Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Qubit Pharmaceuticals Aims for Quadratic Speedup in Simulations
Qubit Pharmaceuticals and Singapore’s Centre for Quantum Technologies have deployed the first quantum Markov Chain Monte Carlo (qMCMC) algorithm on gate‑based quantum hardware. The two‑year partnership blends Qubit’s quantum chemistry know‑how with CQT’s expertise in circuit design, using Quantinuum’s H2 and Helios machines via the National Quantum Computing Hub. Early results, posted on arXiv, aim to demonstrate quadratic or exponential speedups over classical methods for molecular simulations. The teams plan to integrate these algorithms into future drug‑discovery pipelines.

Givaudan Research: Zensera Lemon Balm Supports the Mind During Stress
Givaudan’s patented Zensera lemon‑balm extract (300 mg) was tested in a double‑blind, placebo‑controlled trial with 130 healthy adults under moderate stress. The study measured mood, heart rate, blood pressure and a battery of executive‑function tasks over five hours. Participants who took...
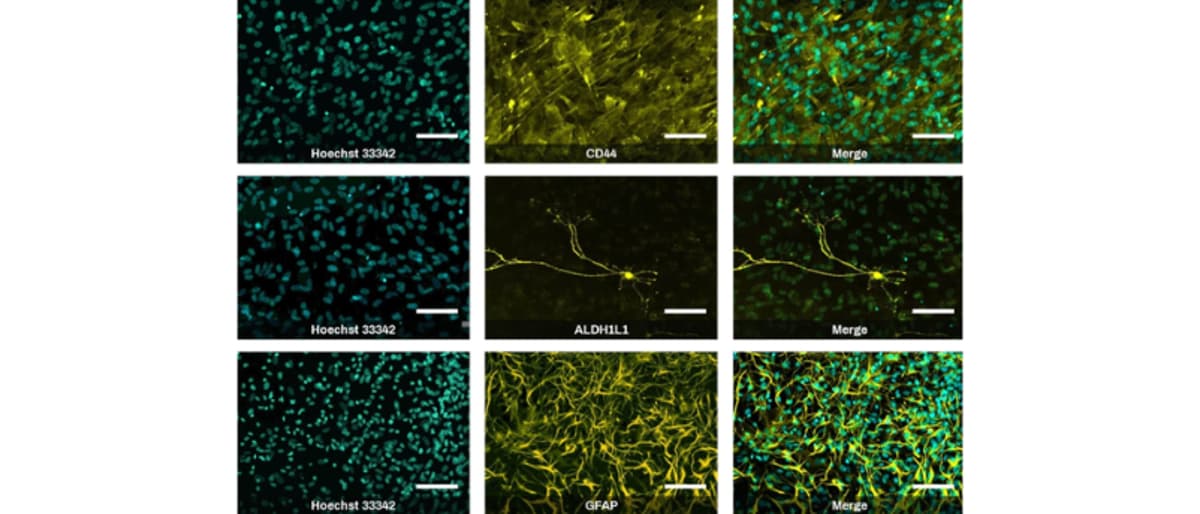
New iPSC Differentiation Kits for Neuroscience Research
AMSBIO introduced the Quick‑Glia™ product line, iPSC‑derived glial cell kits designed for neuroscience research. The kits convert human induced pluripotent stem cells into functional astrocytes or microglia in 1–2 weeks, delivering high‑purity, cryopreserved cells ready for disease modeling and drug...

Buried in Soil, a 100-Million-Year-Old Bacterial Toxin Could Reshape Pest Control and Antibiotic Discovery
Researchers from McMaster, Harvard, Yale and European partners have identified a new class of insect‑killing proteins, SAIPs, produced by rare Streptomyces strains. These toxins, structurally distant from diphtheria toxin, target an insect‑specific surface protein called Flower, leaving humans unharmed. The...

Precision Begins Patient Enrolment for FUNCTION-DMD Trial
Precision BioSciences has activated Arkansas Children’s Hospital as the inaugural site for its Phase I/II FUNCTION‑DMD trial, enrolling boys aged two to seven with Duchenne muscular dystrophy. The study tests PBGENE‑DMD, a first‑in‑class in‑vivo gene‑editing therapy that removes exons 45‑55 to restore...

