
Rare Disease Drug Commercialization With Zevra Therapeutics' Neil McFarlane
In a Business of Biotech interview, Zevra Therapeutics CEO Neil McFarlane outlined the company’s recent rebrand from KemPharm and its sharpened focus on developing and commercializing rare‑disease therapeutics. He traced his unconventional path—from Army Reserve medic to transplant nurse practitioner—to a mission‑first leadership style that now drives Zevra’s strategic roadmap. McFarlane highlighted several levers of growth: the $150 million sale of a pediatric priority‑review voucher in April 2025, a series of targeted acquisitions that bring complementary science and commercial expertise, and the assembly of a dedicated rare‑disease sales force. He emphasized partnership with patient‑advocacy groups to shape trial design and market access, and detailed how the company is building a commercial infrastructure capable of launching ultra‑rare products globally. The interview featured vivid analogies, such as comparing biotech execution to a military field hospital operation, underscoring the importance of clear operational orders and cultural alignment in mergers. McFarlane cited the Animan acquisition as a case where shared mission and agile scientific teams created a “force multiplier,” accelerating the development of less‑invasive transplant therapies. These moves position Zevra as a potential partner of choice for larger pharma seeking rare‑disease pipelines, while the voucher proceeds provide runway for late‑stage trials. The emphasis on culture, advocacy, and focused commercialization suggests Zevra could capture a sizable share of a market that is attracting heightened investor interest and regulatory incentives.
This Vaccine Is Quietly Doing Something to Your Heart
Recent research highlights that the shingles vaccine Shingrix, already approved for preventing herpes zoster, also appears to confer significant heart health benefits. Large‑scale meta‑analyses and real‑world studies report up to a 30% reduction in stroke and a comparable drop in...

GLP‑1 Gene Therapy: A One‑Time Fix for Obesity & Diabetes? | Longevity News Roundup — Week 21, 2026
The longevity roundup highlighted several breakthrough therapies, most notably Fractyl Health securing European approval for RJVA-001, a GLP‑1 gene therapy that programs pancreatic beta cells to secrete GLP‑1 after meals, potentially eliminating the need for chronic injections in obesity and...
How Platform Engineering Unlocks CRA Readiness
Speakers from CloudSmith framed the EU Cyber Resilience Act (CRA) as an urgent, practical priority for platform engineers, not just legal teams. The webinar explained that CRA mandates transparency about software composition—especially generating and providing SBOMs (software bill of materials)—and...

Magic Lifescience at NIH POCTRN | Shark Tank Pitch for Point-of-Care Diagnostics
Magic Lifescience, a Stanford spin‑out, delivered a Shark‑Tank‑style pitch at the NIH POCTRN showcase, positioning its platform as a next‑generation point‑of‑care (POC) diagnostic solution. The founders, CSO Elaine Ng and CFO Tianhao, explained how their original cancer‑focused research was re‑engineered...
The Next Era of Healthcare Is Personal
The Mackenzie podcast episode spotlights Madna’s vision of a personal, information‑driven healthcare era. CEO Stefan Benell explains how the company’s mRNA platform treats DNA as an information molecule, allowing rapid design of vaccines, cancer immunotherapies, and treatments for rare genetic...

Strategy& Insider Podcast - Episode 46 with Lara Gervaise and Edoardo Guidice
The Strategy& Insider podcast featured Lara Gervaise and Edoardo Guidice, co‑founders of Vuosis AI, a Swiss EPFL spin‑off that uses voice analysis to flag early signs of fatal diseases, burnout and cognitive decline. Vuosis AI’s platform extracts hundreds of acoustic features—tone,...

ReVAMPing the Pharmaceutical Supply Chain: Creating a Reliable Domestic Supply
The Revamp Consortium convened leaders from government, industry, and academia to address chronic drug shortages and the broader fragility of the U.S. pharmaceutical supply chain. Speakers highlighted the need to move from a reactive, fire‑drill mindset toward preventive strategies...

