDA's 2023 rule that permitted mifepristone to be prescribed via telemedicine and mailed to patients. In response, Danco Laboratories and generic maker GenBioPro petitioned the Supreme Court for an...
Glowing Nanoparticles Exposed Hidden Cancer-Protein Behavior that Could Reshape Drug Screening
A Broad Institute team led by Sam Peng introduced upconverting nanoparticle probes that remain luminescent for minutes to hours, enabling continuous single‑molecule imaging of cancer‑related receptors in living cells. Using these probes, they captured real‑time dimerization dynamics of EGFR, HER2...

For Once, some Scientific Fraudsters Have to Pay Their Money Back to the Government.
Dana‑Farber Cancer Institute agreed to pay $15 million to resolve False Claims Act allegations that it submitted false certifications on six NIH research grants between 2014 and 2024. The institute admitted that researchers reused and altered images in 14 grant‑linked publications,...

Protein Degraders Gain Speed as Arvinas Scores Landmark Approval
Arvinas received FDA approval for Veppanu, the first PROTAC therapy, marking a milestone for protein degraders. The drug showed modest benefit in a phase 3 trial, with the clearest effect in patients carrying ESR1 mutations, prompting the company to lay off...

Theranostic Fiber with Micro‑Wrinkles Promises Real‑Time Health Monitoring
Scientists Meng, Zou and Lv introduced a multifunctional theranostic fiber that integrates micro‑wrinkles to sense physiological signals, deliver treatment and wirelessly transmit data. The work, published in npj Flexible Electronics, could reshape personalized healthcare and human‑machine interfaces.
Everads Therapy Publishes First-in-Human Data on Suprachoroidal Injector
Everads Therapy announced the publication of first-in-human trial data for its suprachoroidal injector, demonstrating safety, tolerability and rapid posterior drug distribution in patients with diabetic macular edema. The results, appearing in Ophthalmology Science, were showcased at the ARVO 2026 meeting,...

Module 4, Section 2: All About Assays
The Module 4, Section 2 briefing provides a concise overview of modern assay platforms used in early‑stage drug discovery. It references key literature on PRMT5 fragment‑based screening that produced the MRTX1719 candidate, as well as thermal‑shift, surface plasmon resonance (SPR), and polymerase...

Needle-Free Diabetes Care: 6 Devices that Painlessly Monitor Blood Sugar
Needle‑free glucose monitors are moving from research labs to commercial shelves, with six innovative devices highlighted for their non‑invasive approaches. Abbott’s FreeStyle Libre remains the market leader for interstitial sensing, while newcomers such as Occuity Indigo, D‑Pocket, Light Touch Technology,...

PCI Adds US Sterile Fill/Finish Capabilities Through $1B Global Expansion Drive
PCI Pharma Services announced a $1 billion global expansion, allocating $100 million to upgrade its San Diego campus with a high‑speed isolator line that will more than double its prefilled syringe capacity. The company also launched a GMP‑ready isolator vial and lyophilization line...

BEAM Wins TIME 100, Delivers First Gene Base Editing Cure
Congrats to $BEAM on being named as one of @TIME’s 100 most influential & innovative companies that are shaping the world & our future. @beamtx’s leading Gene Editing platform - Base editing, has achieved a significant milestone when KJ Muldoon...

Acarbose Protects Retina by Modulating Microglial Metabolism
Acarbose modulates microglial Pkm2 acetylation to reshape immunometabolism and preserve retinal neurons after ischemia-reperfusion 👉"Our findings support that ACA exerts retinal protection through the Sirt1-Pkm2-NAD axis, suggesting a metabolic checkpoint that integrates immune and mitochondrial regulation." https://t.co/V3WGPV26qk

BioCryst Partners with Irish Affiliate of Neopharmed Gentili in ~$345M EU Commercialization Deal for Navenibart
BioCryst Pharmaceuticals has signed a licensing agreement with the Irish affiliate of Neopharmed Gentili, granting exclusive rights to commercialize its long‑acting plasma kallikrein inhibitor, Navenibart, across the European Union. The deal provides BioCryst with a $70 million upfront payment and potential...

German Lab Prints Tumors to Study Neutrophil Interactions
One of the cooler things I saw during my visit to Germany last week was this tumor printer. The company is using it to test how neutrophils affect the tumor microenvironment. https://t.co/jXzxDpj6nZ

Funding Cuts and Visa Limits Spark US Brain Drain
#NIH funding cuts & visa crackdowns are driving away talented foreign researchers who normally would have flocked to the US, a @statnews.com survey shows. "It is going to cause a long-term brain drain," one US based researcher told @DrewQJoseph. ...

