Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Insilico Medicine Secures IND for AI-Designed Rentosertib Inhalation, First Direct‑to‑Lung Trial
Insilico Medicine announced IND clearance from China's CDE for its AI‑designed Rentosertib inhalation solution, marking the 13th AI‑driven program to reach clinical testing and the first to use a direct‑to‑lung delivery route. The Phase I study will enroll about 80 healthy volunteers and IPF patients, aiming to prove safety and lung‑focused exposure.

Pfizer Settles with Generic Drugmakers to Protect Blockbuster Drug Until 2031
Pfizer reached settlements with three generic manufacturers—Dexcel Pharma, Hikma Pharmaceuticals and Cipla—to postpone the launch of off‑patent copies of its heart‑disease drug Vyndamax until mid‑2031. The agreement stabilizes U.S. sales, which hit $3.8 billion in 2025, and averts the steep revenue...

When ADCs Meet Targeted Protein Degraders: The Emerging Field of Degrader-Antibody Conjugates
The biotech sector is exploring degrader‑antibody conjugates (DACs), a hybrid that merges antibody‑drug conjugate targeting with catalytic protein‑degradation payloads. C4 Therapeutics has expanded its partnership with Roche to co‑develop two undisclosed oncology DAC programs, while Orum Therapeutics secured $100 million to...

Should Pharma Be Swinging Bigger with AI? One Top Researcher Wants to Try
Kyunghyun Cho, a leading AI researcher known for co‑authoring the attention mechanism, used his platform at the International Conference on Learning Representations to argue that pharmaceutical companies should dramatically increase AI investment. He outlined a plan for a $50 million venture...

Teva to Acquire Emalex Biosciences for ~$900M
Teva Pharmaceutical Industries has signed a definitive agreement to acquire Emalex Biosciences for an estimated $900 million, comprising $700 million upfront and up to $200 million in commercial milestones. The purchase brings Emalex’s lead asset, ecopipam—a selective dopamine D1 receptor antagonist—into Teva’s neuroscience...

AstraZeneca Makes Surprise U-Turn with £300m Pharma Investment in UK
AstraZeneca announced a surprise £300 million (≈$381 million) investment in the UK, unfreezing a £200 million (≈$254 million) Cambridge expansion and allocating £100 million (≈$127 million) to a new "lab of the future" in Macclesfield. The move follows a pause last year after the company expressed...

Psychedelics Go Mainstream: Medicine, Mania Or Both?
Psychedelics are re‑emerging as a potential breakthrough in behavioral health, spurred by loosening regulations and a Trump‑issued executive order that accelerated research. Early clinical data suggest benefits for depression, PTSD and other conditions, prompting biotech stocks to rally after FDA...

Amgen, AZ Will Pilot FDA's Real-Time Clinical Trial Plan
The FDA unveiled a plan to receive clinical‑trial data in real time, aiming to accelerate drug development. AstraZeneca and Amgen have agreed to pilot the model with two studies that will stream endpoints and safety signals to regulators as they...

Revolution’s on a Pancreatic Cancer Winning Streak. What Comes Next for the Biotech?
Revolution Medicines announced that its oral RAS(ON) inhibitor daraxonrasib nearly doubled overall survival in previously treated metastatic pancreatic cancer, while its second candidate zoldonrasib showed tumor shrinkage in over half of KRAS‑driven lung cancer patients. The breakthrough data sent the...

The 20 Enterprise AI Drug Discovery & Life Sciences CEOs You Need to Know in 2026
The AI Insider has compiled a roster of the 20 most influential CEOs steering enterprise AI drug discovery and life‑sciences firms in 2026. The list showcases companies that are compressing decade‑long development cycles into months through generative protein design, AI‑optimized...

Drug Trials Snapshot: DAXXIFY
Revance Therapeutics’ DAXXIFY, a daxibotulinumtoxin A formulation, received FDA approval on September 7 2022 for temporary reduction of moderate to severe glabellar lines. Approval was based on two randomized, double‑blind, placebo‑controlled trials (GL‑1 and GL‑2) that enrolled 609 adults across the United States...

HUTCHMED Reports NMPA’s NDA Acceptance Under Priority Review for Sovleplenib to Treat wAIHA
HUTCHMED’s oral JAK2 inhibitor sovleplenib has received NDA acceptance and priority review from China’s National Medical Products Administration for warm‑antibody autoimmune hemolytic anemia (wAIHA). The submission is backed by Phase II/III data showing a 43.8% overall response rate versus 0%...

