Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Rockwell Automation and Cytiva Launch Platform to Accelerate Digital Transformation for Biopharma Companies
Rockwell Automation and Cytiva have launched Figurate, a supervisory control and data acquisition (SCADA) platform aimed at streamlining digital integration in biopharmaceutical manufacturing. The system combines Cytiva’s bioprocessing hardware with Rockwell’s FactoryTalk software, offering an open‑architecture layer that connects third‑party equipment. Designed for scalability, Figurate can grow from early‑stage process development to full‑scale commercial production without redesign. Pre‑engineered templates and modular components promise faster deployment, reduced validation time, and enhanced cGMP compliance.

AI Drug Target Platform Pairs Prediction with Benchmarking to Improve Early Discovery
Insilico Medicine unveiled an integrated AI framework that couples its Target Identification Pro (TargetPro) predictive engine with the TargetBench 1.0 benchmarking suite to improve early‑stage drug target discovery. The system uses disease‑specific models trained on 22 omics and text scores,...

Early Data Links Wegovy to Risk of 'Eye Stroke' — Here's What to Know
Early signals from a British Journal of Ophthalmology analysis suggest Wegovy, the semaglutide‑based weight‑loss injection, may be linked to ischemic optic neuropathy (ION), a rare form of eye stroke that can cause rapid vision loss. The study examined 31,774 FDA...

BREAKING: Senate Investigation Finds FDA Officials Covered-Up 25 COVID Shot Safety Signals
The episode examines a Senate investigation revealing that FDA officials allegedly suppressed about 25 statistically significant safety signals linked to COVID‑19 vaccine adverse events, including serious conditions such as Bell's palsy, cardiac failure, sudden cardiac death, and strokes. It details...

AbbVie Faces Questions About Skyrizi Competition From J&J
AbbVie’s first‑quarter earnings call highlighted growing pressure on its immunology franchise as analysts probed the company’s defense against Johnson & Johnson’s upcoming IL‑23 inhibitor. Skyrizi, AbbVie’s flagship psoriasis drug, posted $1.2 billion in Q1 sales, a 12% year‑over‑year increase, but faces...

Pharmaceutical Executive Daily: Rocket Pharmaceuticals Sells Pediatric Disease Priority Review Voucher
Rocket Pharmaceuticals agreed to sell its rare‑pediatric disease priority‑review voucher for $180 million in cash, providing non‑dilutive funding as the PRV program was reauthorized in early 2026. Teva Pharmaceutical announced a definitive agreement to acquire Emalex Biosciences for up to $900 million,...

Senate Report: Early COVID Vaccine Warning Signs Were Overlooked
A Senate Permanent Subcommittee report alleges the FDA ignored early COVID‑19 vaccine safety signals uncovered by an alternative data‑mining method in 2021. Analyst Dr. Ana Szarfman identified dozens of statistically significant adverse‑event signals, including cardiac death and Bell’s palsy, but...

AbbVie Tops Q1 Estimates, Raises Outlook and Discontinues Cancer Candidate
AbbVie reported $15 billion in first‑quarter revenue, a 12.4% year‑over‑year increase, driven by strong immunology and a 26% surge in neuroscience sales. Immunology products Skyrizi and Rinvoq generated $7.29 billion, while the migraine drug Qulipta boosted neuroscience revenue to $2.87 billion. The company...

The Pharma Sovereignty Playbook: How I’m Playing the $1.5 Trillion Drug Supply Crisis
A 2023 FDA inspection of Intas Pharmaceuticals’ plant in India halted half of the U.S. cisplatin supply, triggering nationwide shortages of both cisplatin and its substitute carboplatin. The crisis highlighted that only 24% of active pharmaceutical ingredient (API) manufacturing for...
Genome Editing Can Be Risky. Meet the Epigenome Editors
Scientists are turning to epigenome editors—tools that rewrite gene activity without altering the DNA sequence—to sidestep the safety concerns of traditional genome editing. By coupling dead Cas9 (dCas9) with epigenetic modifiers, researchers can turn genes on or off with high...

