How Eli Lilly LDL Therapy VERVE 102 Could End Heart Disease
Eli Lilly’s Verve 102 gene‑editing therapy aims to eradicate high LDL cholesterol with a single intravenous infusion, targeting the PCSK9 gene in liver cells. The phase‑1 trial involved 35 participants, many already on high‑intensity statins, and achieved an average 62% drop in LDL and an 88% reduction in PCSK9 levels, with effects persisting for up to 18 months and no dose‑limiting toxicities reported. The data leverages a base‑editing CRISPR platform that swaps a single DNA base, permanently silencing PCSK9 without cutting the chromosome. Researchers highlighted that the edit is durable as edited hepatocytes divide, while the mRNA and guide RNA degrade after delivery. Safety signals were limited to mild infusion reactions, transient ALT elevations, and isolated fatigue, though a single case of acid‑reflux raised a question about rare adverse events. Prominent voices, including David Sinclair’s tweet and Verve co‑founder Dr. Sakar Cathther’s claim that a one‑time dose could mimic lifelong genetic protection, underscore the excitement. Yet the discussion also flagged unresolved concerns: a modest association between PCSK9 loss‑of‑function and new‑onset diabetes, and Eli Lilly’s hinted subscription‑style pricing for a permanent genetic cure. If approved, Verve 102 could transform cardiovascular care from chronic statin or monoclonal‑antibody regimens to a one‑off gene therapy, reshaping pricing models, insurance coverage, and patient adherence. However, regulatory timelines push market entry to the early 2030s, and long‑term safety and cost‑effectiveness will determine whether the therapy fulfills its promise of ending cholesterol‑driven heart disease.

Is Psychedelic Therapy Ready for FDA Approval?
The conversation with UCSF neuroscientist Robin Carhart‑Harris examines whether psychedelic‑assisted therapies are poised for FDA approval. He frames the treatment as a combination of a drug that opens a "plastic" mental state and a carefully managed therapeutic context that must...
A Man with Terminal Glioblastoma Was Given 12–15 Months to Live.
The video follows a man diagnosed with terminal glioblastoma, given a 12‑15‑month prognosis, who is now on his fourth dose of Anktiva and whose latest MRI returned a normal result. Dr. Patrick Soon‑Shiong, the billionaire surgeon who created the drug,...
Peer AI - Medical Writing Platform for FDA Submissions - Life Sciences Today Podcast Episode 63
The Life Sciences Today podcast featured Anita Modi, founder and CEO of Pure AI, unveiling the company’s AI‑driven platform that automates the creation and management of FDA regulatory documents. After starting as a medical‑writing drafting tool, Pure AI now...

Menopause, Part 1: What It Actually Is and the 24-Year WHI Correction
The episode launches a menopause series after an opening plug for a new book, Signal, on testosterone misuse and hormone evaluation. Hosts trace two centuries of medical missteps around menopause—from early quack organotherapy and 20th-century estrogen promotion to the 2002...
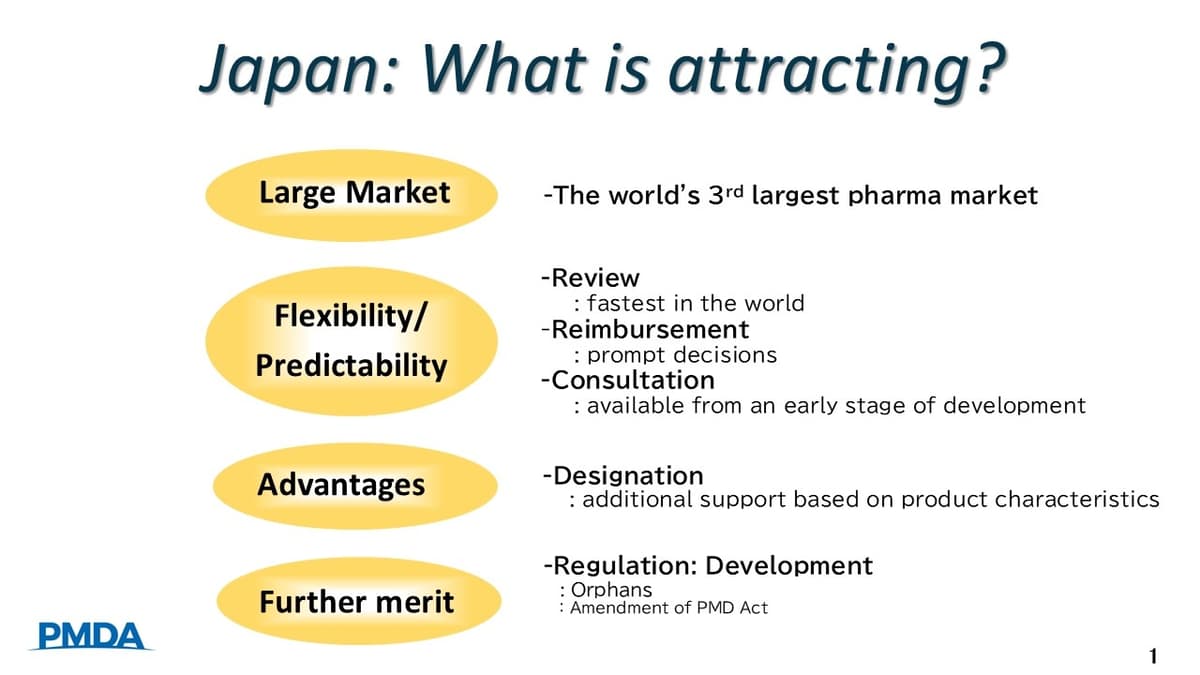
Why Japan for Drug Development Vol.2 Taking the Next Steps with Your Drug in Japan
The webinar, co‑hosted by NIK Biotechnology and sponsored by Nexera, focused on the latest PMDA initiatives that make Japan an increasingly attractive market for drug developers. Speakers highlighted Japan’s position as the world’s third‑largest pharmaceutical market, the transparency of PMDA’s...

