
Oncology Nurses - The Frontliners in Oncology
The FDA Oncology Center of Excellence hosted a panel for National Nurses Week, highlighting oncology nurses as the front‑line drivers of cancer drug development. Senior advisors and seasoned nurses shared personal stories, underscoring the profession’s high public trust and its pivotal role in translating scientific protocols into compassionate patient care. Panelists described a typical day: reviewing consent forms, monitoring adverse events, coordinating with investigators, and ensuring patients receive timely, evidence‑based interventions. They emphasized that nurses are often the first to notice subtle toxicities, manage side‑effects, and act as the conduit between complex trial designs and the lived experiences of patients and families. Memorable remarks included, “Nurses are the heartbeat of clinical research,” and the assertion that their education efforts—spending hours explaining protocols to patients—build the trust essential for trial enrollment and retention. Participants also highlighted the logistical challenges of coordinating multi‑site studies and the need for clear, patient‑centered communication. The discussion reinforces that oncology nurses are indispensable to both patient safety and the efficiency of drug approvals. Their advocacy, coordination, and education accelerate development pipelines while safeguarding outcomes, signaling a need for continued institutional support and investment in nursing resources.

CDER - REdI Annual Conference 2026: Innovative Regulatory Strategies to Advance Medical Products
The FDA’s REdI Annual Conference 2026 gathered over 8,300 registrants from 106 countries to showcase innovative regulatory strategies that will shape the future of medical product oversight. The event’s theme highlighted the agency’s shift from traditional paradigms toward data‑rich,...
Trump Administration Blocks Publication of Vaccine Research
The video reports that the Trump administration, through the Department of Health and Human Services, blocked the publication of several vaccine‑safety studies, a move described as breaking decades‑old precedent. The FDA allegedly refused to release the research because the authors...

Bloomberg Money Minute
The Bloomberg Money Minute highlighted three distinct market stories: Biontech’s operational cutbacks, Pfizer’s earnings beat, and soaring U.S. beef prices. Biontech announced the shutdown of several manufacturing sites, eliminating as many as 860 positions after COVID‑19 vaccine sales fell sharply...

$STVN: Are Oral GLP-1s Really a Death Blow? | Aurelian Research's Leo Trudel
The podcast examines Stevanato Group (STVN), an Italian glass‑vial and containment‑system maker serving the world’s largest pharma firms. Its business surged during the COVID‑19 vaccine boom, later flattened as customers destocked, and is now rebounding as demand for biologic injectables...

CDER & CBER All Hands: Implementing the FDA's Plausible Mechanism Framework
The FDA held its first joint CDER‑CBER all‑hands to unveil the Plausible Mechanism Framework, a regulatory approach designed to keep pace with rapid advances in individualized medicine. Speakers highlighted the landmark case of baby KJ, whose successful gene‑editing therapy...

NIH SciBites: A Smarter Way to Silence Inflammation
NIH postdoctoral researcher Matteo Pavan unveiled a novel therapeutic strategy aimed at chronic inflammation, a condition implicated in roughly 60% of worldwide deaths and a driver of heart disease, cancer, diabetes and Alzheimer’s. Current anti‑inflammatory drugs act like a sledgehammer, suppressing...

Introduction to Gene Therapy From Biotechnological Perspective (10 Minutes)
The video provides a biotech‑focused overview of gene therapy, tracing its evolution from early concepts in the 1970s to the current portfolio of FDA‑approved products. It explains how modifying a patient’s genetic code differs from conventional symptom‑based treatments and why...

