Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
FDA Clears Eli Lilly’s Foundayo, First New Oral Anti‑Obesity Drug in Years
The U.S. Food and Drug Administration approved Eli Lilly’s once‑daily oral weight‑loss pill, Foundayo, making it the first new anti‑obesity medication in years and only the second oral GLP‑1 option. Priced between $149 and $349 a month, the drug promises a simpler regimen that could broaden access to obesity therapy.

FDA Grants RMAT Status to Orca-Q in High-Risk Hematologic Malignancies
The U.S. Food and Drug Administration has awarded Regenerative Medicine Advanced Therapy (RMAT) designation to Orca‑Q, an investigational allogeneic T‑cell immunotherapy targeting high‑risk hematologic malignancies. The designation follows early phase 1 data that hint at improved overall survival, lower graft‑versus‑host disease...

Pfizer Deals Extend Patent Life for a Top-Selling Rare Disease Drug
Pfizer announced settlements with Dexcel Pharma, Hikma Pharmaceuticals and Cipla that push the patent expiry of its rare‑disease drug Vyndamax to June 1, 2031. The deals delay generic tafamidis entry, keeping U.S. sales stable through the early 2030s after the company warned...

FDA Grants Priority Review and Breakthrough Designation to GSK‑Ionis Hepatitis B Drug Bepirovirsen
The U.S. FDA has accepted Ionis Pharmaceuticals’ bepirovirsen for priority review and awarded it breakthrough therapy designation, setting a PDUFA target date of Oct. 26, 2026. The antisense drug, developed with GSK, could become the first therapy to achieve meaningful functional cures...

Restylane, Sculptra Combo Improves Skin Dryness, Collagen Loss for Menopausal Women
Interim data from two investigator‑initiated trials show that a sequential regimen of Restylane Skinboosters followed by Sculptra markedly improves skin hydration, firmness and collagen density in menopausal women. The 9‑month study reported peak patient satisfaction at month six, with measurable...

Scientists May Have Finally Created a Male Birth Control Pill—So Far, It Shows Very Few Side Effects
YourChoice Therapeutics reported that its experimental male contraceptive pill YCT‑529 was well‑tolerated in a Phase 1 trial of 16 healthy, vasectomized men. The non‑hormonal compound halted sperm production without altering testosterone, heart rate, mood, or sexual desire. Pre‑clinical work in mice...

YURTEC Corp Reports 9% Drop in Full-Year Profit as Revenue Slides 1.9%
YURTEC Corp announced full-year earnings of ¥10.33 bn ($68.8 m), a 9% decline from the prior year, while revenue fell 1.9% to ¥252.3 bn ($1.68 bn). The drop underscores pressure on the Japanese antibody‑drug conjugate developer as it balances R&D spending with market expectations.

New York Proposes an Additional Layer of Legal Protection to Those Profiting From Injecting Vaccines
New York is introducing Senate Bill A9140, which would grant blanket legal immunity to doctors, pharmacists and other entities that prescribe, dispense, order, furnish, or administer vaccines to minors. The bill bars any civil lawsuit alleging injury, including residual effects,...

Charting FDA‑Guided Trial Pathways for Merkel Cell Carcinoma
Really enjoyed my fireside chat at MCC4 with Harpreet Singh, MD, the CMO at Precision Medicine, focused on establishing pathways for clinical trials in Merkel cell carcinoma. The physician-scientists in the audience were clearly excited about the prospect of working...

STAT+: Supreme Court to Hear Case About ‘Skinny Labeling’ and Generic Access
The U.S. Supreme Court will hear oral arguments on a case involving “skinny labeling,” a practice where generic manufacturers seek approval to market a drug for only a subset of its approved uses. By limiting the label, generics aim to...
$739 Million Acquisition of XOMA Strengthens Ligand’s Biopharma Portfolio
Ligand Pharmaceuticals announced a $739 million acquisition of XOMA Royalty, expanding its royalty‑financing portfolio to over 200 assets. The deal adds more than 120 new royalty streams, including seven marketed drugs such as Vabysmo and Ojemda, and deepens exposure to oncology,...
Ashvattha Therapeutics Announces Presentations Highlighting Mechanism of Action for Migaldendranib in Diabetic Macular Edema and Neovascular Age-Related Macular Degeneration
Ashvattha Therapeutics presented Phase 2 data on its subcutaneous nanomedicine migaldendranib (MGB) for diabetic macular edema and neovascular age‑related macular degeneration at ARVO. The two‑stage trial showed stable central subfield thickness for up to 12 weeks and maintained visual acuity without...