How Epic Bio Is Leveraging CRISPR without Cutting DNA
Epic Bio, founded by Stanford professor Stanley Qi, is developing an epigenetic editing platform called GEMS that uses the smallest known Cas protein to modulate gene expression without cutting DNA. The system can be delivered in a single viral vector...

Re: Efficacy and Safety of VPM1002 and Immuvac in Preventing Tuberculosis: Phase 3 Randomised Clinical Trial (PreVenTB Trial)
The phase‑3 PreVenTB trial found that neither VPM1002 nor Immuvac reduced microbiologically confirmed tuberculosis, missing its primary efficacy endpoint. The authors of a BMJ rapid response highlight that the headline claim of 50% efficacy against extrapulmonary TB rests on only...

Henlius and Organon Receive the EC Approval for Poherdy (Biosimilar, Perjeta)
The European Commission has granted approval for Poherdy, a 420 mg/14 mL interchangeable biosimilar of pertuzumab (Perjeta), covering all approved indications. The decision rests on extensive analytical, pharmacokinetic, efficacy, safety and immunogenicity data that demonstrate equivalence to the reference product. Henlius has...
A Study in Regulatory Conservatism versus Clinical Innovation
At today’s ODAC meeting, the FDA will review AstraZeneca’s oral selective estrogen receptor degrader camizestrant for hormone‑receptor‑positive, HER2‑negative breast cancer with ESR1 mutations. Late‑stage data showed a 56% reduction in risk of disease progression and consistent safety, along with improved...

Defect-Engineered Pt/Nb2O5 Boosts Radical-Driven Benzimidazole Production and Hydrogen Evolution Efficiency
Researchers have created a defect‑engineered Pt/Nb₂O₅ catalyst with abundant oxygen vacancies and platinum nanoparticles that dramatically improves photocatalytic benzimidazole synthesis and concurrent hydrogen evolution. The system delivers 4.0 mmol g⁻¹ h⁻¹ production of 2‑methylbenzimidazole and 10.2 mmol g⁻¹ h⁻¹ hydrogen under mild light, surpassing prior benchmarks....

Follow the Money: Autoimmune, Inflammatory Disease Therapies, Antibody-Drug Conjugates, AKT1-Selective Inhibitor Programs
A wave of sizable financing rounds swept the biotech sector in late April 2026, with more than $1.3 billion pledged across 15 companies. Beeline Medicines led the pack with a $300 million Series A to advance lupus‑focused small‑molecule and protein therapeutics, while Sidewinder...
Single-Cell Sequencing Reveals Why some CAR-T Therapies Succeed While Others Fail
Researchers reviewed 44 single‑cell RNA sequencing studies covering about 500 patients to pinpoint cellular traits linked to CAR‑T therapy outcomes. The analysis identified exhaustion marker expression, low memory‑like cell fractions, and limited clonal diversity as hallmarks of relapse, while persistent,...

Cardiovascular Health 2026
Recent studies highlight pitavastatin’s pleiotropic benefits beyond LDL‑C reduction, including functional HDL elevation, enhanced cholesterol‑efflux capacity, and antioxidative actions in dyslipidemic patients. Pre‑clinical work shows the drug strengthens blood‑brain barrier integrity and mitigates lipopolysaccharide‑induced BBB dysfunction, suggesting neuroprotective potential. Real‑world...

Tackling Drug Resistance Must Become Biotech’s Next Frontier
Drug resistance underlies roughly 90% of the 600,000 cancer deaths in the United States each year, limiting the durability of modern therapies. Kairos Pharma, founded in 2013, is focusing on the biology of resistance with its candidate ENV‑105, which aims...

FDA-Approved Marijuana Products and Those Subject to State-Issued Licenses Rescheduled; Marijuana Hearing Calendared; Dispensary Applications Accepted
Acting Attorney General Todd Blanche and the DEA issued a final rule that instantly reschedules FDA‑approved THC drug products and marijuana sold under state medical licenses from Schedule I to Schedule III of the Controlled Substances Act. The rule aligns federal regulation...