Grey Matters Launches US Brain PET Clinics for Alzheimer’s Diagnostics
Grey Matters Health announced the opening of its first U.S. brain PET imaging clinics, branded NovaScan, to provide amyloid plaque detection for Alzheimer’s diagnosis. The company highlighted a letter of intent with Catalyst MedTech for at least 200 scans at...

The Lizard that Helped Create Ozempic #science #ozempic #podcast
Researchers tracing how certain reptiles survive long fasting periods discovered hormones in the Gila monster that inspired GLP-1–based drugs such as Ozempic and Mounjaro. The conversation highlights how basic biological research on these lizards led to first-in-class therapies for weight...

Where Organ on Chips Will Add the Most Value in 3–5 Years
The discussion centers on how contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) can accelerate the adoption of organ‑on‑chip technologies over the next three to five years. Participants stress that scaling commercial systems and creating a unified...

Audience Poll: Validation and Trust Are Biggests Barriers to Organ on Chip Adoption
An audience poll found that 46% of respondents say validation and trust are the biggest barriers to broader organ-on-chip adoption, with regulatory acceptance cited by 25%, cost and scalability by about 19%, and workflow integration by 9%. Panelists said the...

The Reality of Human on a Chip Systems
Researchers and teams have demonstrated multi-organ “human-on-a-chip” systems—ranging from DARPA-funded 10-organ demos to focused gut–liver and gut–liver–brain models—that reveal meaningful organ interactions and immune trafficking. These platforms have produced actionable biological insights, such as short‑chain fatty acids exacerbating inflammation in...

Mumbai Pharma Strike: E-Pharma Companies, Chemists Slam Aggressive Pricing Strategies | WION
India’s chemists and druggists have launched a one‑day, nationwide bandh, shutting roughly 80% of private medical stores to protest the rise of e‑pharmacy platforms and their aggressive discounting tactics. The All India Organization of Chemists and Druggists (AIOCD) alleges that many...

The Vitals | Pioneering Ketamine Treatment for Depression
The Vitals episode brings together Mount Sinai psychiatrists to discuss ketamine’s emergence as a fast‑acting antidepressant and its expanding role in treating depression and PTSD. Dr. Dennis Charney recounts the mid‑1990s Yale experiments that showed a single sub‑anesthetic ketamine infusion lifted...

Using AI to Outsmart Drug-Resistant Bacteria
The video addresses the escalating crisis of antimicrobial resistance, describing it as a silent global pandemic that forces the medical community to rethink how antibiotics are discovered and deployed. Traditional drug development struggles to keep pace as bacteria rapidly evolve,...
The AI Super Scientist
The video introduces Alex Zhavoronkov, founder and CEO of In Silico Medicine, showcasing how the company leverages artificial‑intelligence‑driven drug discovery to tackle complex diseases. Using AI, the firm mined massive biomedical datasets to map idiopathic pulmonary fibrosis (IPF), identified key pathogenic...

Science Can't Wait: A Discovery Series | Part 3 | Featuring Cancer Researcher Daniel Hollern
The Science Can’t Wait webinar’s third installment spotlighted Salk Institute researcher Daniel Hollern’s work on leveraging the immune system—specifically B cells—to combat breast cancer. The session framed the effort as part of a broader interdisciplinary push, where basic questions...

The Role Of Regulators In Easing SUT Adoption
The panel examined how regulatory expectations shape the adoption of emerging manufacturing technologies in pharma. Participants highlighted that regulators frequently request extensive data sets—often beyond what a beta‑stage technology can realistically provide—creating a de‑facto barrier for innovators. Key insights included a...
One Pill Changed Everything
A clinical-trial medication from Telomere Pharmaceuticals given as a once-daily pill reportedly produced rapid, dramatic improvements in two rescue dogs: Zeus, a 12-year-old German Shepherd with terminal cancer, regained energy and appetite days after starting treatment, and Benson, a severely...
What if Aging Organs Could Actually Repair Themselves?
The video explores emerging regenerative therapies that pair stem‑cell delivery with epigenetic reprogramming and retinoic‑acid signaling to repair age‑related organ damage rather than replace organs outright. Researchers argue that activating retinoic‑acid pathways in kidney tissue, together with guided stem‑cell progenitors, could...