Cellares and Cabaletta Bio Sign 10-Year Commercial Supply Agreement to Scale Rese-Cel
Cellares has entered a 10‑year commercial supply agreement with Cabaletta Bio to manufacture rese‑cel, the company’s autologous CAR‑T therapy for autoimmune diseases, using its fully automated Cell Shuttle and Cell Q platforms. The deal secures long‑term capacity to produce thousands of...
BPL-003
Great piece from @Shea_ARK "AtaiBeckley is advancing BPL-003—an intra-nasal formulation derived from 5-MeO-DMT—in treatment-resistant depression, with positive Phase 2b results supporting a Phase 3 initiation in the second quarter of 2026.[11] With FDA Breakthrough Therapy designation, BPL-003 has the potential...

Rocket's PRV Goes for $180M; Oruka Targets $500M Offering
New Jersey‑based Rocket Pharmaceuticals sold its pediatric‑disease priority‑review voucher for $180 million, setting a near‑record price for such assets. The cash infusion strengthens Rocket’s balance sheet as it advances late‑stage gene‑therapy candidates. Meanwhile, biotech firm Oruka Therapeutics announced plans to raise...

Science in Space
NASA astronaut Chris Williams and ESA astronaut Sophie Adenot conducted the DNA Nano Therapeutics‑3 experiment in the Kibo laboratory’s Life Science Glovebox aboard the International Space Station. The study explores DNA‑inspired assembly techniques to fabricate nanostructured cancer therapies such as chemotherapy and...

BeOne Medicines Licenses Trispecific Antibody Targeting PD-1, CTLA-4 and VEGF
BeOne Medicines, the rebranded former BeiGene, announced a licensing and option agreement to acquire a trispecific antibody that simultaneously targets PD‑1, CTLA‑4, and VEGF. The experimental biologic originates from a Chinese biotech and is positioned for development in solid‑tumor indications....

Boehringer, Zealand’s Obesity Drug ‘More Akin’ to Novo’s Wegovy in Phase 3
Boehringer Ingelheim and Zealand Pharma reported that their glucagon/GLP‑1 dual agonist survodutide achieved a 13.4% (39.2 lb) average weight loss after 76 weeks in the Phase 3 SYNCHRONIZE‑1 trial, meeting primary endpoints and reducing waist circumference. Analysts compared the outcome to Novo...

AACR 2026: Combo Therapies by Moderna, Marengo Show Promise in Skin, Breast Cancer
Combination regimens dominated AACR 2026, with biotech firms showcasing new cocktail data across multiple tumor types. Marengo Therapeutics reported that its dual‑T‑cell agonist invikafusp alfa combined with Gilead’s ADC Trodelvy produced complete or partial responses in 40% of heavily pretreated...

Pregnancy’s Progesterone: Natural Calm, Muscle Relaxant, Immune Tolerance
Ever notice how some pregnant women seem so calm, almost like they are in an altered state? That is progesterone. Progesterone rises dramatically during pregnancy. Outside of pregnancy, your progesterone levels are less than 1 ng/mL. During your luteal phase, it...

Executing Biologics Technology Transfer Through Integrated Operating Models
The article advocates replacing the traditional sequential biologics tech‑transfer model with an integrated operating model that colocates development, manufacturing, MSAT and quality teams. By running small‑scale experiments and large‑scale runs concurrently, organizations can observe scale‑dependent effects in real time and...

Drug Pricing, PBM Reform, and the 2026 Midterms: What You Need to Know
Analysts at Avalere Health’s AXS26 summit warned that the 2026 midterm elections will reshape drug pricing and PBM practices. Recent FTC settlements require Express Scripts, CVS Caremark and Optum Rx to end spread pricing and unlink compensation from rebates, while...
Milestone for Crispr: First-of-Its-Kind Gene Editing Treatment Successfully Passes Clinical Trial
Intellia Therapeutics announced that its CRISPR‑based, one‑time gene‑editing therapy Lonzo‑z cleared a Phase 3 trial for hereditary angioedema, cutting swelling attacks by 87%. The double‑blind, placebo‑controlled study treated 52 of 80 patients and left 62% attack‑free without ongoing medication. Results represent...

STAT+: Boehringer’s Obesity Drug Shows 16.6% Weight Loss; More Data Needed
Boehringer Ingelheim reported that its experimental obesity drug produced an average 16.6% weight loss in a mid‑stage trial, signaling strong efficacy against a disease affecting over 100 million Americans. The study involved roughly 300 participants over 68 weeks and showed improvements...

Probiotic Candidate Shows Potential to Suppress UTIs
Researchers reported that the probiotic strain Limosilactobacillus reuteri 3613‑1, which produces the antimicrobial compound reuterin, can inhibit key urinary and vaginal pathogens. In a randomized, double‑blind, placebo‑controlled trial of 130 healthy women taking the probiotic daily for 24 weeks, the...
FDA Approves Breztri for Patients 12 Years and Older with Asthma
The FDA has approved Breztri Aerosphere, a single‑inhaler triple‑combination of budesonide, glycopyrrolate, and formoterol, for maintenance treatment of asthma in patients aged 12 and older. This marks the first U.S. approval of a triple therapy that adds a long‑acting muscarinic...
Erasca Plummets Despite 'Phenomenal' Results In Pancreatic Cancer
Erasca (ERAS) shares tumbled more than 45% to $10.50 after a patient in its pancreatic cancer trial died from pneumonia, a known side effect of the RAS‑blocking drug class. The death occurred when the patient withdrew from supportive care, despite...

