Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Advances in Neonatal Cell Therapies: 2025 Update
The 2025 Neonatal Cell Therapies Symposium underscored the rapid maturation of manufacturing for cord blood, placental and amniotic‑derived cellular products, now operating under GMP certification. Robust quality‑management systems integrate donor eligibility, informed consent and continuous environmental monitoring to guarantee batch consistency and ethical sourcing. Emerging automation, closed‑system bioreactors and advanced cryopreservation are cutting production timelines and supporting multicenter trials, while regulatory frameworks such as the TGA’s Biologicals guidelines provide a safety net. Together, these advances promise broader, more equitable access to regenerative treatments for newborns and children worldwide.

MCRI Launches World‑first Trial Giving Obese Parents GLP‑1 Drugs to Curb Child Obesity
The Murdoch Children’s Research Institute (MCRI) announced a world‑first clinical trial that will provide GLP‑1 weight‑loss medication to obese parents enrolled in its Generation Victoria cohort, testing whether parental weight loss can shift household food environments and reduce childhood obesity...
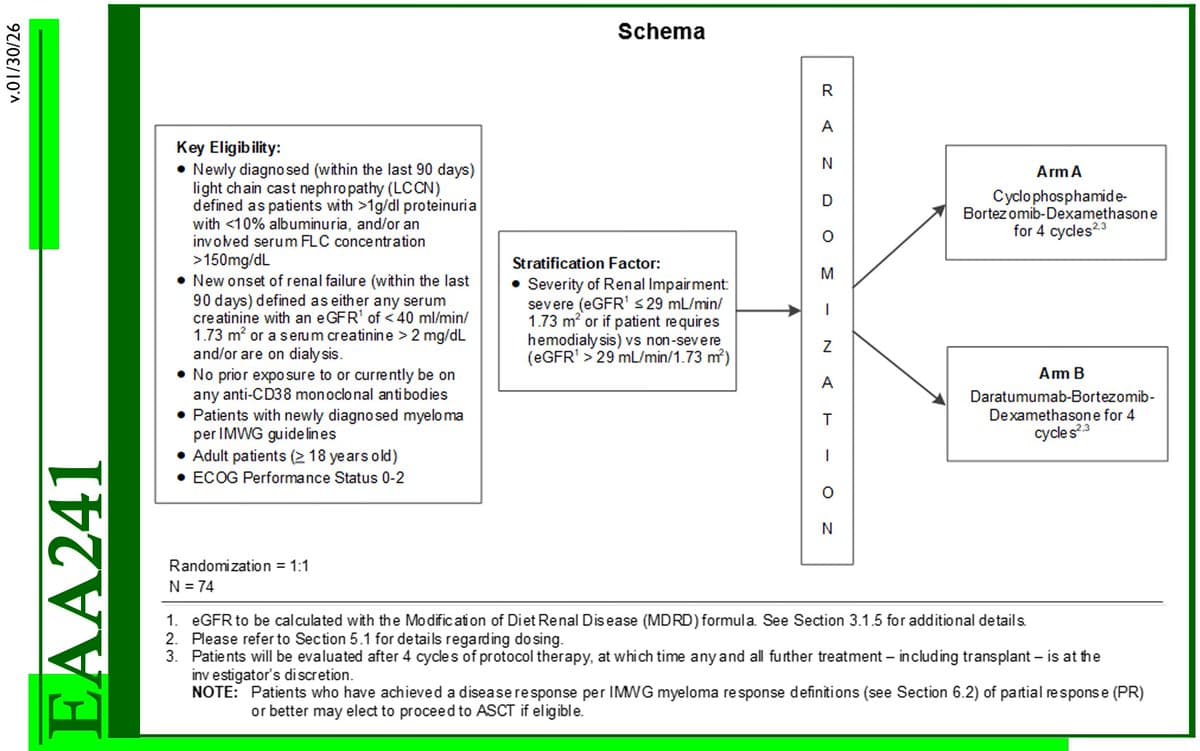
New Trial Compares Dara‑Bor‑Dex vs Cy‑Bor‑Dex in MM with LCCN
EAA241 - Ph 2 RCT Dara-Bor-Dex vs Cy-Bor-Dex in the treatment of Newly Diagnosed Multiple Myeloma with Light Chain Cast Nephropathy (LCCN) [Study activated 8/11/25] @keruakous https://t.co/1NgvVZ3fTA #NCT07085728 #mmsm @eaonc https://t.co/Jrh4teOxUK
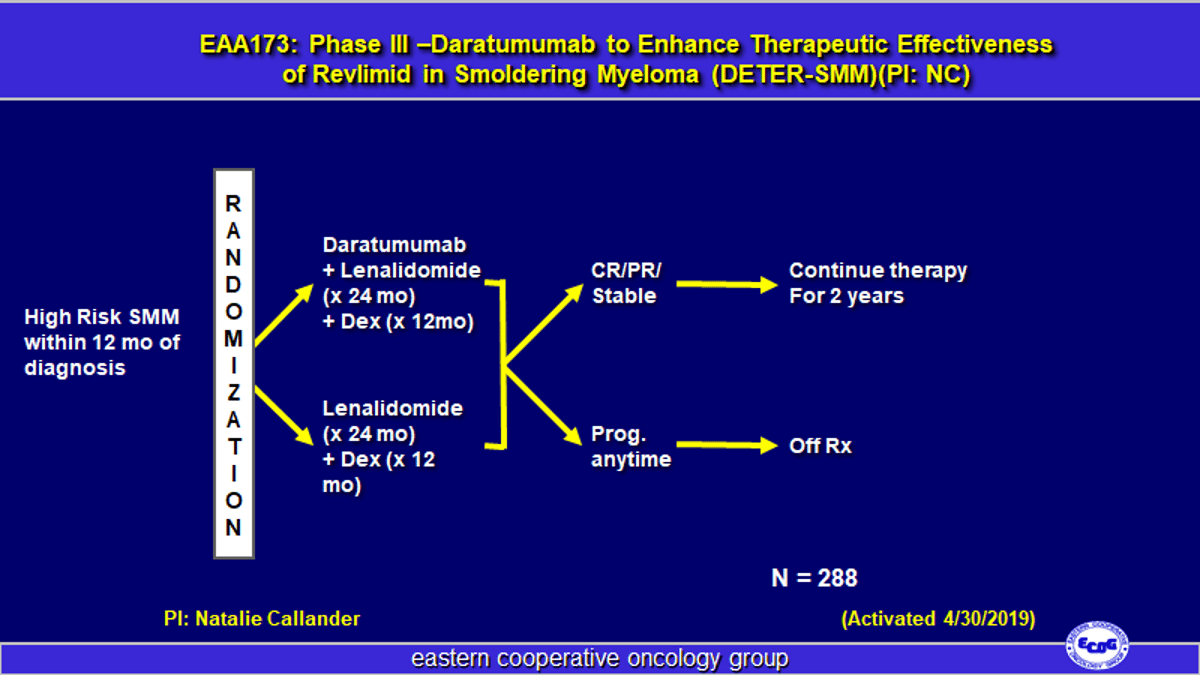
Daratumumab Boosts Revlimid Efficacy in Smoldering Myeloma
#EAonc EAA173 - Daratumumab to Enhance Therapeutic Effectiveness of Revlimid in Smoldering Myeloma (DETER-SMM) - PI: @nsc_natalie https://t.co/VtBMJUjI5X Activated: Apr 30, 2019 #mmsm @eaonc #NCT03937635 @VincentRK @mweissmdphd https://t.co/oQzEYsPoU4