Drug Trials Snapshots: VOQUEZNA TRIPLE PAK, VOQUEZNA DUAL PAK
Phathom Pharmaceuticals’ VOQUEZNA TRIPLE PAK (vonoprazan, amoxicillin, clarithromycin) and VOQUEZNA DUAL PAK (vonoprazan, amoxicillin) received FDA approval for adult Helicobacter pylori infection based on a 992‑patient trial across six countries. In the modified intent‑to‑treat population, the triple regimen eradicated the...

GSK CEO Says Team Reorg Could Be in the Cards Pending Upcoming Phase 3 Readouts
GlaxoSmithKline (GSK) is banking on a slate of late‑stage Phase 3 trials slated for 2024, spanning oncology drugs and next‑generation vaccines. CEO Luke Miels warned on the first‑quarter earnings call that a series of missed readouts could trigger a reshuffle of...

Drug Trials Snapshot: OPDUALAG
OPDUALAG, a fixed‑dose combo of nivolumab and relatlimab, received FDA approval in March 2022 for adults and adolescents with unresectable or metastatic melanoma. The pivotal trial enrolled 714 patients and showed a median progression‑free survival of 10.1 months versus 4.6...

Drug Trials Snapshots: VONJO
VONJO (pacritinib) received FDA accelerated approval on Feb. 28, 2022 for adult myelofibrosis patients with platelet counts ≤50,000/µL. The decision rests on a single trial that enrolled 63 low‑platelet patients, of whom 31 received VONJO and 32 received best available therapy. In...

Drug Trials Snapshot: PYRUKYND
AGIOS Pharmaceuticals’ mitapivat, marketed as PYRUKYND, received FDA approval in February 2022 for treating hemolytic anemia caused by pyruvate kinase deficiency. In pivotal trials, 40% of non‑transfused adults achieved a ≥1.5 g/dL hemoglobin increase versus none on placebo, while 33% of...

Drug Trials Snapshots: ENJAYMO
ENJAYMO (sutimlimab‑jome) received FDA approval in February 2022 as the first therapy to reduce red‑blood‑cell transfusions in adults with cold agglutinin disease (CAD). Approval was based on a single open‑label, single‑arm trial (CARDINAL) that enrolled 24 patients across eight countries. After...

Drug Trials Snapshots: VABYSMO
VABYSMO (faricimab‑svoa), approved in January 2022, was evaluated in four pivotal trials involving 2,591 patients with neovascular age‑related macular degeneration (nAMD) and diabetic macular edema (DME). The drug was administered monthly for four doses before transitioning to a personalized schedule based...

Drug Trials Snapshot: CIBINQO
Pfizer's oral JAK inhibitor CIBINQO (abrocitinib) received FDA approval in January 2022 for adults with refractory moderate‑to‑severe atopic dermatitis. The approval rests on three phase‑3 trials involving 1,615 patients across 18 countries, which demonstrated robust skin‑clearance outcomes at week 12, especially with...

Drug Trials Snapshot: IMAAVY
IMAAVY (nipocalimab‑aahu) received FDA approval on April 29, 2025 for generalized myasthenia gravis in patients 12 years and older with AChR or MuSK antibodies. In a pivotal 24‑week Phase III trial of 196 adults, the drug achieved a statistically significant 1.5‑point improvement...

MPM Has Collected Three China Drugs for Its ‘Best of Both Worlds’ Strategy
MPM BioImpact, a biotech investment firm, has finalized the acquisition of three China‑origin drug candidates as part of its "best of both worlds" strategy. The deals were brokered through its portfolio company K2 Therapeutics, which now controls three distinct programs...

The Strategic Investments Expanding CDMO Capabilities for HPAPIs and ADCs
Contract development and manufacturing organisations (CDMOs) are accelerating investments to meet soaring demand for highly potent active pharmaceutical ingredients (HPAPIs) and antibody‑drug conjugates (ADCs). The focus is on backward integration, high‑containment infrastructure, and advanced processing such as chromatography and lyophilisation...

FDA Accuses ChemoCentryx of Trial Manipulation in Amgen‑Owned Tavneos Approval
The U.S. Food and Drug Administration has formally accused ChemoCentryx of manipulating results from the pivotal trial that secured approval of its drug Tavneos, now under Amgen’s ownership. The agency says the alleged misconduct could prompt a withdrawal of the...