Updated: AbbVie Flirts with Popular Pan-RAS Field in up to $1.45B Option to Acquire Kestrel
AbbVie has signed a deal giving it an option to acquire Kestrel Therapeutics for up to $1.45 billion, targeting Kestrel’s pan‑RAS inhibitor platform. The agreement includes an upfront payment and milestone‑based earn‑outs tied to regulatory and commercial milestones. Kestrel’s lead candidate,...

Eli Lilly’s Zepbound Sales Surge and Launch of Oral GLP‑1 Drug Foundayo Boost Weight‑Loss Play
Eli Lilly reported a rapid acceleration in sales of its anti‑obesity medicine Zepbound and introduced Foundayo, an oral GLP‑1 therapy, expanding its addressable weight‑loss market. Analysts see the moves as a hedge against emerging rivals and a catalyst for the...

FDA Claims Trial 'Manipulation' Tainted Approval of ChemoCentryx Drug
The FDA has accused ChemoCentryx of manipulating key efficacy data in the pivotal Phase III trial that secured approval for its rheumatoid‑arthritis drug Tavneos. The agency says the new information renders the trial "tainted" and is moving to pull the treatment...

Eli Lilly and NVIDIA Deploy 1,016‑GPU LillyPod Supercomputer to Speed AI Drug Discovery
Eli Lilly has launched LillyPod, a dedicated AI supercomputer built with 1,016 NVIDIA Blackwell Ultra GPUs, to accelerate drug discovery. The Indianapolis facility works alongside Lilly’s wet lab in San Francisco, promising faster analysis of legacy data and new molecular designs...

The US FDA Grants Priority Review and Breakthrough Therapy Designation to GSK’s Bepirovirsen for Chronic Hepatitis B
The U.S. FDA has accepted GSK’s new drug application for bepirovirsen, granting both priority review and breakthrough therapy designation for chronic hepatitis B. The decision is based on Phase III B‑Well 1 and B‑Well 2 trials across 29 countries, which showed higher functional‑cure...
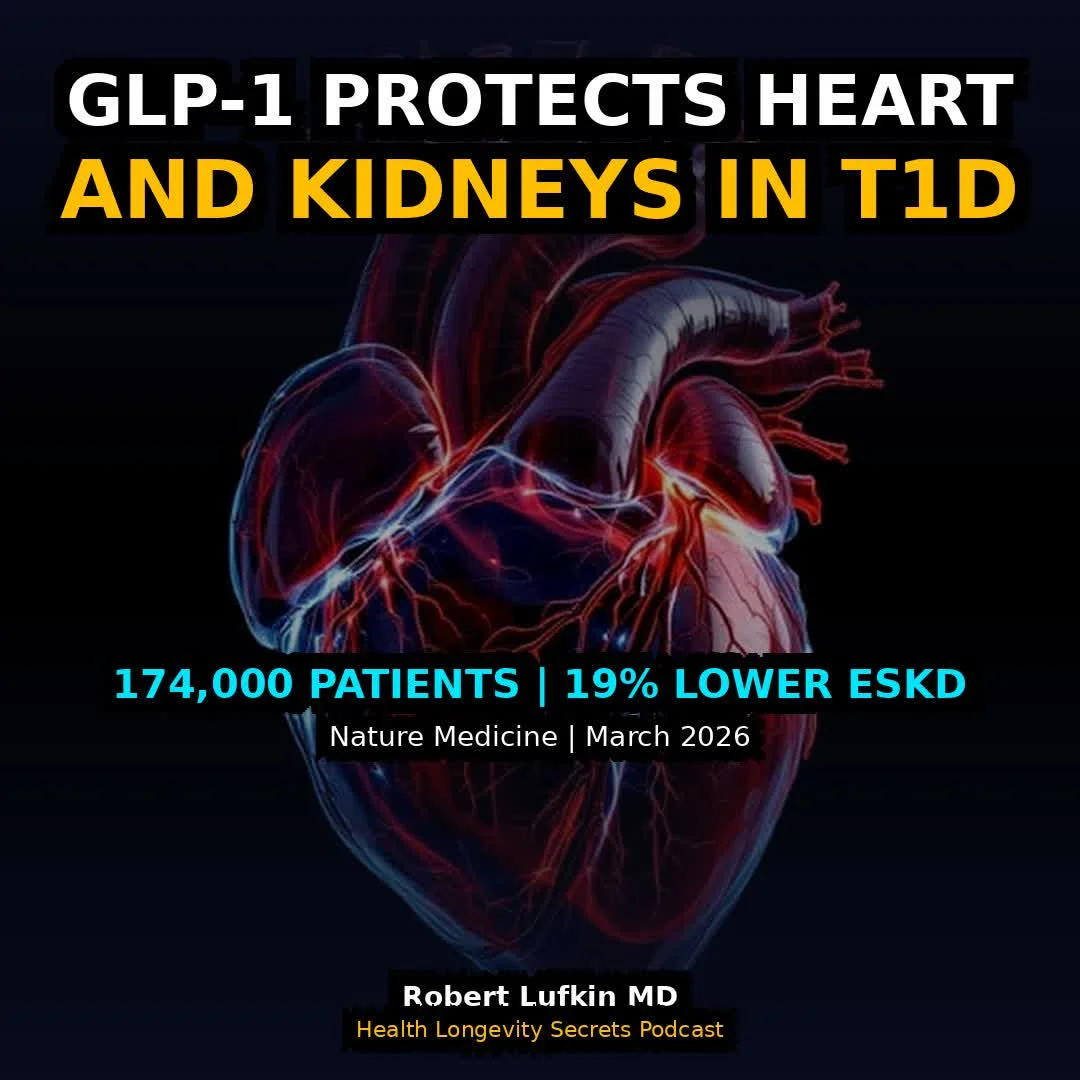
GLP‑1 Therapy Shows Benefit for Type 1 Diabetes Patients
A 174,000-patient analysis just delivered the first hard evidence that GLP-1 drugs work in a population every clinical trial has excluded. As a medical school professor, I teach that Type 1 diabetes is autoimmune, Type 2 is metabolic. But the cardiovascular...