The Future of Fungi
The Stanford Engineering podcast explores the emerging frontier of fungi, hosted by Russ Altman and featuring bioengineer‑chef Vayu Hill‑Maini. Hill‑Maini argues that mushrooms and molds are poised to become a cornerstone of future food, pharmaceuticals, and novel materials, leveraging their...
Building Companies at the Edge of Science and Market - Life Sciences Today Podcast Episode 61
The Life Sciences Today podcast features Jennifer Ernst, a rare hybrid who has moved from high‑tech device work at Xerox PARC to bio‑electronic medicine. Her career is defined by matching breakthrough science with clear market opportunities, from printed‑electronics roll‑to‑roll manufacturing...
TheraCryf CEO on Why Latest Toxicology Results Represent a Major Milestone
The CEO of Theracryf announced that the company has reached a pivotal pre‑clinical milestone, completing a high‑dose toxicology study in rodents and preparing a parallel study in mini‑pigs. This marks the final set of safety assessments required before filing an...

The Women's Health Initiative Wasn't a Bad Study. The Headlines Were. | Dr. Heather Hirsch
In this interview, Dr. Heather Hirsch argues that the Women’s Health Initiative (WHI) was a rigorously designed, double‑blind, placebo‑controlled trial, and that the negative headlines that followed its 2002 release have unfairly tarnished menopausal hormone therapy (HRT). She emphasizes that...

Abortion Access In The High Court, Again | Katie Keith
The podcast focuses on a fresh legal battle over the FDA’s 2023 decision to allow mifepristone – the drug used for medication abortions – to be prescribed via telehealth and dispensed at pharmacies. Louisiana’s attorney general, joined by a private...

Carolyn Rodriguez, MD, PhD | Taming the Unquiet Mind: Next Frontiers in OCD Treatment and Research
In a Stanford‑hosted talk, associate dean Carolyn Rodriguez outlined the next frontiers in obsessive‑compulsive disorder research, emphasizing the need to shorten the 14‑ to 17‑year gap between symptom onset and evidence‑based care. Rodriguez highlighted three pillars of her lab’s work: a...

Sean Spencer, MD, PhD, Fellow ’20, Postdoc ’22 | Harnessing Gut Microbes to Heal Patients
Dr. Sean Spencer, a Stanford gastroenterologist and physician‑scientist, presented the emerging clinical frontier of gut‑microbe therapeutics. He outlined how advances in sequencing, culturing and sampling are moving the microbiome from a research curiosity to a practical diagnostic and therapeutic tool. Three...

Jean Tang, MD ’99, PhD ’03, Resident ’07 | Personalized Gene Therapy to Treat Rare Disease
Dr. Jean Tang, a Stanford dermatologist, detailed her two‑decade journey developing a personalized gene‑therapy for epidermolysis bullosa (EB), a rare disorder affecting one in 100,000 where patients lack functional collagen VII. Using a retroviral vector to deliver the 9 kb...

(Podcast Version) The Battle to Beat Malaria | NOVA Remix | NOVA | PBS
The podcast chronicles the decades‑long fight against malaria, focusing on the breakthrough R21 vaccine developed by Oxford researchers and manufactured at scale by the Serum Institute of India. It contrasts the legacy RTS,S/RTSS vaccine’s modest ~40% efficacy with the World...