From Partnered ASO Therapies To A Wholly-Owned Pipeline With Ionis's Brett Monia, Ph.D.
The Business of Biotech interview with Ionis founder‑scientist Brett Monia, now CEO, explores how the RNA‑therapeutics pioneer moved from a partnership‑heavy antisense oligonucleotide (ASO) model to a wholly‑owned drug pipeline with built‑in commercialization capabilities. Monia recounts the early scientific unknowns—cellular uptake,...
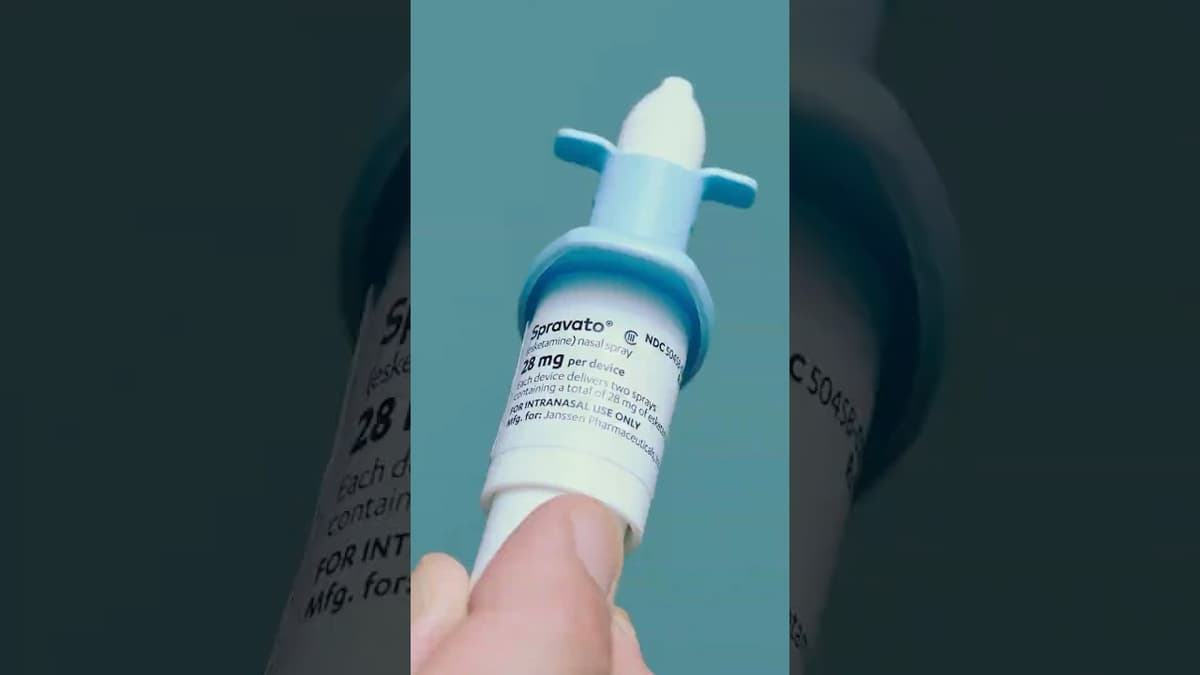
J&J’s New Drug for Treatment-Resistant Depression
Johnson & Johnson’s newly FDA‑approved nasal spray, Spado (esketamine), targets patients with treatment‑resistant depression (TRD) and acute suicidal ideation. The drug, delivered via a nasal atomizer, represents the latest addition to the limited arsenal of rapid‑acting antidepressants. Unlike traditional oral antidepressants,...

Cancer Clinical Trials: Basics, Timeline, Safety, Risks/Benefits & Myths (Part 1)
In this introductory session, Gabrielle Gargano and Cassel Mangalinden walk viewers through the fundamentals of clinical research, emphasizing its relevance across oncology and broader therapeutic areas. They outline the spectrum of trial categories—prevention, screening, diagnostic, treatment, and quality‑of‑life—highlighting that many...
Psychedelic-Assisted Therapy or Therapy-Assisted Psychedelics?
The conversation centers on a semantic and practical split: is the emerging model "psychedelic‑assisted therapy" or rather "therapy‑assisted psychedelics"? As ketamine clinics and new psychedelic startups proliferate, the terminology reflects deeper questions about the role of psychotherapy versus the...