AI Protein Design Advances, Yet Developability Remains Overlooked
Some new protein design competition results from Rio, showing the field keeps tackling harder and harder targets w/ AI methods Might be time to factor in other properties (developability, manufacturability)? https://t.co/h1niVwjnSS

U.S. Medical Centers Need a New Model for Drug Discovery and Development
For more than half a century U.S. academic medical centers (AMCs) have supplied the majority of FDA‑approved drug patents, but China’s rapid R&D expansion threatens that dominance. Chinese biotech now leads in novel medicine approvals and offers clinical trials that...
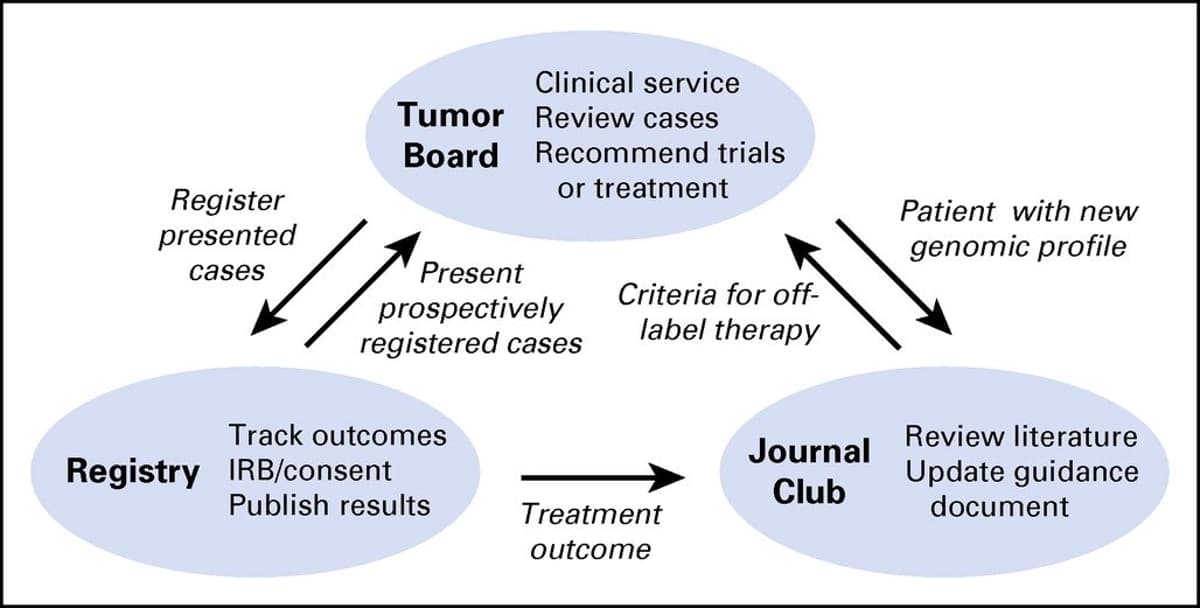
Integrated Academic-Community Tumor Board Enhances Precision Medicine
Implementation & Utility of an Integrated Academic-Community #PrecisionMedicine Molecular Tumor Board [MTB] [Jul 5, 2017] @meburkard et al. @JCOPO_ASCO https://t.co/HOWIx826Lm https://t.co/1JmBdW6K5c