Leipzig’s Primogene Raises €4.1 Million to Scale Enzymatic Biomanufacturing of Complex Bioactive Molecules
Leipzig‑based biotech Primogene announced a €4.1 million (≈$4.5 million) seed round led by High‑Tech Gründerfonds to expand its enzymatic biomanufacturing platform. The funding will be used to scale production, grow the team, and broaden its IP portfolio and strategic partnerships. Primogene’s enzyme‑driven...

Reduced Ghrelin Receptor Activity Improves Mitochondrial Function and Muscle Function in Aged Mice
Researchers demonstrated that reducing activity of the ghrelin receptor (GHSR‑1a) improves muscle endurance and mitochondrial function in aged mice. Both genetic knockout and the inverse‑agonist PF‑5190457 increased markers of mitochondrial biogenesis and mitophagy, enhancing fatigue resistance. The interventions did not...

FDA Review Lags Keep Terminally Ill Children Waiting for Life‑Saving Drugs
Parents of children with rare, terminal diseases allege the FDA’s slow approval process is denying them life‑saving treatments. Caregivers cite broken promises from FDA leadership and demand faster, risk‑aware pathways as lawmakers push for broader early‑diagnosis reforms.

AstraZeneca Q1 Profit Jumps 5% on Oncology, Rare‑Disease Gains
AstraZeneca posted a first‑quarter profit of $3.081 billion, a 5.5% rise year‑over‑year, as revenue climbed 12.5% to $15.288 billion. The boost came from strong sales of its oncology and rare‑disease medicines, and the company now faces pivotal FDA decisions on its cancer...

Could This Be the First Parkinson's Disease Modifier?
In this brief episode, the host and a GAIN representative discuss GT02287, a novel allosteric modulator targeting the glucocerebrosidase (GCase) enzyme, as a potential disease‑modifying therapy for Parkinson’s disease. They explain how the drug stabilizes the misfolded enzyme’s shape, enhancing...

Arcera and Fosun Sign MoU for Neuroscience Innovation
Arcera Life Sciences and Fosun Pharma have signed a memorandum of understanding to create a long‑term strategic partnership focused on licensing, technology sharing, and neuroscience innovation. The deal taps Fosun’s research and manufacturing capabilities and Arcera’s access to international markets,...

How a Supreme Court Fight over Fish Oil Could Raise Your Prescription Drug Costs
U.S. Supreme Court is hearing Hikma Pharmaceuticals’ challenge to Amarin’s patent enforcement over Vascepa, a fish‑oil drug. The dispute centers on “skinny labeling,” which lets generics launch for unpatented uses while brand patents remain. A decision favoring Amarin could tighten...

Massive Bio Partners with OpenAI to Broaden Clinical Trial Access
Massive Bio has teamed up with OpenAI under the Impact Hours programme to automate clinical‑trial eligibility screening. The AI engine translates complex sponsor criteria into machine‑readable parameters, enabling real‑time, automated patient pre‑screening for oncology and haematology studies. The partnership includes...

Rocket to Sell PRV for $180m to Advance Gene Therapy Pipeline
Rocket Pharmaceuticals has agreed to sell its rare‑pediatric disease priority review voucher for $180 million after the FDA granted accelerated approval for its Kresladi gene therapy. The voucher, issued for addressing a rare paediatric condition, can be used to speed up...

CAR-T Cell Therapies Going in Vivo
Ex‑vivo CAR‑T therapies have saved tens of thousands of patients, but the industry is now pivoting to in‑vivo approaches that can be administered off‑the‑shelf. Over the past year, major pharma players have poured billions into in‑vivo CAR‑T platforms, highlighted by...
Tumor/Lymph Node Dual‐Targeting Ultrasonic Nanoconverter Orchestrates Spatiotemporal ROS Regulation for Dual‐Zone Programmed Sono‐STING Immunotherapy
Researchers have engineered a dual‑targeting ultrasonic nanoconverter (OPD@PSF) that co‑delivers the sonosensitizer protoporphyrin IX and the STING agonist Vadimezan to breast tumors and their draining lymph nodes. High‑power ultrasound at the tumor site generates abundant reactive oxygen species, inducing immunogenic...