Regulatory Submissions with Real-World Evidence: Successes, Challenges, and Lessons Learned - 09/23/2025
In a September 23, 2025 speech, FDA Principal Deputy Commissioner Sara Brenner outlined how real‑world evidence (RWE) has been used to support regulatory decisions and announced the agency‑wide FDA‑RWE‑ACCELERATE initiative. She highlighted recent examples from CDER, CBER and CDRH, and introduced Sentinel 3.0,...

Top AI Researcher Departs Genentech, Rethinks Pharma Future
“It got a bit too distant for me." Some insights into big pharma & AI from @kchonyc, a top AI researcher who talked with me about leaving Roche's Genentech and what he's thinking about next: https://t.co/IvzWUjc9Qd

First FDA ODAC Meeting without Pazdur Highlights Conceptual Challenges with Trial
The FDA’s Oncology Drugs Advisory Committee (ODAC) convened for the first time in nine months to review two AstraZeneca cancer therapies. The panel voted 6‑3 against approving camizestrant, an oral SERD for HER2‑negative advanced breast cancer, citing the trial’s failure...

Here's How Medication Abortion Works with Just One Drug That's Still Fully Available
A federal appeals court ordered the FDA to revert to in‑person prescribing rules for mifepristone, overturning the Biden administration’s telehealth flexibility. The decision takes effect nationwide, while the makers of mifepristone have appealed to the Supreme Court for a rapid...

Celcuity Reports the P-III (VIKTORIA-1) Trial Data on Gedatolisib Combination for HR+/HER2- PIK3CA Mutant Advanced Breast Cancer
Celcuity announced phase‑III VIKTORIA‑1 data showing that gedatolisib combined with fulvestrant, with or without palbociclib, achieved a statistically significant improvement in progression‑free survival (PFS) versus the alpelisib‑fulvestrant standard of care in HR+/HER2‑ advanced breast cancer patients harboring PIK3CA mutations. The...

Summit Shares Descend as PD-1/VEGF Asset Misses Early Survival Mark
Summit Therapeutics announced that its anti‑PD‑1/VEGF bispecific, ivonescimab, failed to meet the interim progression‑free survival threshold in the Phase 3 HARMONi‑3 trial for squamous NSCLC. The miss triggered a 26% plunge in Summit’s share price, closing at $16.12. An independent data...

Drugmaker AbbVie Chooses North Carolina for $1.4B Manufacturing Campus
AbbVie announced a $1.4 billion investment to build a 185‑acre manufacturing campus in Durham, North Carolina, targeting its immunology, neuroscience and oncology portfolios. The site will create 734 jobs over four years, incorporate AI‑driven production tools, and be operational by the...

UCB Captures Candid in $2.2B Autoimmune Deal as Pharma’s M&A Train Chugs Along
UCB announced a $2 billion acquisition of Candid Therapeutics, adding a pipeline of T‑cell engager drugs aimed at autoimmune and inflammatory diseases. The deal also includes up to $200 million in milestone payments and is expected to close by Q3 2026. Candid’s...

How a Vision-Restoring Gene Therapy Proved that We Can Treat Inherited Diseases
Luxturna, the first FDA‑approved gene‑augmenting therapy for inherited retinal disease, received the 2026 Breakthrough Prize after restoring sight to patients with Leber’s congenital amaurosis type 2. Developed by Spark Therapeutics founders Katherine High, Jean Bennett and surgeon Albert Maguire, the treatment...

OneSource Notches a Second Generic Semaglutide Approval in Canada, Through Partner
Canada approved its second generic version of Ozempic (semaglutide) through Apotex, which partnered with Orbicular Pharmaceutical Technologies and India‑based CDMO OneSource Specialty Pharma. OneSource handled end‑to‑end manufacturing, leveraging its FDA‑approved Bengaluru facility, while Orbicular led product development. The approval follows...

ARCHIMED to Acquire Esperion Therapeutics for ~$1.1B
Archimed announced a definitive agreement to acquire Esperion Therapeutics for up to $1.1 billion, taking the company private and delisting it from NASDAQ. Esperion shareholders will receive $3.16 in cash per share plus a contingent value rights (CVR) package worth roughly...

FDA Greenlights First Ibogaine Trial as Hype Eclipses Limited Data
The U.S. Food and Drug Administration announced it will permit the first clinical trial of ibogaine, a psychedelic derived from a West African shrub, after President Donald Trump highlighted the drug at a White House event. Researchers caution that the...

Andalusia Unveils Ruthenium‑Uracil Nanoparticle Coating to Combat Hospital Superbugs
A team from the Instituto de Investigaciones Químicas, backed by the CSIC and the University of Sevilla, has created a ruthenium‑uracil nanoparticle that eliminates Staphylococcus aureus in lab tests. The breakthrough, funded by the Andalusian Ministry of University, Research and...