Molecular Tumor Boards Cut Costs, Reduce Recommendation Discordance
Quantifying the Value of Molecular Tumor Board: Discordance Recommendation Rate & Drug Cost Avoidance [10/20/22] Walters @JimW9200 @antonyruggeri @mullane_michael Hunt, Wilson, Ramczyk, @mtmdphd @JCOPO_ASCO https://t.co/OORXChBGjx #PrecisionMedicine #hpeonc @Aurora_Cancer https://t.co/ZUQXq4npfh
As Biosimilars Gain US Traction, Patent Thickets Are Under More Scrutiny
Sun Pharma announced a $12 billion acquisition of Organon to accelerate its entry into the global biosimilar market. The FDA has now approved roughly 90 biosimilars, which account for about 23 % of the U.S. biologics market, reflecting growing competition. Patent thickets—large...
Overcoming Barriers to Community Oncology Precision Medicine
Implementing #PrecisionMedicine Programs and #ClinicalTrials in the Community-Based Oncology: Practice: Barriers and Best Practices - @JLErsek @lorablk @mtmdphd & @EdKimMDLCI #ASCO18 Education Book https://t.co/ahsmfePatk #NCORP

PurIST Classifier Validated for Tailored Pancreatic Cancer Therapy
Real-World Validation of the Purity Independent Subtyping of Tumors Classifier for Informing Therapy Selection [PurIST @TempusAI] in Pancreatic Ductal Adenocarcinoma [Sep 4, 2025] @stephwen et al. @JCOPO_ASCO https://t.co/R24DErvaB4 #pancsm #PrecisionMedicine https://t.co/KNJHtWovqT
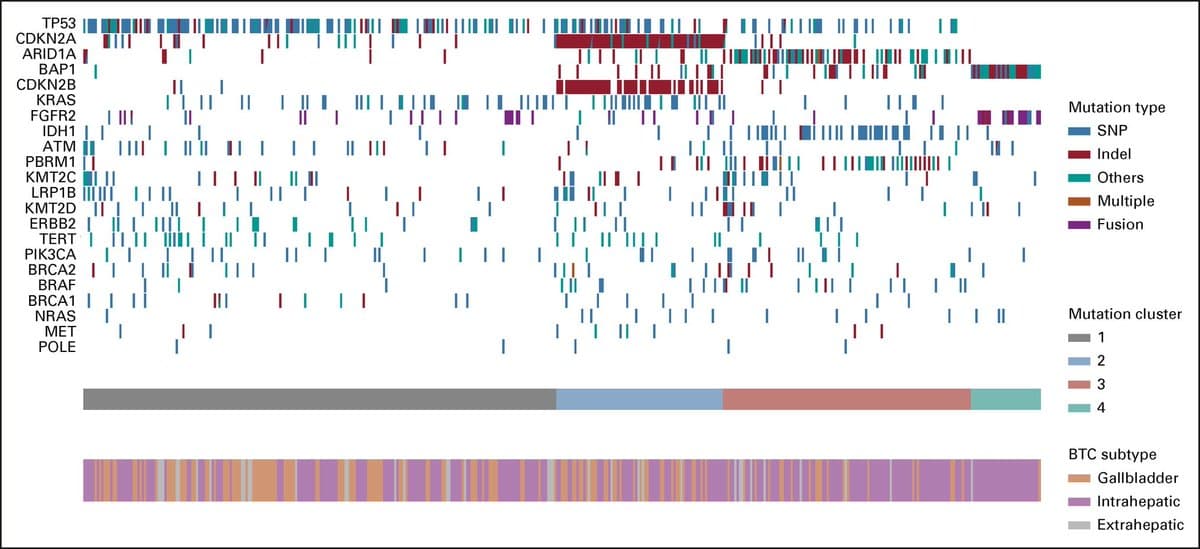
Biliary Cancer Subtypes Show Distinct Immune Signatures
Clinical, Genomic, and Transcriptomic Data Profiling of Biliary Tract Cancer Reveals Subtype-Specific Immune Signatures [Jun 8, 2022] @kabir_mody et al. @MarkYarchoan @JCOPO_ASCO https://t.co/IN6UsL5jvN #hpbcsm #PrecisionMedicine https://t.co/p3BzJ82oxA