Mimio Health’s Fasting‑Mimetic Supplement Cuts Cholesterol and Glucose in RCT
Mimio Health’s fasting‑mimetic supplement Mimio lowered total cholesterol, LDL, oxidized LDL and fasting glucose in an eight‑week, double‑blind trial of 42 older adults. The study, published in Scientific Reports, also reported improved appetite regulation and reduced abdominal discomfort, suggesting a...

AbbVie Submits FDA NDA for Upadacitinib (RINVOQ) to Treat Severe Alopecia Areata
AbbVie announced it has submitted a new drug application to the U.S. FDA for upadacitinib (RINVOQ) in severe alopecia areata, based on Phase 3 results showing significant scalp hair regrowth. The filing targets both adult and adolescent patients, a population...

FDA to Pilot Real-Time Clinical Drug Trials Through Cloud and AI
The FDA announced a pilot that will stream clinical‑trial data to the agency in real time using cloud platforms and artificial‑intelligence analytics. Commissioner Marty Makary said the effort could shave up to 40% off the time between Phase 1 trials and...

FDA Launches Real‑Time Clinical Trial Tracking Pilot with AstraZeneca and Amgen
The FDA announced a pilot that will stream real‑time data from two pharmaceutical trials—AstraZeneca’s mantle‑cell lymphoma study and Amgen’s early‑stage lung cancer trial. The effort seeks to shrink the decades‑long lag between trial results and regulatory decisions, creating new IT...

MIT Team Unveils Magnetically‑controlled Soft Hydrogel Microrobots for Medical Use
Researchers from MIT, EPFL and the University of Cincinnati have 3D‑printed soft magnetic hydrogel structures that transform into microrobots controllable by external magnets. The “magno‑bots” can deform and grip at sub‑millimeter scales, opening pathways for biopsy retrieval and targeted drug...

Supply Chain Digital Twins: An Evolution, Not a Breakthrough
Researchers at NIST and EMD Millipore argue that digital twins can model the intricate biopharmaceutical supply chain, from demand shocks to distribution bottlenecks. By creating in‑silico replicas of cells, raw materials, and logistics flows, twins could identify alternative distribution centers and...

Milk Exosomes Transform Therapeutic Bioprocessing
Milk-derived extracellular vesicles, known as milk exosomes, are emerging as a biocompatible platform for therapeutic delivery. Researchers have loaded the JAK inhibitor tofacitinib into exosomes (mEXOs@TOF) for ulcerative colitis, achieving high drug‑loading efficiency, stability and strong anti‑inflammatory effects without toxicity....

Hopes Raised for More Sustainable Oligonucleotide Manufacturing
QurAlis CTO Hagen Cramer says enzymatic synthesis could make large‑scale oligonucleotide production far more sustainable than the solvent‑intensive solid‑phase method. While solid‑phase synthesis remains fast and automated, it generates high process mass intensity due to extensive solvent washes. Enzymatic, aqueous‑based...

Sandoz Q1 Net Sales Rise 3% to $2.76 Bn, Biosimilars Up 18% and 2026 Guidance Reaffirmed
Sandoz reported first‑quarter net sales of $2.76 billion, a 3% increase at constant currency, and confirmed its 2026 financial guidance. The growth was driven by an 18% jump in biosimilar sales, while generic sales slipped modestly.

AssureCare and Pillr Health Partner to Help Health Systems Tackle Specialty Pharmacy Costs
AssureCare and Pillr Health announced a strategic partnership to embed AssureCare's Akumen analytics platform into Pillr Health's Pharmacy Accelerator solution. The integration gives hospitals real‑time data and AI‑driven predictive modeling to manage specialty pharmacy costs, which are expected to exceed...

Late‑Stage Drug Development Trends: Phase III, AI, Global Partnerships
LOOKING forward to tomorrow's webinar on late-stage drug development trends with Susan Galbraith (AstraZeneca), Levi Garraway (Roche) and Ken Getz (Tufts). We'll be discussing the fundamentals of Phase III, with thoughts on efficiencies, interplay with biotech partners, China, AI, and...