FDA Launches Real‑Time Clinical Trial Initiative, AstraZeneca and Amgen Lead First Pilots
On April 28, 2026, the U.S. Food and Drug Administration unveiled a two‑step plan to roll out real‑time clinical trials, launching two proof‑of‑concept studies and issuing a Request for Information on a summer pilot. AstraZeneca and Amgen are already running...

Phase 3 Trial
.@SWOG S2213 Ph3 RCT Dara-VC Induction Followed by ASCT or Dara-VCD Consolidation & Daratumumab Maintenance in Pts w/ Newly Diagnosed AL Amyloidosis [Activated: 12/1/23] https://t.co/OizUfJCc2c #mmsm #bmtsm https://t.co/zWMK1RfCFW

Senate Introduces Clinical Trial Modernization Act to Cut Patient Costs
Senators Tim Scott (R‑SC) and Mark Warner (D‑VA) unveiled the Clinical Trial Modernization Act (S.4440), a bipartisan effort to remove financial and geographic obstacles for patients in clinical trials. The legislation would permit sponsors to reimburse up to $2,000 annually...

Bristol Myers Squibb Posts $11.5B Q1 Revenue, Reaffirms Full-Year Guidance
Bristol Myers Squibb announced first‑quarter 2026 revenue of $11.5 billion, up 1% year‑over‑year, and reaffirmed its full‑year guidance. The company highlighted a 9% growth in its oncology portfolio, a 280‑basis‑point dip in gross margin, and several late‑2026 regulatory milestones.

Tonight in Your Rights: A Shadow Docket Surprise
The Supreme Court’s shadow docket, led by Justice Samuel Alito, issued a one‑week administrative stay that temporarily restores nationwide mail‑order access to the abortion pill mifepristone. The stay halts a Fifth Circuit ruling that had blocked remote dispensing after Louisiana...
GRIN Therapeutics Launches European Phase 3 Beeline Trial of Radiprodil
GRIN Therapeutics announced the initiation of its pivotal Phase 3 Beeline trial in eight European countries, expanding a global registrational program for radiprodil. The study targets GRIN‑related neurodevelopmental disorder (GRIN‑NDD) caused by gain‑of‑function variants, aiming to become the first disease‑modifying therapy...
Special Packaging Enables Effective Mitochondrial Delivery
Researchers have engineered "mito‑capsules" by wrapping donor mitochondria in erythrocyte‑derived plasma membranes, a technique that markedly improves delivery and integration into recipient cells. In vitro, the capsules restored bioenergetic function in mitochondrial disease models, while in vivo studies demonstrated functional...
Cytokinetics Inc (CYTK) Q1 2026 Earnings Call Transcript
Cytokinetics reported a strong commercial debut for MYCorzo, generating $4.8 million in product revenue within the first nine weeks and securing an $11.9 million Bayer milestone payment. The drug saw rapid adoption, with over 425 prescribers and 70% of dispensed...

Grail Inc (GRAL) Q1 2026 Earnings Call Transcript
GRAIL reported Q1 2026 results showing revenue of $36.2 million, a 26% year‑over‑year increase, driven by 45,000 Gallery tests sold—a 39% volume rise. The company completed a $325 million private placement and secured a pending $110 million equity investment from Samsung to accelerate...

Ocugen Inc (OCGN) Q1 2026 Earnings Call Transcript
Ocugen reported major clinical progress across its gene‑therapy pipeline, completing enrollment for the OCU400 Phase 3 Limelight trial and delivering strong 12‑month efficacy data for OCU410 in geographic atrophy and OCU410ST in Stargardt disease. The company announced a rolling Biologics License...
Cumberland Pharmaceuticals Inc (CPIX) Q1 2026 Earnings Call Transcript
Cumberland Pharmaceuticals reported an 18% revenue increase to $44.5 million for 2025, driven by strong sales of Vibativ, Sancuso and the newly launched Talicia. Adjusted earnings turned positive at $1.7 million and cash flow from operations rose to $4.9 million, narrowing the annual...

Alkermes Plc (ALKS) Q1 2026 Earnings Call Transcript
Alkermes reported $1.5 billion in 2025 revenue, driven by a 9% rise in its proprietary product portfolio, and closed the $775 million cash Avadel acquisition in February 2026, adding the LUMRIZE sleep‑medicine platform. The company forecast 2026 total revenue of $1.73‑$1.84 billion with...

Exelixis Inc (EXEL) Q1 2026 Earnings Call Transcript
Exelixis reported first‑quarter 2026 revenue of $611 million, driven by an $555 million cabozantinib franchise that grew 8% year‑over‑year and lifted global sales to $764 million, a 12.5% increase. The company posted record new patient starts for CABOMETYX, expanding its renal cell carcinoma...

Supernus Pharmaceuticals Inc (SUPN) Q1 2026 Earnings Call Transcript
Supernus Pharmaceuticals announced FDA approval of Qelbree, a non‑controlled ADHD medication for children aged 6‑17, with a U.S. launch planned for the second quarter of 2021. The company reported first‑quarter revenue of $130.9 million, a 38% year‑over‑year increase, driven by growth...