Your Weekly TechBio News: High-Throughput Screening
High‑Throughput Screening (HTS) remains a cornerstone of modern drug discovery, allowing researchers to evaluate millions of chemical compounds against biological targets in a single campaign. Recent advances in robotics, miniaturized assay formats, and cloud‑based data pipelines have dramatically increased throughput...

Generally Good Indian Pharma Companies
An audit of top‑tier Indian generic manufacturers evaluated options for 20 therapeutic compounds, ranking firms by market capitalization, API vertical integration, ANDA volume, and GMP compliance. Cipla, Abbott, Sun Pharma, Zydus, Biocon and others were highlighted for specific products such...

How Multi-Omics Is Changing What Scientists Can See in the Human Immune System
Multi-omics technologies are reshaping human systems immunology by delivering high‑dimensional, single‑cell and spatial data that capture the full complexity of immune responses. Researchers now integrate scRNA‑seq, scATAC‑seq, CITE‑seq and spatial transcriptomics with large public atlases to identify molecular signatures predictive...

Morning Brief Podcast: Sun–Organon: The Scope, Risks, and Future of India's Biggest Pharma Deal
Sun Pharma announced an all‑cash $11.75 billion acquisition of U.S.‑based Organon, the largest overseas purchase by an Indian firm since 2007. The deal doubles Sun Pharma’s revenue to about $12.4 billion, propelling it into the top‑25 global pharmaceutical companies. It instantly gives...

New Copper Nanozyme Shows Powerful Tumor Suppression with High Precision
Researchers at the Chinese Academy of Sciences have created a coordinatively unsaturated copper single‑atom nanozyme (Cu‑N₂‑CDs) that exhibits markedly higher catalytic activity than traditional Cu‑N₄ nanozymes. The unsaturated Cu‑N₂ sites boost H₂O₂ adsorption by 3.49 times and generate hydroxyl radicals 3.62 times...

ONAVIDA Study Finds High‑Protein Oral Supplement Improves Recovery in Malnourished Cancer Patients
The ONAVIDA trial, published on 29 April 2026, demonstrated that a novel concentrated high‑protein, high‑calorie oral nutritional supplement (cHPHC‑ONS) significantly enhanced nutritional and morphofunctional recovery in 230 malnourished cancer patients over three months. The prospective, multicenter study combined the supplement...

Survodutide Shows 16.6% Weight Loss in Phase 3 Trial, Boosting Obesity Drug Race
Zealand Pharma reported that its dual GLP‑1/glucagon agonist survodutide produced an average 16.6% body‑weight reduction in a Phase 3 obesity trial. The data, presented from the SYNCHRONIZE‑1 study, also showed 85.1% of patients lost at least 5% of weight versus 38.8%...

NeoGenomics Q1 2026 Revenue Jumps 11% as COO Warren Stone Charts Aggressive Sales Expansion
NeoGenomics reported first‑quarter 2026 revenue of $186.7 million, an 11% year‑over‑year increase that beat guidance and prompted a lift in full‑year outlook. President and COO Warren Stone used the call to unveil a sales‑force expansion and deeper community‑oncology coverage aimed at...

FDA Launches Real‑Time Clinical Trial Monitoring to Accelerate Drug Reviews
The U.S. Food and Drug Administration announced the completion of its first real‑time clinical trial monitoring tests, partnering with AstraZeneca and Amgen to stream safety and efficacy data live to regulators. The initiative uses AI and electronic health‑record integration to...

Dr. Reddy’s Announces Health Canada Approval of Generic Semaglutide Injection
Dr. Reddy’s Laboratories received a Notice of Compliance from Health Canada, becoming the first firm to secure market authorization for a generic semaglutide injection in Canada. The approval covers both 2 mg and 4 mg pen formulations, and the company is preparing...

GSK‑Ionis Antisense Drug Bepirovirsen Gets FDA Priority Review, Breakthrough Designation
GSK and its partner Ionis Pharmaceuticals announced that the FDA has placed bepirovirsen, an antisense oligonucleotide for chronic hepatitis B, on the priority‑review track and granted it breakthrough therapy designation. The move follows Phase 3 B‑Well data showing statistically significant functional‑cure...