Astellas’s R&D Head Talks Strategy Ahead of Looming Patent Cliff
Astellas Pharma faces a looming patent cliff as its blockbuster prostate‑cancer drug Xtandi, which generated about $4.6 billion in the 2025 fiscal year, loses U.S. patent protection next year. To offset the expected revenue hit, chief R&D officer Tadaaki Taniguchi is accelerating...
Henlius and Organon Gain EC Approval for Pertuzumab Biosimilar
Henlius and Organon have received the first European marketing authorization for a pertuzumab biosimilar, Poherdy, targeting HER2‑positive breast cancer. The EC approval mirrors the product’s earlier U.S. clearance and covers all indications of Roche’s reference drug, Perjeta, including metastatic, neoadjuvant...

Pfizer Earns Positive Phase 3 in Multiple Myeloma; ICON Overstated Revenue
Pfizer announced that its antibody‑drug conjugate Elrexfio achieved a statistically significant and clinically meaningful improvement in progression‑free survival for patients with double‑class exposed relapsed or refractory multiple myeloma. The Phase 3 trial met its primary endpoint, positioning the drug as a...

Quintuple Receptor Agonist Outperforms Tirzepatide in Trials
New @Nature A quintuple [GLP-1 + 4 other] receptor agonist drug that exceeds effects of the dual receptor (GLP-1 and GIP, tirzepatide) in the experimental model vs diabetes and obesity (in case you thought a dual receptor was max effect, as...

After Apellis Deal, Biogen Looks to Fill Early-Stage Pipeline
Biogen announced a $5.6 billion acquisition of Apellis Pharmaceuticals, adding a complement‑inhibition platform to its portfolio. While the deal closes this month, the company emphasized that its growth engine will now hinge on early‑stage development and research rather than late‑stage launches....

Brenntag Pharma Unveiled BYPHAR, the New Regulated Biopharma Manufacturing Brand
Brenntag Pharma introduced BYPHAR, a new regulated biopharma manufacturing brand that consolidates high‑quality raw materials, advanced facilities, and value‑added services into a single, compliance‑ready portfolio. The brand groups materials into three tiers—Explore, Enhance, and Excel—covering non‑GMP to LBLE critical grades,...

With Growth Curve Trending up, Biogen Looks to Early-Stage Assets for ‘Next Generation’
Biogen, fresh from its $5.6 billion acquisition of Apellis, announced on its Q1 earnings call that it will pivot toward early‑stage assets to thicken a pipeline it described as “quite thin.” The shift comes as Alzheimer’s patients transition off Eli Lilly’s Kisunla,...