Neurocrine Biosciences Inc (NBIX) Q1 2026 Earnings Call Transcript
Neurocrine Biosciences reported Q1 2026 results with total product sales exceeding $2.8 billion, a 22% year‑over‑year increase driven by strong performance of INGREZZA and the first‑year launch of CRENESSITY. INGREZZA generated over $2.5 billion, up 9%, while CRENESSITY posted more than $300 million, covering...

Vertex Drops mRNA Cystic Fibrosis Program over 'Tolerability' Issues
Vertex Pharmaceuticals announced it is halting development of its mRNA‑based cystic fibrosis (CF) therapy after encountering tolerability and delivery challenges. The decision follows similar setbacks at other biotech firms pursuing mRNA treatments for CF. Vertex will refocus resources on its...

DOJ Moves Certain Marijuana Products to Schedule III, Sets June Rescheduling Hearing
The U.S. Department of Justice issued an order on April 22, 2026 moving FDA‑approved and state‑licensed medical marijuana products into Schedule III of the Controlled Substances Act. A separate order sets a June 29, 2026 hearing to consider broader rescheduling of marijuana from Schedule I to...

FDA Expectations Create Potential Friction in New Form 483 Response Guidance
The FDA has issued a draft guidance outlining heightened expectations for Form 483 response submissions, emphasizing greater detail and faster turnaround. Cooley’s life‑sciences regulatory chair, Sonia Nath, warned that these demands could generate friction between regulators and manufacturers. She urged...

Magnesium Supplementation Cuts Early Colorectal Cancer Risk in New Trial
Researchers at Vanderbilt University Medical Center reported that a 12‑week, personalized magnesium glycinate regimen reduced early signs of colorectal cancer in 240 adults with prior polyps. The supplement reshaped gut microbes to produce vitamin D locally, offering a novel, non‑sunlight‑dependent...

The Fight to Lower Prescription Drug Prices in America #CareTalk
In a May 4 2026 CareTalk episode, host Laura Packard sits with Vinny DeMarco of Maryland Health Care for All to dissect the latest state‑level tactics for lowering prescription‑drug prices. The conversation spotlights Maryland’s $35‑per‑month insulin cap and Colorado’s new drug‑price transparency...

WSJ Calls Trump’s FDA Pick Marty Makary a Potential Disaster for Drug Approvals
The Wall Street Journal warned Sunday that President Donald Trump’s FDA nominee, surgeon‑scientist Dr. Marty Makary, could become a disaster for the agency. The editorial cites Makary’s pattern of blocking rare‑disease and gene‑therapy drugs, alleged conflicts of interest, and a...

Leroy Hood Unveils Dark Proteome Frontier at SynBioBeta 2026
“I am interested in deciphering human complexity in terms of wellness and disease. I guess you would describe me as a multi-disciplinary human biologist.” That is Dr. Lee Hood @ISBLeeHood in one quote. We're excited and honored to have Dr. Leroy Hood...
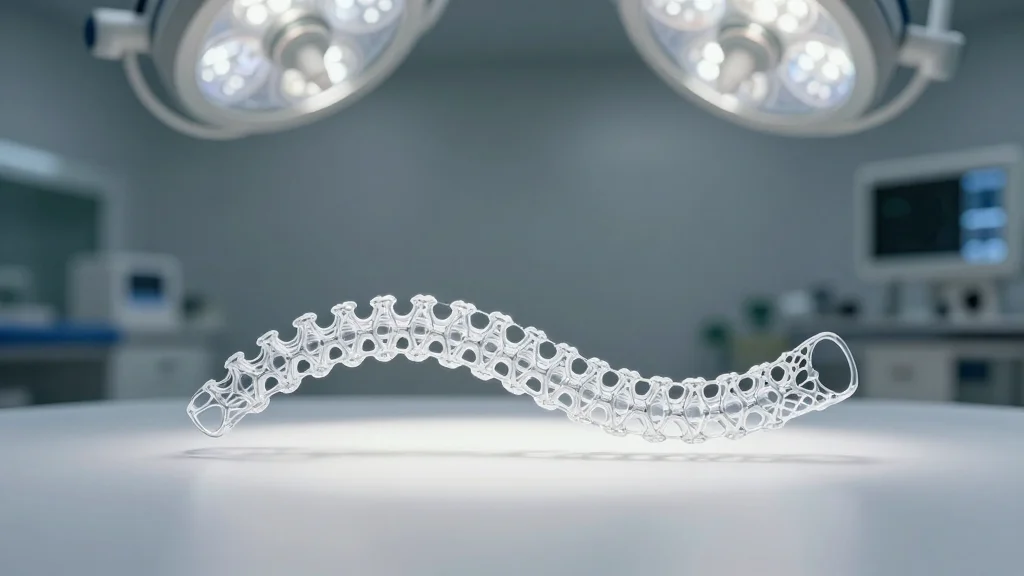
Israeli Team to Perform First Nanotech Spinal‑Cord Implant in Human Trial
Professor Tal Dvir of Tel Aviv University will lead the world’s first human implantation of a nanotech‑enhanced spinal‑cord scaffold, slated for surgery in the coming weeks. The Health Ministry has cleared compassionate‑use trials for eight patients, following mouse studies that...