Ebola Virus BDBV Fundamentals and Best Hope for Treatment
The video focuses on the rapidly expanding Bundibugyo Ebola virus (BDBV) outbreak in the Democratic Republic of Congo and neighboring Uganda, which has surpassed 700 suspected cases and 176 confirmed deaths as of May 2026. Health authorities have declared a...

Increasing Precision In Inhalation Delivery
The discussion centers on how advancing formulation science can sharpen the precision of inhaled therapeutics, especially as companies target both lung and nasal routes. Speakers note that roughly half of the pipeline now consists of biologics, prompting a push to deliver...
The Low-Dose Peptide I Take
The video follows a 34‑year‑old general practitioner who injects 1.25 mg of tirzepatide weekly despite lacking diabetes or obesity, using the drug to tap into emerging evidence that GLP‑1 and GIP agonists confer health benefits beyond weight loss. Recent large‑scale trials—SELECT, FLOW,...

Shining a Light on Bladder Cancer Detection
MIT researchers have engineered a urinary catheter coated with carbon‑nanotube nanosensors that light up when they encounter bladder‑cancer‑specific protein biomarkers. The device scans the bladder with a laser‑based system, producing a fluorescent “chemical image” that pinpoints where malignant molecules are...

EMV Capital Advances XF-73 Toward Late-Stage Trials for Surgical Infection Prevention
EMV Capital announced that its AMR Bio subsidiary is moving XF‑73, a novel anti‑infective, into the final FDA‑design phase of a Phase 3 trial aimed at preventing surgical site infections. XF‑73 has already cleared a successful Phase 2b study, showing near‑100% efficacy in...

Former Top Gun Pilot on Hallucinogens, PTSD & the Abyss | Brian "Ponch" Rivera at Macrocosm 2026
The talk by former Top‑Gun pilot Brian “Ponch” Rivera blends military decision‑making, team dynamics, and the mental‑health crisis among veterans. He frames his experience in the Bay of Bengal as a case study of how world models—our internal maps of...

Is There an Effective Vaccine for the Ebola Outbreak? | Asked & Answered
An Ebola outbreak in the eastern Democratic Republic of Congo, identified as the Bundibugyo strain, has sparked urgent questions about vaccine availability. Health officials confirm no approved vaccine exists that reliably protects against this specific strain, and the existing Zaire‑targeted...

Could a Pill Prevent the World’s Deadliest Cancer?
The video explores a emerging strategy to prevent lung cancer by targeting inflammation rather than solely focusing on genetic mutations. Researchers at Mount Sinai, led by Dr. Miriam Merad and Dr. Tom Marron, argue that up to 80% of lung...

Dr. Glaucomflecken Explains: Intensive LDL Cholesterol Targeting in Atherosclerotic CVD (Ez-PAVE)
The video features Dr. Glaucomflecken breaking down a New England Journal of Medicine trial that compared intensive LDL‑cholesterol lowering (target <55 mg/dL) with conventional management (target <70 mg/dL) in patients with established atherosclerotic cardiovascular disease. Over 3,000 adults were randomized and followed for three years....