March 2026 Patent Highlights
The March 2026 Patent Highlights page aggregates the latest drug‑discovery milestones, from 38 first‑time small‑molecule approvals by Europe’s EMA, China’s NMPA and Japan’s PMDA to a deep dive on protein‑structure advances and machine‑learning tools. It spotlights a newly optimized HPK1 inhibitor...
Circio Partners with TraffikGene Project to Advance Non-Viral circVec Delivery
Circio, an Oslo‑based circular RNA company, has partnered with the Universidad de Santiago de Compostela’s TraffikGene project to explore non‑viral delivery of its circVec circular RNA expression vectors. The collaboration merges Circio’s circVec platform with TraffikGene’s peptide amphiphile carrier system...
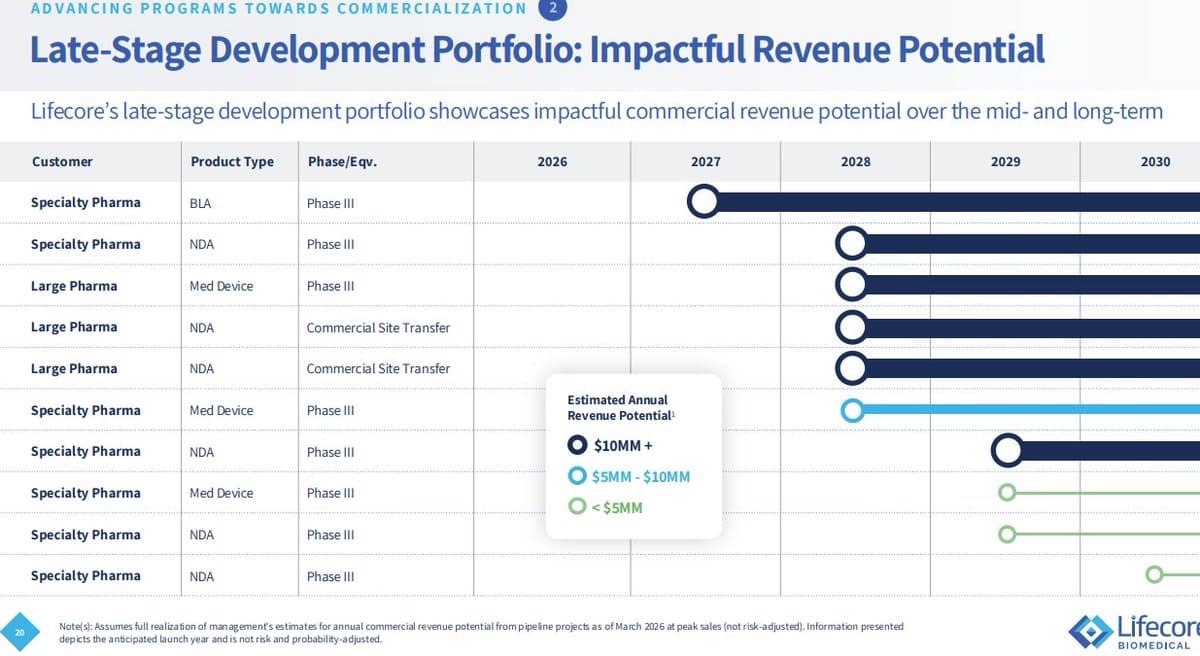
LFCR Undervalued; Upcoming Site Transfers Could Drive 25% Margins
$LFCR beaten down again on fears of equity issuance, lingering preferred concerns, and general liquidity worries. They have enough liquidity to cover the preferreds. Debt is not a concern as of yet. Meanwhile, the company has reported 5 site transfers in the...
SYRE Jumps to $8B After Breakthrough UC Trial
$SYRE pushing $8 billion cap currently on a major $XBI red day following their open label a4b7 data proving that a4b7 continues to work. Valuation was ~$3B at the start of the year. Must’ve been a massive surprise that their...