Elutia Inc. (NASDAQ: ELUT) Reducing Surgical Infection Risk & Scaling Commercial Adoption
Elutia Inc. (NASDAQ: ELUT) is positioning its antibiotic‑infused biomaterial platform to slash the roughly 20% post‑operative infection rate that plagues breast reconstruction after mastectomy. The company highlighted its upcoming NXT‑41X product, which integrates a sustained‑release antibiotic payload into standard surgical...
A Daily Pill Could Help Keep Weight Off After Stopping Obesity Jabs. #WeightLoss #BBCNews
A new oral medication, Orthogon, is being positioned as a follow‑up therapy for patients who have stopped GLP‑1 weight‑loss injections such as semaglutide. The pill aims to preserve the weight loss achieved during the injectable phase. In a randomized, placebo‑controlled trial...
Paxlovid for Covid?
Two recent randomized trials in the U.K. and Canada enrolling about 4,000 mostly vaccinated outpatients — age 50+ or younger with comorbidities — found Paxlovid did not lower the already low combined rate of hospitalization or death (around 1%). However,...

Immunic CEO on Q1 Highlights, New Appointments and Upcoming Milestones
Immunic’s CEO Daniel Witt used the Proactive interview to outline a busy first quarter, highlighting a slate of senior appointments, a $400 million oversubscribed financing round, and the company’s roadmap toward commercializing its lead candidate, VETA calcium. The new chief...
Dr. Glaucomflecken Explains: Enfortumab Vedotin and Pembrolizumab in Bladder Cancer (KEYNOTE-905)
The video reviews the New England Journal of Medicine publication of the phase III KEYNOTE‑905 trial, which evaluated neoadjuvant Enfortumab Vedotin combined with Pembrolizumab in patients with muscle‑invasive bladder cancer who could not receive cisplatin‑based chemotherapy. The study randomized 340 eligible...
Why Delivery May Be the Biggest Problem in Longevity
The video discusses how delivery, not just molecular design, is the biggest obstacle to translating rejuvenation biotechnologies—such as Yamanaka factor gene therapies—into practical longevity treatments. It highlights that only a few tissues are naturally amenable to current delivery methods. The eye...
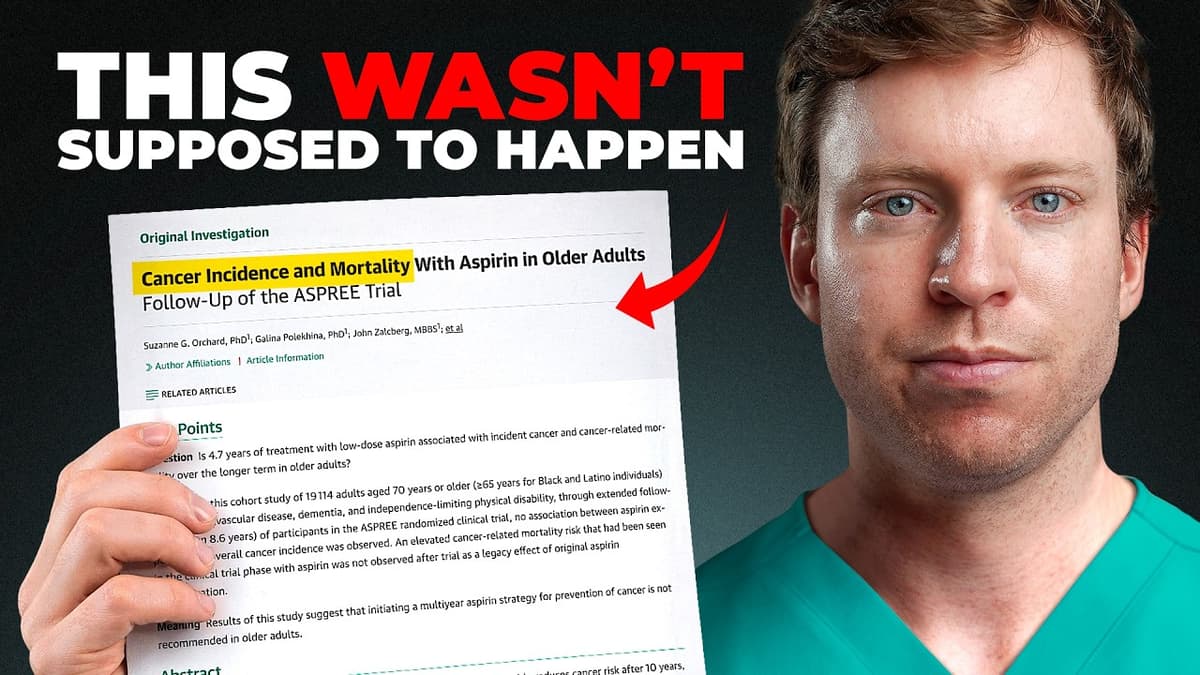
We Were Wrong About Aspirin (New Evidence)
The video examines how new randomized evidence overturns the long‑standing belief that daily low‑dose aspirin prevents cancer in otherwise healthy adults. Early observational studies and a 2010 meta‑analysis by Peter Rothwell suggested a one‑third reduction in cancer deaths, prompting the...