(Review) Regulation and Review Process of OTC Drugs in Japan - PMDA-ATC Learning Viedos
The video outlines Japan’s regulatory framework for over‑the‑counter (OTC) medicines, emphasizing self‑medication as a policy response to an aging population and fiscal pressures on universal health coverage. Over the past five years, roughly 600‑700 OTC applications are filed annually, with a...

(Review) Orphan Drug Designation System in Japan - PMDA-ATC Learning Videos
The video explains Japan's orphan drug designation system, governed by the PMD Act, its enforcement regulations, and a specific notification. It outlines the three eligibility criteria—patient population, medical need, and development feasibility—that a drug must meet to qualify. A drug qualifies...

WHO Director-General Dr Tedros on the Pathogen Access and Benefit Sharing (PABS) Annex
World Health Organization Director‑General Dr. Tedros highlighted the Pathogen Access and Benefit Sharing (PABS) annex as the final piece needed to activate the WHO pandemic treaty. He explained that the annex consolidates lessons from COVID‑19 and links a suite of...

From Passenger to Driver: How Clonal Hematopoiesis Rewires Cancer Risk | MGR | 8 April 2026
The talk chronicles a physician‑scientist’s transition from treating acute myeloid leukemia patients to uncovering the genetic underpinnings of related myeloproliferative disorders. By leveraging early‑era genomic sequencing on patient‑derived blood and buccal samples, the speaker’s lab identified the JAK2 V617F gain‑of‑function...

IBT Portfolio Manager Says KalVista Deal Signals Biotech 2.0 Growth
International Biotechnology Trust’s portfolio manager Ailsa Craig explained that the KalVista Pharmaceuticals acquisition marks the fund’s fifth deal in 2026, underscoring a surge in biotech‑focused M&A. The transaction, which lifted KalVista’s share price from $5 to $27, delivered a 36%...

Bending the Cost Curve: Understanding the GLP-1 Era | Global Conference 2025
The panel at Global Conference 2025 tackled the emerging GLP‑1 era, focusing on how these highly effective obesity treatments are reshaping employer‑sponsored health benefits and overall cost structures. Speakers from AON, MIT Sloan, and Merrick Ventures examined the paradox of...
Drug Discovery From 10 Years to Days | Demis Hassabis, Google DeepMind
Demis Hassabis, CEO of DeepMind, outlined how artificial intelligence is poised to transform drug discovery through Isomorphic Labs, a new spin‑out focused on chemistry and biochemistry. Building on AlphaFold’s breakthrough in protein‑structure prediction, the venture seeks to close the loop...

Advances in Liquid Biopsy for Diagnosis, Surveillance, and Treatment Response | MGR | 1 April 2026
The talk reviewed recent advances in liquid‑biopsy technologies, focusing on how cell‑free DNA and RNA in plasma can serve as a non‑invasive window into tumor genomics, epigenomics, and transcriptomics. Ashis highlighted three clinical arenas—early cancer detection, treatment monitoring, and organ‑injury...

U.S. Food and Drug Administration Webinar on the RCT-DUPLICATE Initiative
The FDA’s RCT‑DUPLICATE webinar presented a systematic effort to assess when real‑world evidence (RWE) can stand in for randomized clinical trials (RCTs). The initiative benchmarks observational database studies against completed trials, then extends the methodology to predict outcomes of...

FDA Direct: The Power of Real-Time Clinical Trials
The FDA unveiled its inaugural real‑time clinical trial system, announced by chief AI officer Jeremy Walsh alongside Dr. Emma Meagher of the University of Pennsylvania and Dr. Jennifer Litton of MD Anderson. The initiative moves beyond theoretical panels, delivering an...

When a Pharma Giant Comes to Town: The Promise and Politics of Eli Lilly's LEAP District
The video examines the creation of the LEAP District outside Lebanon, Indiana, a place‑based economic‑development initiative built around Eli Lilly’s next‑generation drug‑manufacturing hub. The partnership pools billions of dollars from the pharma giant and local governments to transform farmland into a...