BRAF Class II/III Mutations Represent Unmet Clinical Need
Real-world clinical genomic analysis of Pts w/ BRAF mutated cancers identifies BRAF class II & III as a population of unmet medical need [3/7/22] Severson etal #ESMO2240P @TempusAI https://t.co/s5PEBEczlv @Annals_Oncology DOI: https://t.co/c3wxWyUuX0 #PrecisionMedicine #caxtx
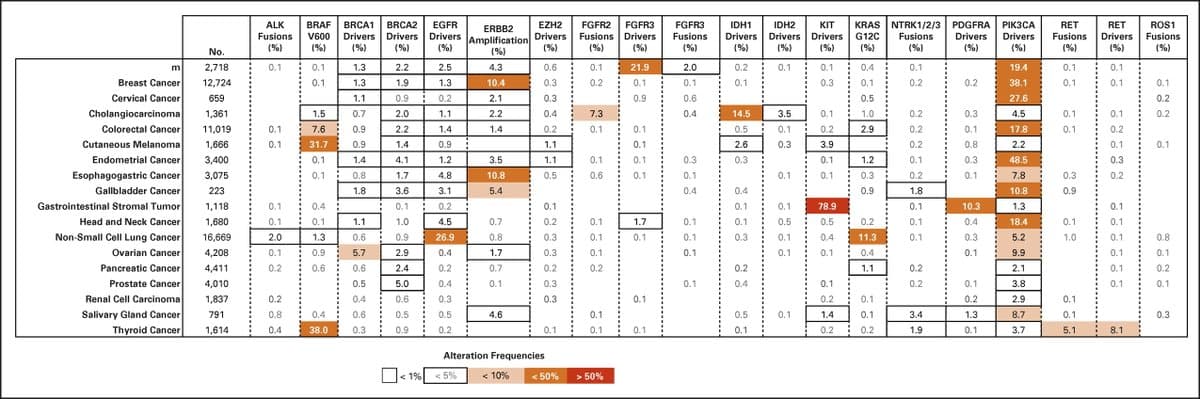
ASCO Endorses Somatic Genomic Testing for Advanced Cancers
Somatic Genomic Testing in Patients With Metastatic or Advanced Cancer: @ASCO Provisional Clinical Opinion [Feb 17, 2022] @CDebyaniPhD et al. @JCO_ASCO https://t.co/hfHUBS3Unx #PrecisionMedicine https://t.co/GwYOMRD1hh
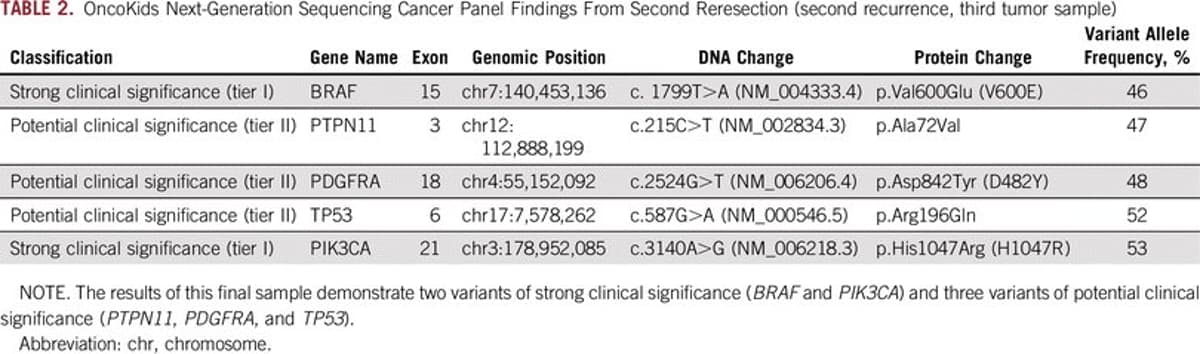