Gene Editing Swaps Whole Genes, Fixes 1,000 Mutations
A new genome editing technique enables efficient replacement of entire genes, allowing correction of up to 1,000 mutations simultaneously by inserting large DNA segments without causing toxic double-strand breaks. genetherapy
New Genome Editing Method Could Swap Entire Genes and Correct 1000 Mutations at Once
Scientists have unveiled a new genome‑editing platform called prime assembly that can insert DNA segments up to 11,000 base pairs, enabling the replacement of entire genes rather than single‑point edits. The method uses overlapping flaps to attach donor DNA without...

Cartesian Therapeutics Inc (RNAC) Q1 2026 Earnings Call Transcript
Wave Life Sciences reported a Q1 net loss of $46.9 million, with cash reserves of $243.1 million expected to fund operations into 2027. The DMD candidate WVE‑N531 delivered a statistically significant 3.8‑second improvement in time‑to‑rise and 7.8% average dystrophin expression,...

Syndax Pharmaceuticals Inc (SNDX) Q1 2026 Earnings Call Transcript
Syndax Pharmaceuticals reported $172 million total revenue for 2025, driven by $124.8 million from its menin inhibitor RevuForge and $151.6 million from the chronic‑GVHD therapy Nictimvo. RevuForge posted 38% quarter‑over‑quarter revenue growth and reached roughly 50% penetration of the KMT2A AML population, while...

Alnylam Pharmaceuticals Inc (ALNY) Q1 2026 Earnings Call Transcript
Alnylam reported a strong Q1 2021 performance, with total product sales reaching $135.8 million, an 89% year‑over‑year increase. ONPATTRO generated $102 million in revenue, growing 13% quarter‑over‑quarter, while newly launched OXLUMO contributed $9 million and GIVLAARI $24.7 million. The company announced positive Phase 3 data...

Amgen Inc (AMGN) Q1 2026 Earnings Call Transcript
Amgen reported first‑quarter 2026 sales of $5.6 billion, a 4% year‑over‑year increase, driven by six high‑growth products that contributed 70% of total revenue. Repatha, Evenity, Test Buyer, TEPEZZA and the rare‑disease portfolio posted double‑digit growth, while legacy brands Prolia and XGEVA...
Kymera Therapeutics Inc (KYMR) Q1 2026 Earnings Call Transcript
Kymera Therapeutics reported Q1 2026 revenue of $2.8 million, entirely from its Gilead collaboration, and ended the quarter with $978.7 million in cash, extending its runway into the second half of 2028. The company completed enrollment and dosing for the Phase Ib atopic...
Angiocrine Signaling Drives Liver Fibrosis: From Mechanism to Early Clinical Translation
Researchers led by Hu et al. discovered that ROCK2 activity in liver sinusoidal endothelial cells is a pivotal driver of liver fibrosis. Single‑cell transcriptomics and knockout models revealed that ROCK2‑mediated cytoskeletal remodeling releases angiocrine factors that activate hepatic stellate cells. Early...
Long-Lived Immune Cells Show Promise Against Cancer in World-First Trial
A world‑first clinical trial tested CAR‑T therapy enriched with stem‑cell memory T cells, a long‑lived immune subset. In a small cohort of 11 patients with refractory blood cancers, five achieved complete remission and one partial remission, outperforming historical outcomes of...

343. Summary: Can This Nutrient Help Alzheimer's? - Life Extension
In this episode, Dr. Mike and Dr. Crystal discuss a recent pilot study on creatine supplementation as a potential therapy for Alzheimer's disease, featuring insights from lead author Aaron Smith. They explain how creatine, known for its role in muscle...
Trial of Non-Invasive Endometriosis Scan Boosts Hopes for Quicker Diagnosis
A small trial of 19 women showed that the experimental radiotracer maraciclatide can illuminate endometriotic lesions on a Spect‑CT scan, matching surgical findings in 16 cases with no false positives. Current diagnosis in the UK often requires invasive laparoscopy, leading...