Advancing Real-Time Clinical Trials
The FDA announced a landmark initiative: the first ever real‑time clinical trial, where regulators can monitor safety signals and efficacy endpoints instantly via a cloud‑based dashboard. Commissioner Dr. Marty Makary framed the effort as a response to the growing "dead...

Proteins for Lead Detection - Mike Jewett
Mike Jewett explains how his lab engineers proteins—either entirely new or modified natural variants—to serve as lead‑detection sensors. Proteins are strings of 20 possible amino acids; their order dictates three‑dimensional structure and function. Because the relationship between sequence and a desired...
Dr. Glaucomflecken Explains: Tenecteplase for Acute Central Retinal Artery Occlusion (TenCRAOS)
The video features Dr. Glaucomflecken reviewing a recent New England Journal of Medicine trial that tested intravenous tenecteplase as an emergency treatment for acute central retinal artery occlusion (CRAO), a sudden, painless loss of vision often seen in older hypertensive...

Implementing A New CEO Strategy With NervGen's Adam Rogers, M.D. And Rich Macary
The interview with NerveGen’s CEO Adam Rogers and adviser Rich McCary centers on the company’s new growth strategy as it prepares for a pivotal Phase III trial of its peptide therapy NVG291 for spinal‑cord injury. Rogers, a former biotech founder who...
Automating Clinical Trial Data Collection for Better Research Outcomes
The video outlines how artificial intelligence and automation are reshaping clinical‑trial data collection, positioning a unified health‑life‑sciences platform as the catalyst for faster, more efficient research. Key insights include AI‑driven patient trial matching that scans electronic health records in real time,...
This Vaccine Could Stop the Next Pandemic | The Economist
The video explores the prospect of universal, broad‑spectrum vaccines that harness trained innate immunity to blunt future pandemics, highlighting recent research and expert commentary. It contrasts the fast‑acting innate system with the slower, highly specific adaptive response, noting that vaccines...
Arthritis Cure BREAKTHROUGH: Regrow Young Cartilage
The video highlights a Stanford breakthrough where inhibiting the enzyme 15‑PGDH triggers regeneration of articular cartilage, a condition affecting over 50 million Americans with osteoarthritis and lacking disease‑modifying treatments. In aged mice, twice‑weekly injections of a small‑molecule 15‑PGDH inhibitor thickened joint surfaces...

Increasing Access to Nonprescription Drugs
The Duke Margolis Institute for Health Policy and the U.S. Food and Drug Administration convened a stakeholder meeting to discuss expanding access to non‑prescription (OTC) medicines through a new drug‑application framework. FDA officials outlined three pathways—direct‑to‑OTC submissions, prescription‑to‑OTC switches, and applications...
The 340B Program: Why It Avoids Budget Scrutiny
The video examines the 340B drug pricing program, a federal initiative that requires pharmaceutical manufacturers to sell medicines to eligible hospitals at deep discounts. Originally limited to roughly 90 providers, the program now includes more than 2,600 hospitals, many of...

US to Reclassify Medical Marijuana
The United States is moving to reclassify federally licensed medical marijuana from Schedule I to Schedule III, a shift that would place it alongside substances such as ketamine and certain anabolic steroids. The Department of Health and Human Services recommended the...

The Future of Cell-Free Biotechnology
In this Stanford Engineering interview, Professor Mike Jewett explains cell‑free biotechnology—a platform that harvests the molecular machinery inside lysed cells and repurposes it as a stand‑alone protein‑production factory. By stripping away the living cell’s chassis, the approach sidesteps the evolutionary...

What Happens When You Stop a GLP-1: The Data From Three Trials
The video dissects recent evidence on how durable the weight‑loss and cardiovascular benefits of GLP‑1 receptor agonists are once the drugs are stopped. It centers on a British Medical Journal analysis and three pivotal trials—SURMOUNT‑4, STEP‑1 extension, and a semaglutide...