FDA Knew of Better Method to Detect COVID Vaccine Safety Signals — But Refused to Use It
Senator Ron Johnson released a report showing that FDA officials during the Biden administration knew a superior empirical Bayesian data‑mining method existed for detecting COVID‑19 vaccine safety signals in VAERS but ordered staff to continue using the older, flawed tool....
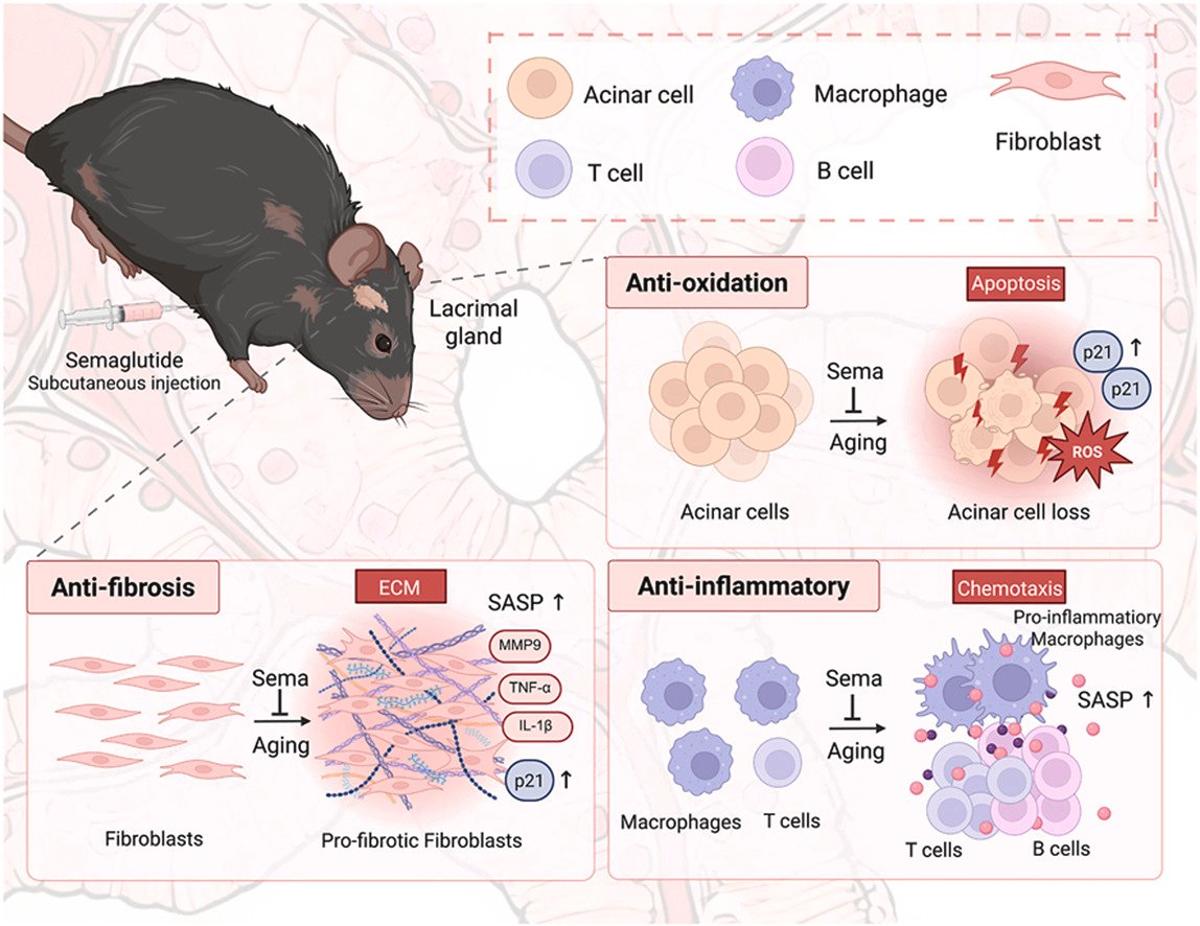
Semaglutide Restores Lacrimal Gland, Relieves Age‑Related Dry Eye
Semaglutide alleviates age-related dry eye disease by restoring lacrimal gland structure and function https://t.co/EDvPtdeiqg https://t.co/Kpku8SHEOH

Media Overlooks Upcoming RMP Level Concerns
How is no one in the press asking about level of RMPs going forward? @dampedspring @barryknapp
Nanozymes Against Brain Tumors
Researchers at Empa and HOCH Health Ostschweiz are developing biocompatible nanozymes that can be applied directly during brain‑tumor surgery to attack astrocytoma cells. The nanozymes act like enzymes, generating reactive‑oxygen species and activating drug precursors, and they are triggered by...

Teva Spends $700M on Tourette Drug, Shares Jump 11%
Teva nabs experimental Tourette drug in $700M Emalex buyout https://t.co/z1byYRgYul by @realJacobBell $TEVA + 11%

FDA Pilots Real-Time Trials as AbbVie Eyes KRAS Biotech
FDA tests out ‘real-time’ clinical trials; AbbVie closes in on a KRAS biotech https://t.co/diuieVTJ3g $ABBV $LLY $GSK $IONS $PFE #biotech
Biogen Ready to Catch Alzheimer’s Patients Transitioning Off Lilly’s Kisunla
Biogen is positioning its Leqembi therapy to capture Alzheimer’s patients who will finish Eli Lilly’s 18‑month Kisunla regimen and need a maintenance option. Leqembi, approved in January 2023, saw a 74% year‑over‑year sales jump to $168 million in Q1 2026, beating expectations. Biogen is...