Tiny Biotech’s Experience Raises Questions About FDA’s Rare Disease Policies
BioCentury’s website now publishes a detailed cookie policy that separates cookies into five categories: strictly necessary, functional, marketing, advertising, and analytics. Strictly necessary cookies are always active and essential for authentication and navigation, while functional cookies enable personalization of the...

FDA Proposes AI-Driven Real-Time Trials, Ending Phase Gaps
No more Phase 1, 2, or 3 in clinical trials? The FDA is proposing using AI to get trial data in real-time from EHRs and giving trial design feedback based on what it sees. No more batch processing could eliminate the wait...

In Chiesi’s Biggest M&A Deal yet, Pharma Buys HAE Company KalVista
Italian pharmaceutical group Chiesi announced the acquisition of U.S. biotech KalVista for approximately $1.5 billion in cash, marking its largest M&A transaction to date. KalVista, a specialist in hereditary angioedema (HAE), brings a late‑stage monoclonal‑antibody program and a pipeline of complement‑targeted...

The Trump Administration Is Shifting Federal Policy On Cannabis And Psychedelics
The Trump administration announced that marijuana will be re‑classified from Schedule I to Schedule III in states that have authorized medical use, and the Justice Department scheduled a June hearing on a nationwide re‑classification. The same administration issued executive orders to increase...
BIOTECanada Responds to Health Canada’s Gazette on Modernizing Clinical Trials Regulations
Health Canada has released a Gazette notice proposing modernized clinical‑trial regulations and draft guidance for decentralized trials. BIOTECanada welcomed the initiative but urged that the new rules align with the U.S. FDA and European EMA to avoid duplicative requirements. The...
GLP-1 Drugs May Lower CV Risk in TAVI Patients With Diabetes or Obesity
A retrospective analysis of 1,708 matched TAVI patients shows that glucagon‑like peptide‑1 (GLP‑1) receptor agonists cut the relative risk of major adverse cardiovascular events (MACE) by 37% and all‑cause mortality by 39% at one year. The benefit was consistent in...

Small Stem Cell Edit Generates Persistent Antibody Protection
Researchers showed that editing a small number of blood stem cells can reprogram the immune system to continuously produce therapeutic proteins, including powerful antibodies that are normally hard to generate. In mice, this approach created long-lasting, boostable protection against infections...

2C‑B Shows Dose‑Dependent Effects Similar to MDMA and Psilocybin
Acute dose-dependent effects of 4-bromo-2,5-dimethoxyphenethylamine (2C-B) compared with 3,4-methylenedioxymethamphetamine (MDMA) and psilocybin in a double-blind, placebo-controlled study in healthy participants https://t.co/OEJPk0XTqg
PTC Therapeutics Sees 52% Slowing of Huntington's Disease with Votoplam
PTC Therapeutics reported that participants with Stage 2 Huntington's disease receiving 10 mg of Votoplam experienced a 52% slowdown in disease progression over 24 months, compared with a matched natural‑history cohort. The data, presented in a press release on April 28, 2026, reinforce...

We Covered Psychedelics for Brain Injury Back in 2021
psychedelics for brain injury trending now, you say? yes, we wrote about it in 2021. full article below. https://t.co/RcXtd2XIe0

Payment Models Taking Shape for Advanced Therapies: Fran Gregory, PharmD
Advanced therapies priced up to $4 million are prompting a rethink of how they are funded. Fran Gregory of Cardinal Health says the gap between limited launch data and payer expectations can be bridged with robust real‑world evidence. The company is...

Olema Oncology Appoints Prakash Raman, Ph.D., to Board of Directors
Olema Oncology announced the appointment of Dr. Prakash Raman, a veteran biotech executive, to its Board of Directors. Raman, currently CEO of InduPro Therapeutics, brings more than two decades of experience in business development and corporate strategy, including senior roles...

Rocket Pharmaceuticals Secures $180 Million From Priority Review Voucher Sale
Rocket Pharmaceuticals sold its FDA rare‑pediatric‑disease priority review voucher for $180 million, extending its cash runway into the second quarter of 2028 and providing non‑dilutive capital for its cardiovascular gene‑therapy pipeline. The deal underscores the growing market for FDA vouchers as...