Quantum-Designed Drug Hits Nature Biotech Cover, New Pan‑KRAS Inhibitors Available
Our drug from a quantum computer paper made the cover of Nature Biotechnology. It was amazing to hold the physical copy of the journal while visiting our UAE site. We will order a bunch for the office - I love...

FDA Halts Manual Trial Data Entry, AI Opportunity Ignored
The FDA Finally Stopped Re-Typing Clinical Trial Data. That's the AI Story Nobody Is Writing. https://t.co/hAH43sFSTg

Word Games: How Moderna Is Selling Its Newest Vaccine without Using the “V” Word
Moderna’s $776 million federal award for a bird‑flu vaccine is under scrutiny after U.S. officials targeted mRNA technology, prompting the company to warn it may halt late‑stage vaccine programs. Simultaneously, Moderna and Merck are advancing an mRNA‑based cancer treatment, which Merck...
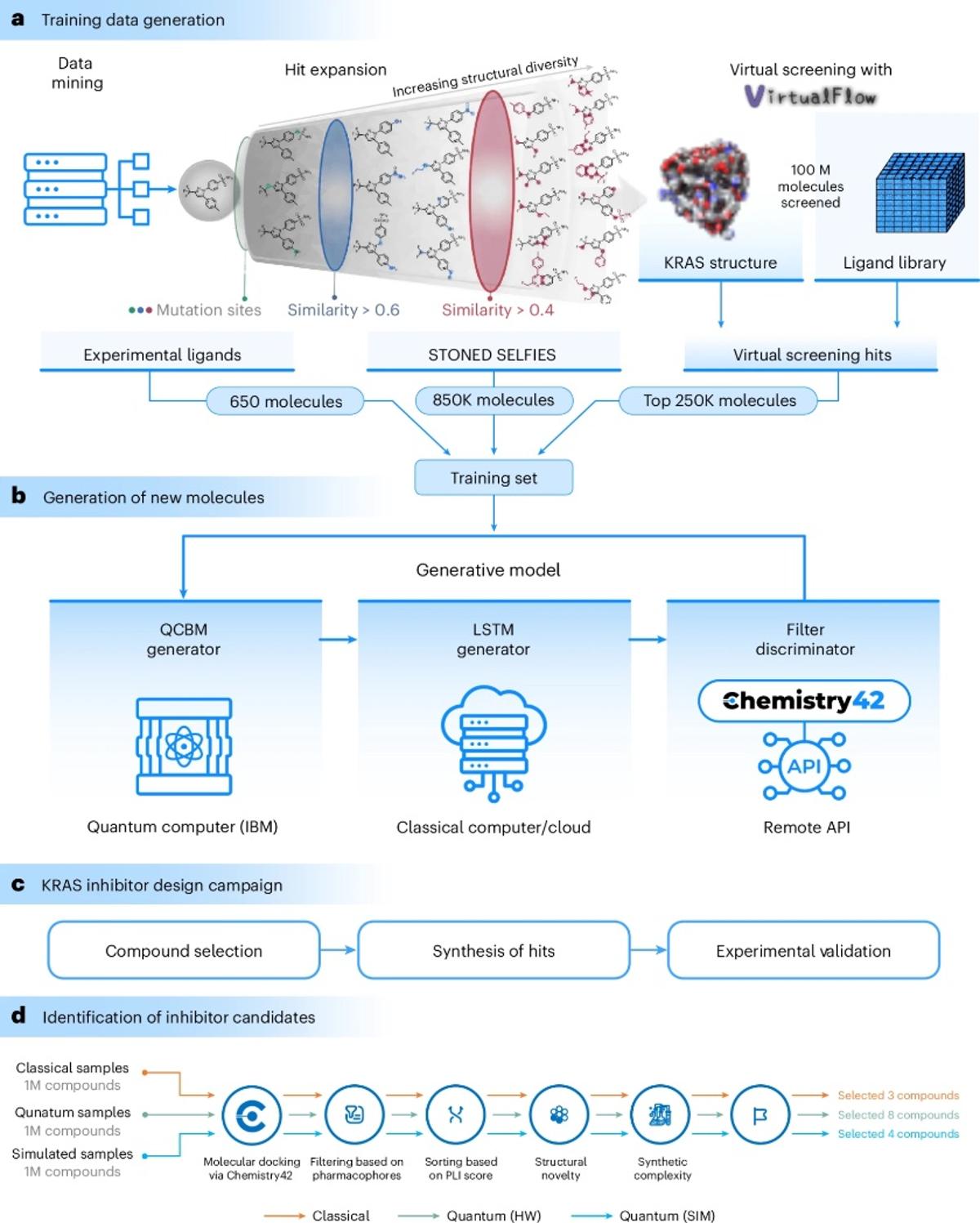
Quantum Algorithm Generates Valid KRAS Inhibitor Hits
Quantum-computing-enhanced algorithm unveils potential KRAS inhibitors 👉 “We introduce a quantum–classical generative model for small-molecule design… his work showcases the potential of quantum computing to generate experimentally validated hits that compare favorably against classical models. @biogerontology https://t.co/s7LjNpu2CP