Protein Biomarkers in Practice: Strategies to Reduce Drug Development Risk
Protein biomarkers are emerging as pivotal tools for reducing risk across the drug development lifecycle. Advances in high‑throughput proteomic platforms now allow real‑time functional insights, enabling stronger target validation, patient segmentation, and measurable efficacy signals. An eBook from GEN compiles...

Clene Secures FDA Pathway to File Accelerated‑Approval NDA for ALS Drug CNM‑Au8
Clene Inc. announced it will submit an accelerated‑approval New Drug Application for its ALS therapy CNM‑Au8 in the third quarter of 2026, following a FDA Type C meeting that said the company’s data could support the pathway. The filing hinges...

More OMQ Letters After FDA Inspections? Don't Treat Them as Noise
Pharmaceutical manufacturers are receiving more post‑inspection letters from the FDA’s Office of Manufacturing Quality (OMQ), even after inspections with only one or two Form FDA 483 observations. The rise aligns with a 27% jump in FY2024 drug quality inspections—62% of...

Axsome Therapeutics, Inc. (AXSM) Q1 2026 Earnings Call Transcript
Axsome Therapeutics held its first‑quarter 2026 earnings conference call on May 4, 2026. CEO Herriot Tabuteau, CFO Nick Pizzie and CCO Ari Maizel presented the company’s financial results, commercial strategy and pipeline milestones. The discussion featured forward‑looking statements about upcoming Phase 3 data and reaffirmed...

Pharma Exports Surpass $31 Billion in FY26 Despite Global Headwinds
India’s pharmaceutical exports exceeded $31 billion in FY 26, marking a robust overall performance despite a sharp 23% drop in March shipments. The decline was driven by a 10% fall in U.S. exports and an 11.5% dip to China, both grappling with...

Pharmaceutical Executive Daily: UCB to Acquire Candid Therapeutics
UCB signed a definitive agreement to acquire privately held Candid Therapeutics for $2 billion upfront with up to $200 million in milestones. The deal, slated to close by late Q2 or early Q3 2026 pending antitrust clearance, adds a suite of T‑cell engager...

Two Strategies for Specialty Drug Savings—And How to Choose the Right One
Specialty drug prices are climbing, prompting employers to seek sustainable savings through two main pathways: alternative funding programs (AFPs) and in‑benefit optimization. AFPs tap manufacturer assistance or non‑traditional sourcing to lower out‑of‑pocket costs for high‑price therapies, but they add coordination...

AAPS NBC 2026 To Highlight Predictive Tools in Drug Discovery with Opening Plenary
The AAPS National Biotechnology Conference 2026 will open with a plenary by Johns Hopkins professor Thomas Hartung, focusing on artificial intelligence and new‑approach methods (NAMs) that enhance predictive toxicology and human‑relevant models. Hartung will detail how AI‑driven in‑vitro systems, organoids...