What Is Your AI Drug Repurposing Strategy Missing?
The article argues that AI‑driven drug repurposing for oncology often fails because models are fed fragmented, noisy data despite abundant datasets. It stresses that more data alone won’t improve outcomes; instead, high‑quality, curated, structured data—such as knowledge graphs linking genes,...

EU Backs Itvisma Gene Therapy for Spinal Muscular Atrophy
European regulators have issued a positive opinion on Itvisma, Novartis' onasemnogene abeparvovec gene therapy for spinal muscular atrophy (SMA). The Committee for Medicinal Products for Human Use (CHMP) recommended marketing authorisation on 23 April 2026, pending final approval by the European Commission....

Rhythm Pharmaceuticals Reports the EC Approval of Imcivree (Setmelanotide) for Acquired Hypothalamic Obesity
Rhythm Pharmaceuticals announced that the European Commission has granted marketing authorization for Imcivree (setmelanotide) to treat acquired hypothalamic obesity in patients aged four years and older. The approval is based on the Phase III TRANSCEND trial, which enrolled 120 participants...
Novo Nordisk to Launch Ozempic for Type 2 Diabetes in US
Novo Nordisk will launch an oral version of Ozempic in the United States, following FDA approval that marks the first oral GLP‑1 drug cleared for both blood‑sugar control and cardiovascular risk reduction. The tablets, available in 1.5 mg, 4 mg and 9 mg...

Arvinas and Pfizer Report the US FDA Approval of Veppanu (Vepdegestrant) for ESR1-Mutated Breast Cancer
The U.S. FDA has approved Veppanu (vepdegestrant) for adults with ESR1‑mutated ER+/HER2‑ advanced or metastatic breast cancer who have received at least one line of endocrine therapy. The approval follows the Phase III VERITAC‑2 trial, which compared Veppanu to fulvestrant...

MAHA Vs. The FDA: Dredging up Old Anti-Regulation Revisionist History
A new essay in Science-Based Medicine denounces the “health‑freedom” narrative that seeks to dismantle the FDA. It traces the agency’s authority back to the 1962 Kefauver‑Harris amendment, which linked drug approval to rigorous safety and efficacy trials. The author dismantles...

65 % of Eligible Lung Cancer Patients Do Not Receive the Most Appropriate Targeted Therapies, Diaceutics Report Finds
Diaceutics’ 2026 Clinical Practice Gaps report shows that 65% of U.S. patients with advanced non‑small cell lung cancer still miss the most appropriate targeted therapy, a figure unchanged since 2019. While biomarker testing has improved, the biggest loss now occurs...

FDA Action Alert: Argenx, AstraZeneca/Daiichi Sankyo, Biogen/Eisai and Cingulate
May’s FDA docket is light, but the decisions on four high‑profile drugs could reshape market dynamics. Argenx is seeking to broaden Vyvgart’s label to seronegative myasthenia gravis patients, potentially adding 11,000 new users. AstraZeneca and Daiichi Sankyo aim to secure...

Viewpoint: How ‘Health Care Guru’ Joe Rogan Circumvented the FDA’s Skepticism on Psychedelics
Joe Rogan directly messaged President Donald Trump about the therapeutic potential of psychedelics, prompting the president to sign an executive order that fast‑tracks FDA review of these drugs. The order creates a priority‑voucher system that accelerates approvals for psychedelic manufacturers,...

Hidden Ingredient in GLP-1 Tablets Raises New Gut Health Questions
Oral semaglutide tablets rely on the absorption enhancer SNAC (salcaprozate sodium) to cross the stomach lining, but only 0.4%‑1% of the drug reaches the bloodstream. A 21‑day rat study published in the Journal of Controlled Release found that the majority...

5-HT1A Blockade Amplifies DMT’s Subjective Experience
5-HT1A receptor blockade potentiates the subjective effects of DMT Zarmeen Zahid, Rick J. Strassman, Clifford R. Qualls, Sandeep M. Nayak, 2026 https://t.co/5tCpjw2Iaz

FDA Clears GSK’s Gepotidacin, First New Oral UTI Antibiotic in 30 Years
The U.S. Food and Drug Administration has approved GSK’s gepotidacin (Blujepa) for uncomplicated urinary tract infections in females aged 12 and older, marking the first new oral antibiotic class for this indication in nearly 30 years. The decision follows pivotal...

Eli Lilly Inks $2.25 B Profluent Deal to Fast‑track AI‑driven Gene‑therapy Platform
Eli Lilly signed a research agreement worth up to $2.25 billion with AI protein‑design firm Profluent to develop next‑generation gene‑therapy candidates using AI‑engineered recombinases. The deal leverages cash from Lilly’s obesity drug franchise and adds to a string of multi‑billion‑dollar gene‑therapy...

Pharma Embraces AI, Yet Breakthrough Drugs Remain Elusive
JUST IN: The pharmaceutical industry is adopting AI, but new drug discovery through it remains elusive.