Research Highlights | ART-Free HIV Remission
The Lancet HIV study led by Johns Hopkins demonstrates that initiating antiretroviral therapy (ART) within 48 hours of birth can dramatically limit the formation of the latent HIV reservoir in perinatal infections, opening the possibility of ART‑free remission. In a multinational...

The Impossible Maths Behind Trump's Drug Price Reductions. #BBCNews
The video debunks President Donald Trump’s claim that his administration has slashed prescription‑drug prices by hundreds of percent. It explains that the administration’s spokespersons, including RFK Jr. and Trade Secretary Howard Lutnick, mistakenly applied the wrong formula for percentage change, inflating...

New Prior Authorization Proposals: Implications for Prescription Drug Access
The podcast discusses CMS’s latest proposal to extend prior‑authorization deadlines to prescription drugs, building on a 2024 rule that applied only to surgeries and services. The rule would set a one‑week response window for standard requests and three days for expedited...

Warren to RFK Jr: Drugs Less than $17 Cost Patients $200 on TrumpRx
The clip pits Warren against RFK Jr., illustrating how the Trump‑run pharmacy program, TrumpRx, inflates prices for common prescriptions. It shows Protonix, a heartburn drug, listed at $200 for a 30‑day supply on TrumpRx while the identical generic pantoprazole sells for $16...

Psychedelic Therapy's $100B Moment: Why Compass Pathways' COMP 360 Changes Investing
The video examines the rapid emergence of psychedelic‑based mental‑health treatments, focusing on Compass Pathways’ COMP360 psilocybin program and its recent 23% stock surge after a successful Phase 3 trial. Experts highlight that psychedelic therapy is an interventional model—typically one to...

90% of Statin Side Effects Happened on Placebo Too
Statins remain cornerstone lipid‑lowering therapy, but patient‑reported muscle complaints often exceed true pharmacologic toxicity. The video dissects why many side effects stem from expectation rather than the drug itself. Biochemical changes such as modest CoQ10 reduction occur in most users, yet...

Can We Predict Heart Attacks Years Before They Happen? | The Future of Cardio Genomics
The video introduces Target MI, a €4 million EU‑funded initiative led by Professor Stephanie Bassina Wittinger in Malta, that seeks to predict heart attacks years before they occur using a multi‑omics approach. By leveraging the island’s compact population, the team assembled a richly...
"Mushrooms" For Depression: New Science | NEJM Clinician
NEJM Clinician reports on a JAMA Psychiatry trial evaluating a single 25 mg dose of psilocybin for treatment‑resistant depression. The double‑blind study randomized 144 patients to psilocybin, a low 5 mg dose, or nicotinamide, aiming to mask allocation. At six weeks, 17 % of...

We Spent $724,637 Testing Rapamycin. What We Found Shocked Us.
The video details a five‑year, $724,637 crowdfunded clinical trial that tested whether weekly rapamycin, combined with home‑based cycling exercise, could improve muscle performance in adults aged 65‑85. Results were published in the Journal of Cexia Psychopenia and Muscle, and the...

FDA Grand Rounds: Clinical Omics Biomarker Discovery and Validation in Precision Medicine
The FDA Grand Rounds session featured Dr. Richard Beger discussing clinical omics biomarker discovery and validation for precision‑medicine applications. He outlined the breadth of systems‑biology omics—genomics, transcriptomics, proteomics, metabolomics, lipidomics—and described a structured workflow that stresses early sample‑type decisions, rigorous...