Chiesi Acquires KalVista for $1.9B Rare Disease Drug
Chiesi to buy KalVista in $1.9B deal for rare disease drug https://t.co/VR9hDYGLGJ by Kristin Jensen $KALV $PHVS $NTLA $BCRX $IONS

FDA to Relax Peptide Rules After High‑profile Endorsement
FDA moves toward easing restrictions on certain peptides The agency’s decision to hold an advisory committee meeting on the topic comes after HHS Secretary Robert F. Kennedy Jr. told podcaster Joe Rogan he’s a “big fan” of peptides. https://t.co/wynhLuBUPE https://t.co/y9dgG0xWAq

AstraZeneca’s £300m UK Return Shows the Price Britain Paid to Win Back Pharma Money
AstraZeneca is restarting a £300 million (≈$380 million) research investment in the UK after the UK‑US pharmaceutical agreement boosted expected returns. The plan funds the completion of the Rosalind Franklin building in Cambridge and a new digital drug‑development lab, reflecting higher NHS...

Tomorrow's Dual AZN ODAC: Serena-6 AM, Truqap-281
Reminder that the dual $AZN ODAC takes place tomorrow. Camizestrant Serena-6 trial being debated in the morning, then Truqap Capitello-281 in the afternoon. Youtube link -> https://t.co/RDcvhRqVWR

Inside Servier’s New €200M Venture Fund
Servier has unveiled a new corporate venture fund, committing roughly $216 million to back early‑stage biotech focused on oncology and neurology. The fund, Servier Ventures, will target late pre‑clinical and Series A assets where it can add capital and industrial expertise, especially...

Candel Therapeutics Announces Commercial Agreement with EVERSANA to Support U.S. Launch of Prostate Cancer Therapy
Candel Therapeutics announced a commercialization agreement with EVERSANA to support the potential U.S. launch of its gene‑therapy candidate aglatimagene besadenovec (CAN‑2409) for intermediate‑ to high‑risk localized prostate cancer. EVERSANA will deliver a full suite of services, including data analytics, medical...

Chiesi Signs $1.9B Deal to Acquire KalVista and Its Approved Drug
Italian pharmaceutical group Chiesi announced a $1.9 billion cash acquisition of U.S. biotech KalVista Pharmaceuticals, paying $27 per share. KalVista brings an FDA‑approved therapy for a rare disease into Chiesi’s portfolio, bolstering its specialty drug offerings. The deal is part of...

SRN‑901 Extends Mouse Lifespan by 33% in Preclinical Trial, Raising Longevity Hopes
SinoGen, Tsinghua University and China National Pharmaceutical announced that the oral anti‑aging candidate SRN‑901 extended median remaining lifespan by 33% in naturally aging mice. The study also cut tumor incidence by 30% and slowed visible aging signs, positioning the drug...

Paradigm Health Teams with FDA and Pharma Giants to Speed Trial Data Review
Paradigm Health announced a partnership with the U.S. Food and Drug Administration, Amgen and AstraZeneca to pilot an integrated technology platform that delivers real‑time trial data to regulators, promising to shrink review cycles from months to days. The model, already...

Chiesi Expands Rare Disease Portfolio, Acquires KalVista for $1.9 Billion
Chiesi Group announced a $1.9 billion cash acquisition of KalVista Pharmaceuticals, its largest deal to date. The purchase adds EKTERLY (sebetralstat), the first oral, on‑demand therapy for hereditary angioedema (HAE), to Chiesi’s rare‑disease pipeline. EKTERLY is already approved in major markets...

STAT+: Pharmalittle: We’re Reading About the FDA Speeding up Trials, a Supreme Court Hearing on ‘Skinny Labels,’ and More
The FDA announced a pilot program that will review real‑time data from AstraZeneca and Amgen cancer trials using a platform built by Paradigm Health, aiming to speed regulatory feedback. The agency also opened a public comment period on a separate...