BRAFV600E‑mutated Pediatric Gliofibroma Shows Fleeting Therapy Response
Rare Pediatric Invasive Gliofibroma Has BRAFV600E Mutation and Transiently Responds to Targeted Therapy Before Progressive Clonal Evolution [Mar 27, 2019] @KanevaMD et al. @JCOPO_ASCO https://t.co/oNbfrK7EZr #PrecisionMedicine #btsm https://t.co/V7k9vEiqKm
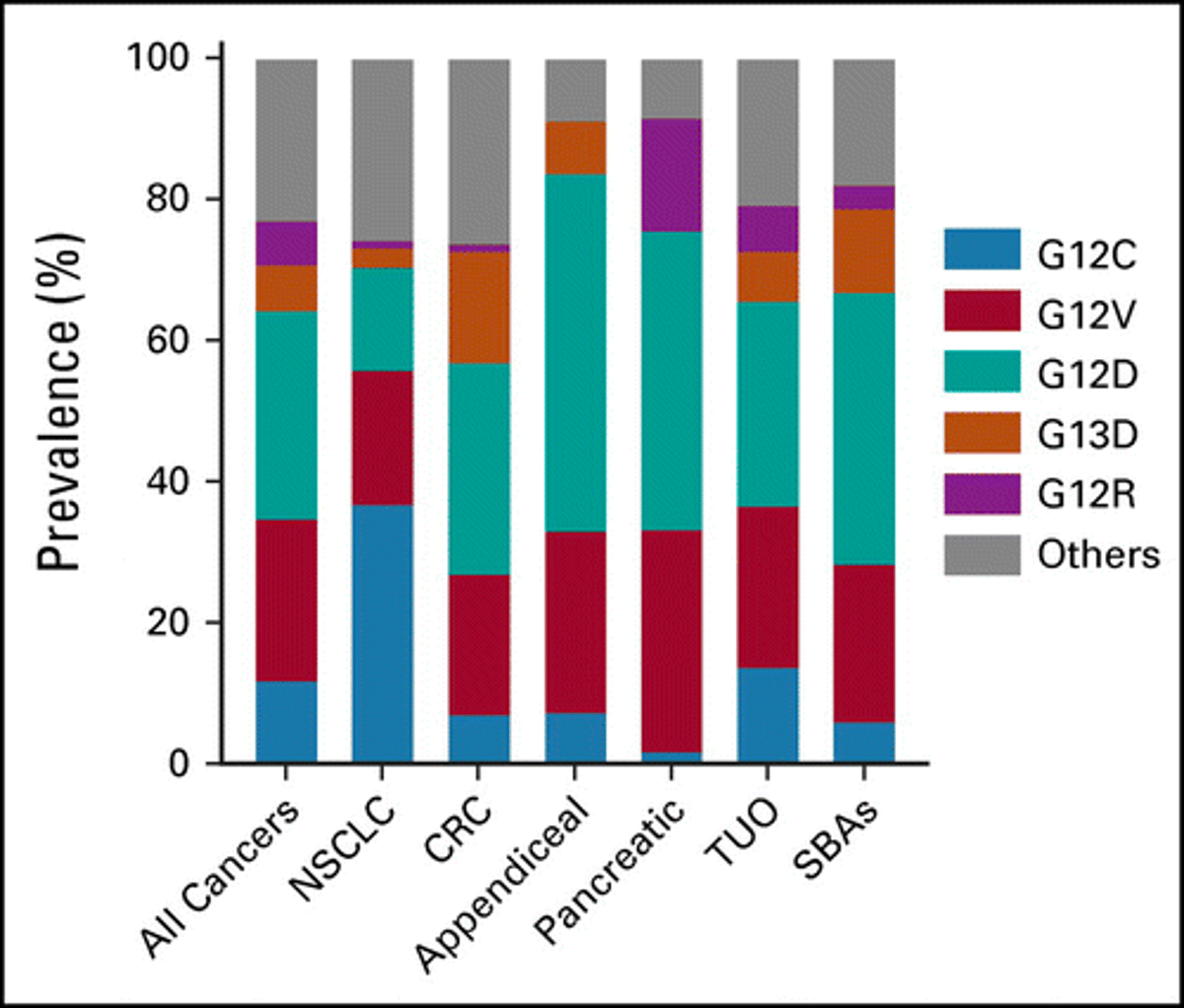
KRASG12C Mutations Linked to Distinct Immuno‑oncology Biomarkers
Landscape of KRASG12C, Associated Genomic Alterations, and Interrelation With Immuno-Oncology Biomarkers in KRAS-Mutated Cancers [Mar 23, 2022] @SalemGIOncDoc et al. @JCOPO_ASCO https://t.co/iehC82lyA4 #ImmunoOnc #PrecisionMedicine @TempusAI https://t.co/wZzfdnBoXU