MedStory: The Hidden Gene Behind Sudden Cardiac Death
The video explains how a rare inherited disorder, arrhythmogenic right‑ventricular cardiomyopathy (ARVC), is driven by mutations in the plakophilin‑2 (PKP2) gene, a hidden cause of sudden cardiac death in young athletes. Stanford’s HEROIC PKP2 trial uses a non‑replicating adeno‑associated virus to...

Dr. Valter Longo on GLP-1, Growth Hormone Peptides and the Diet That Reverses Aging | EP#417
In this episode, longevity researcher Dr. Valter Longo critiques the hype surrounding GLP‑1 agonists and growth‑hormone‑releasing peptides, arguing that they are not the panacea for anti‑aging. He contrasts these pharmacologic shortcuts with dietary regimens that aim to reverse aging through...

The Real Future of Personalized Medicine
The video examines the economics of personalized medicine, contrasting the astronomical price tags of current one‑off gene‑therapy treatments with the potential for mass‑produced, low‑cost alternatives. Today, a single curative gene therapy can cost $1.7‑2.7 million, while manufacturing a short peptide costs roughly...

90% Nocebo (SAMSON) Trial
The Samson trial examined 60 patients who had stopped statins due to perceived side effects. Over a 12‑month blinded crossover, participants took a statin for four months, a placebo for four months, and no tablet for the final four months...

Building A New Antibody Discovery Platform With Infinimmune's Wyatt McDonnell, Ph.D.
In the latest episode of Business of Biotech, Life Science Leader’s Ben Comr sits down with Wyatt McDonnell, Ph.D., co‑founder and CEO of Infinimmune, to discuss the company’s novel antibody discovery platform that combines a vast repository of human blood...

AI, Cancer & Programmable Biology
The video explores how artificial intelligence and programmable biology are converging to create bespoke molecular tools that can deliver mRNA, CRISPR components, vaccines or immunotherapies directly to diseased cells. By leveraging single‑cell RNA sequencing, researchers can map the expression levels of...
AI Isn’t Just Changing Software. It’s Changing Medicine.
The video illustrates how artificial intelligence is reshaping medicine through a real‑world case study. GitLab’s founder, diagnosed with bone cancer and denied trial access, leveraged AI to dissect his own tumor’s molecular profile and then released the data publicly for...

Statin Intolerant? What to Do Instead (2026 Update)
The video tackles statin intolerance, outlining the 2026 clinical update on how to manage patients who cannot tolerate traditional statin‑fibrate therapy. It follows a case study of a man whose statin‑induced myositis prompted a shift to a health‑promoting diet, fish‑oil...

Why These Former FDA Officials Left the Agency
The STATus Report video examines why a wave of seasoned FDA officials departed during the second Trump administration, highlighting a confluence of mass layoffs, political meddling, and cultural shifts that pushed long‑time scientists out of the agency. Interviewees describe abrupt...