ONC Secures $20M for Huahi's Hexavalent Trispecific
If you're wondering what $ONC got for its $20 m, here's the #AACR25 detail on Huahi's HH160, a symmetrical hexavalent trispecific anti PD-1 x CTLA-4 x VEGF-A MAb https://t.co/Y0jVjTuMMo

DOJ Reclassifies Medical Marijuana to Schedule III: Key Implications for Florida and National Cannabis Operators
The U.S. Department of Justice has reclassified state‑licensed medical marijuana from Schedule I to Schedule III, the most significant federal cannabis reform in five decades. The change applies only to FDA‑approved cannabis products and state‑run medical programs, leaving adult‑use and illicit markets...

Real Drug Development Experience Needed to Validate AI Claims
If you have never gone through even one cycle of the whole drug development process, it is hard for me to believe your AI solution is ‘transformative’ for this process…

CRL Release Update – No Litigation War yet, but New Citizen Petition Beats the Drums
The FDA has begun posting Complete Response Letters (CRLs) for unapproved NDAs, ANDAs, and BLAs, releasing 127 documents to date, including 36 that had never been publicly disclosed. A Washington‑based law firm filed a Citizen Petition on April 20, 2026,...

Jazz Pharma’s Ziihera Combos Get FDA Priority Review for First‑line HER2‑positive Gastric Cancer
Jazz Pharmaceuticals announced that the FDA has accepted its supplemental Biologics License Application for Ziihera (zanidatamab‑hrii) combinations and placed the filing under priority review. The decision sets a PDUFA target action date of August 25, 2026, and follows breakthrough‑therapy designation...

Eli Lilly Inks AI Partnership with Profluent, Eyeing $2.25 B in Gene‑editing Milestones
Eli Lilly announced a collaboration with AI‑focused biotech Profluent to create advanced gene‑editing tools that could insert whole genes. The agreement may trigger $2.25 billion in milestone payments, highlighting Lilly’s rapid expansion into genetic medicines.

Sanofi Appoints Belén Garijo as CEO, Vows to Curb $18.4 Bn Dupixent Reliance
Sanofi announced Belén Garijo will assume the chief‑executive role on April 29, inheriting a business where Dupixent generates $18.4 bn in annual revenue. Garijo’s mandate includes leveraging a $30 bn M&A war chest to broaden the pipeline before Dupixent’s 2031 patent expiry.

Independent, Academic Cancer Trials Are Vital to Improve Patient Outcomes Worldwide
A Lancet Oncology Commission has been launched to evaluate the role of independent, academic cancer trials worldwide. The initiative stems from a coalition of 35 investigators and patient advocates spanning six continents, coordinated by the European Organisation for Research and...

Novartis' Rhapsido Wins EU Approval as First Oral Treatment for Chronic Spontaneous Urticaria
Novartis' oral BTK inhibitor Rhapsido (remibrutinib) has secured European Commission approval, marking the first oral targeted therapy for chronic spontaneous urticaria (CSU) in the EU. The decision follows a positive CHMP opinion in February 2026 and opens a new market...

Johnson & Johnson Halves Drug Lead‑optimization Time with AI
Johnson & Johnson announced that its artificial‑intelligence platform has reduced the time required to generate drug‑development leads by half. CIO Jim Swanson highlighted accelerated progress on an oncology and an immunology compound, underscoring AI’s growing role in pharma R&D.
FTC Settlement Compels Express Scripts to Offer Cheaper Insulin Options
The Federal Trade Commission reached a settlement with Cigna's Express Scripts, the first of three major pharmacy benefit managers to be sued over insulin pricing. The agreement obligates the PBM to place the lowest‑cost drug on patients' formularies, though it...