Novo Nordisk’s Wegovy Head Start on Pills Forces Investors to Rethink Eli Lilly's GLP-1 Dominance
Novo Nordisk’s oral Wegovy pill, launched in January at $149 a month, quickly doubled patient intake for telehealth provider LifeMD and attracted tens of thousands of new GLP‑1 users. The strong start has forced investors to reassess Eli Lilly’s position after...
STAT+: French Regulator Fines Novo and Lilly over Weight Loss Ad Campaigns
France’s medicines regulator ANSM imposed a $2 million fine on Novo Nordisk for misleading advertisements promoting its Wegovy and Saxenda weight‑loss drugs. Eli Lilly was also fined about $127 000 for an ad campaign that indirectly promoted prescription‑only Mounjaro. The actions reflect heightened...
Cancer Patient Advocates Endorse Bill To Allow Reimbursement Of Trial Expenses
Cancer patient advocates, led by the American Cancer Society Cancer Action Network, have endorsed a new bipartisan Senate bill that would allow clinical‑trial sponsors to reimburse patients for travel and lodging costs. The legislation aims to dismantle geographic barriers that...
One Hepatic Failure Case Sparks LLY vs Novo Debate
$LLY $NVO So one case of hepatic failure reported with $LLY Foundayo. To put this in perspective, with the injectables in this class, the reported hepatic incidents have been well under 1 in 100,000. Other oral GLP-1 agonists have also...

Deviation Capital Launches $300M Fund, Seeks Bio‑tech Founders at SynBioBeta
Deviation Capital just launched with a $300M fund target, and founding partner Dusan Perovic will be at #SynBioBeta2026 this week in San Jose looking for the next generation of founders to back. Deviation spun out of @TwoSigmaVC with a clear thesis:...

FDA Search for New CBER Head Focused on Small Group of Final Candidates
The U.S. Food and Drug Administration has narrowed its hunt for a new head of the Center for Biologics Evaluation and Research (CBER) to three or four finalists. CBER is the agency’s hub for overseeing vaccines, blood products, and emerging...
FDA Likely to Fast‑Track KRAS Drug for PR Boost
To try to offset this, I predict the FDA will approve the $RVMD KRAS drug in record time. Not just much quicker than usual. FDA needs a public relations win, and this will be their vehicle.
Gene Therapy Targets Childhood Blindness by Fixing Retinal Gene
Jean Bennett and Albert Maguire developed a gene therapy approach to correct a gene in the retina to treat childhood blindness. https://t.co/WJ99qvcUT1

NIH Study Finds Weekly Semaglutide Cuts Heavy Drinking by 41% When Paired With CBT
NIH scientists and Copenhagen University Hospital investigators found that a 26‑week course of weekly semaglutide injections alongside cognitive‑behavioral therapy lowered heavy‑drinking days by 41.1% in obese patients with alcohol‑use disorder, a 13.7‑point gain over placebo. The result suggests a new,...

Celcuity Bolsters ASCO‑highlighted Breast Cancer Drug Case
Celcuity strengthens case for ASCO-spotlighted breast cancer drug https://t.co/HdekunGXsu by @Lilah_Alvarado $CELC + 18% $NVS $AZN $RHHBY

Romanian 'Project Manhattan' Therapy Begins Human Trials to Reverse Aging
Romanian researchers have launched the first human trials of the experimental "Project Manhattan" therapy, which claims to reverse cellular aging and address dozens of age‑related diseases. The initiative arrives as the global longevity industry is valued at roughly $20 trillion, drawing...

Celcuity Strengthens Case for ASCO-Spotlighted Breast Cancer Drug
Celcuity announced that its experimental PI3K/mTOR inhibitor gedatolisib achieved statistically significant and clinically meaningful disease‑progression delays in two‑ and three‑drug combinations for patients with PIK3CA‑mutated, hormone‑receptor‑positive, HER2‑negative breast cancer. The data will be presented at the ASCO meeting in Chicago...
Tilray’s Rescheduling Rally Fizzles as DOJ Plan Stalls, Shares Slip Below $6
Tilray Brands saw its stock plunge from a brief high of $8 to around $6 after the Justice Department announced only a partial, immediate rescheduling of FDA‑approved medical marijuana products. The limited scope of the move and pending administrative hearing...

RxUtility Launches Personalized AI Companion Mimi™ to Compare Every Drug Price as Affordability Crisis Grows
RxUtility announced the beta launch of Mimi™, an AI‑driven conversational companion that compares real‑time prices for every prescription across U.S. pharmacies. The free‑to‑use service asks users for medication, dosage, insurance and location to surface the lowest‑cost options, including coupons and...

Latus Bio Secures $97 Million Series A to Scale Gene‑Therapy Manufacturing
Latus Bio announced a $97 million Series A financing, including a $43 million extension led by 8VC, to fund its proprietary AAV capsid platform and move two lead programs toward IND filing. The capital raise underscores investor confidence in scalable gene‑therapy solutions...