HVIVO Lands Landmark Phase III Trial
HVO announced a landmark contract to run the world’s first Phase III human challenge trial for Iliad’s whooping‑cough vaccine, positioning the company at the forefront of next‑generation vaccine development. The agreement, signed after a year‑long letter‑of‑intent, is HVO’s largest ever in both...
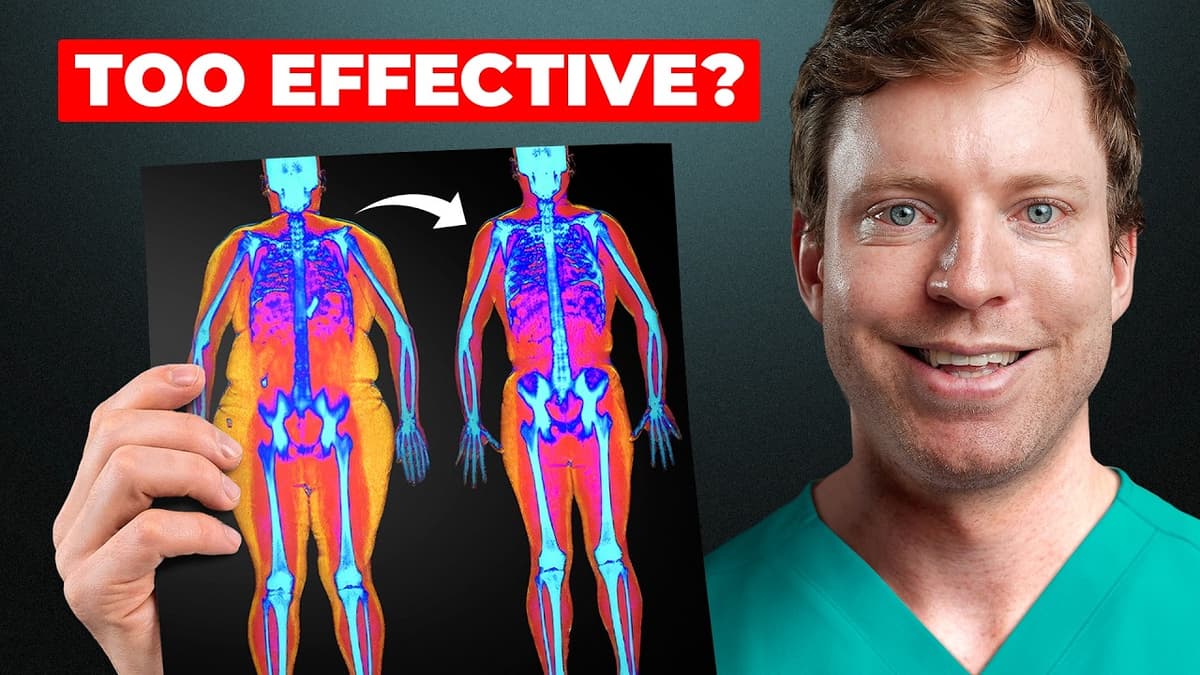
The 'Toxic' Hormone That Just Broke Every Obesity Record
The video examines retatrutide, Eli Lilly’s triple‑hormone receptor agonist that activates GLP‑1, GIP and glucagon. By turning on all three pathways, the drug has shattered obesity‑treatment records, delivering average weight losses of 24‑28% in phase‑3 trials—far exceeding the 15‑20% achieved by...

Johns Hopkins inHealth Precision Medicine | Driving Research and Clinical Innovation
The video introduces Johns Hopkins InHealth Precision Medicine platform, a data‑centric infrastructure that aggregates de‑identified electronic health records to power research and clinical care. It highlights tools like a unified dashboard that surfaces patient metrics, a large‑language‑model “Health General Reasoner” that...

GLP-1s Are a Gift From God
The video argues that glucagon‑like peptide‑1 (GLP‑1) agonists will become a more consequential technology than artificial intelligence, positioning them as a breakthrough medical tool. Beyond dramatic weight loss, clinical studies show GLP‑1s cut cardiovascular events, improve fatty‑liver disease, reduce sleep‑apnea severity,...

Standards' Role In Biosimilarity Assessment
The video examines the US Pharmacopeia’s recommendation that the FDA promote the use of reference standards in biosimilarity assessments. It questions whether such standards can resolve the inherent variability seen across commercial biologic products. The speaker acknowledges that standards are indispensable...