Rare Disease Families Find Roadmap to Drug Development at Bootcamps
The video spotlights a growing movement of rare‑disease bootcamps that equip non‑scientist parents with the tools to launch drug‑development projects for their children. Hosted by biotech firm Ultragenics, the multi‑day, sponsor‑funded curriculum walks participants from early research concepts through...

Safer Stem Cell Transplants — without Chemotherapy or Radiation | Stanford Medicine
Stanford Medicine researchers have introduced a novel conditioning regimen that replaces traditional chemotherapy and radiation with an antibody, Briquilimab, for bone‑marrow transplants in patients with Fanconi anemia—a disorder marked by defective DNA repair. The approach targets the CD117 receptor on...

HVIVO Secures £6m Influenza Human Challenge Trial Contract
hVIVO announced a £6 million contract to run a human challenge study testing a prophylactic antiviral against influenza. The agreement, detailed by Chief Scientific Officer Andrew Catchpole, marks a significant step for the company’s antiviral pipeline. The trial is divided into two...
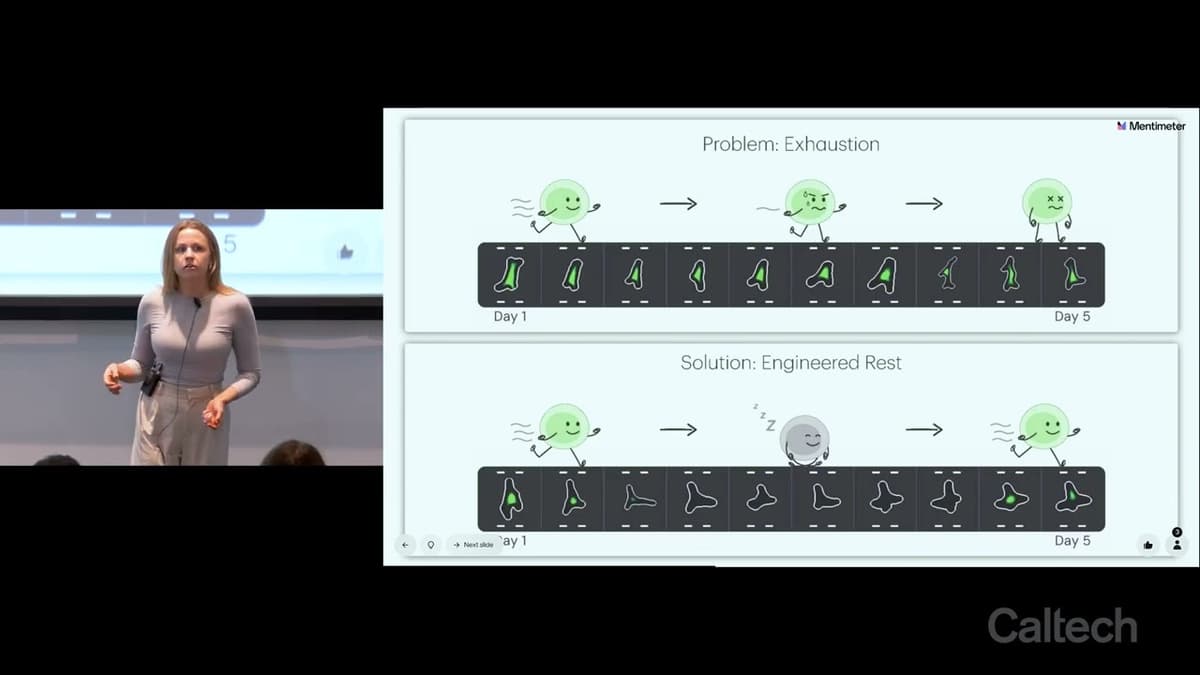
Victoria Tobin - Building Circuits to Give Cancer-Fighting Cells a Break
The video introduces Victoria Tobin’s work on engineering genetic circuits that give CAR T cells scheduled “breaks,” addressing the exhaustion that hampers their cancer‑killing performance. Tobin explains that CAR T cells, harvested from patients and reprogrammed to target tumors, work well...