Comprehensive Genomic Panel Broadens Community Oncology Treatment Options
Unique Features of a Comprehensive Genomic Profiling Panel: Expanding Treatment Options in a Value-Based Community Oncology Network [Mar 9, 2026] La Porte et al. @DrEzraCohen @JCOPO_ASCO https://t.co/tDi3kbxdvX #PrecisionMedicine #hemeonc @TempusAI https://t.co/6QKo7c3oH2

FDA Accuses Amgen of Data Manipulation; Erasca Falls
FDA claims Amgen drug data were ‘manipulated’; Erasca slides despite ‘home run’ results https://t.co/UbftjXwEwa $AMGN $ERAS - 53% $RCKT $JANX
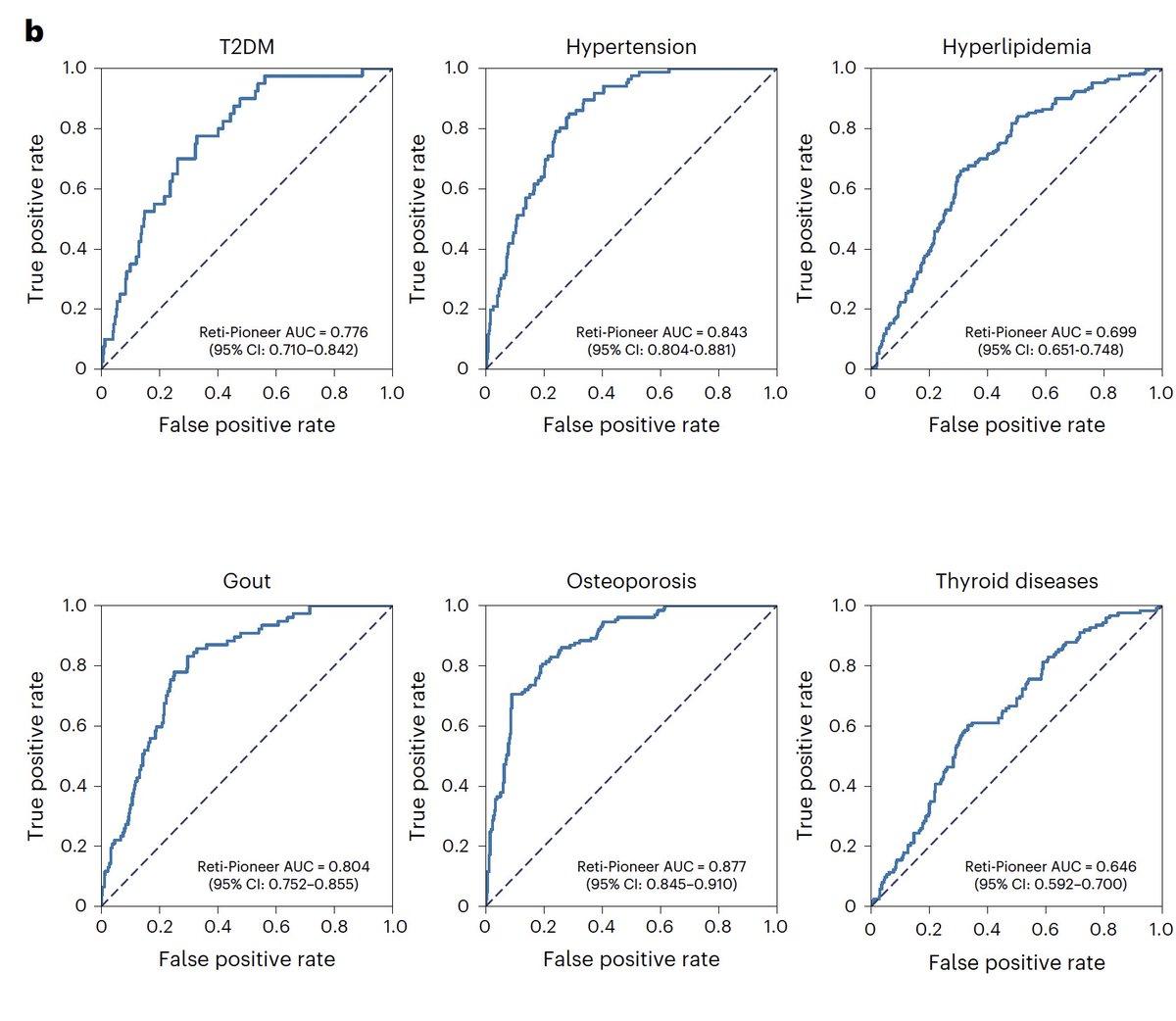
AI Reads Retinal Images to Screen Multiple Diseases
What superhuman vision can detect from the retinal photo, which human eyes cannot, is stunning. A new foundation AI model screening for diabetes hypertension, hyperlipidemia, gout, osteoporosis, and thyroid disease @NatureMedicine https://t.co/GhKvUqz4Vy https://t.co/iKcXCbLceu
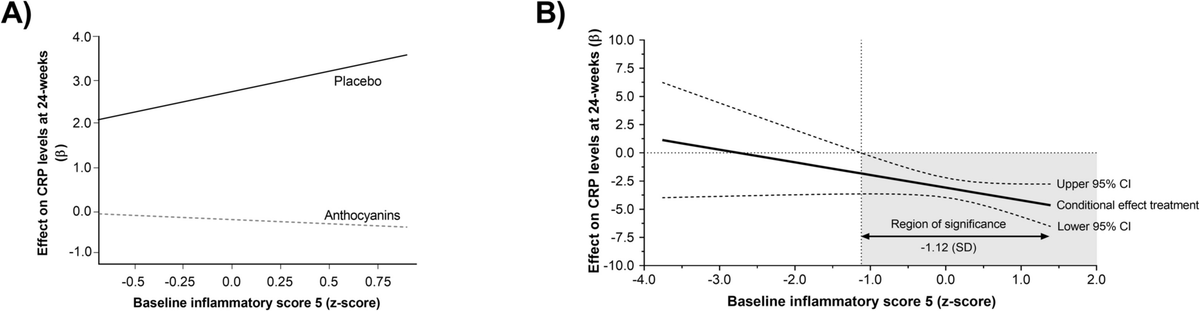
Anthocyanins Improve Cardiometabolic and Anti‑
Anthocyanin supplementation in adults at risk for dementia: a randomized controlled trial on its cardiometabolic and anti-inflammatory biomarker effects https://t.co/tHwVYQ3yvQ https://t.co/cxiRpkIj7W

Coultreon Secures $125M to Test Former Galapagos Immune Drug
Coultreon banks $125M to support testing of former Galapagos immune drug https://t.co/PUsEu2i7Vv by @Lilah_Alvarado $GLPG #biotech #startups