Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

EyePoint Inc (EYPT) Q1 2026 Earnings Call Transcript
EyePoint Pharmaceuticals reported a dramatic revenue drop to $600,000 for the quarter, reflecting the reversal of deferred YUTIQ license revenue, while operating expenses surged to $71 million as Phase 3 trials for its DuraVu platform intensified. The company posted a net loss of $68 million for the quarter and $232 million for the year, but its cash and investments of $306 million, bolstered by a $173 million financing, should sustain operations into 2027. Clinical milestones include the first patient dosing in pivotal Phase 3 DME studies and anticipated mid‑2026 top‑line data for wet AMD, supported by a safety record of no serious ocular or systemic events. A new chief commercial officer, Mike Campbell, was added to steer a potential U.S. launch, and the cGMP manufacturing facility is fully operational for future supply.

Cytokinetics Scores Late-Stage Win in Non-Obstructive HCM as BMS Plans Camzyos Restart
Cytokinetics announced that its experimental drug Myqorzo (aficamten) achieved both primary endpoints in the Phase III ACACIA‑HCM trial, marking the first late‑stage success for a therapy targeting non‑obstructive hypertrophic cardiomyopathy (HCM). The result propelled the company’s stock up 17%, pushing its...

Tricare Drug Prices Hidden, Generics Offer Cheaper Alternative
Now I’m going to flip it. Tricare has the VA pharma contract. @ewarren tried to get a copy of their contract, with pricing to the taxpayer. They won’t provide it. She has done a good...
Advances in Genetic Medicine Took Center Stage at INSAR
At INSAR in Prague, researchers highlighted a surge of genetic‑medicine breakthroughs aimed at autism, focusing on rare variants such as SCN2A, SHANK3 and UBE3A. Techniques ranging from CRISPR gene editing to antisense oligonucleotides and epigenome editing demonstrated tangible symptom improvements,...

Caris Life Sciences Secures MolDX Approval for Ultra-Deep Myeloid Cancer Sequencing Test
Caris Life Sciences announced that its Caris ChromoSeq test has earned MolDX approval, a CMS‑backed endorsement that could unlock broader reimbursement for comprehensive genomic profiling of acute myeloid leukemia and related blood cancers. The clearance validates the assay’s ultra‑deep sequencing depth—up...

CMS Proposes Overhaul of Drug Prior‑Authorization Rules, Targeting Faster Approvals
The Centers for Medicare & Medicaid Services released a proposal on April 10, 2026 to require electronic prior authorization for drugs across Medicare Advantage, Medicaid, CHIP and ACA Marketplace plans. The rule would impose new data standards, tighter decision windows...

OTC Monograph Drug User Fee Amendments (OMUFA): Understanding FY 2026 User Fees and Registration - 05/05/2026
The FDA’s Center for Drug Evaluation and Research hosted a May 5, 2026 webinar to explain the Over‑the‑Counter Monograph Drug User Fee Amendments (OMUFA) for fiscal year 2026. The session covered fee structures, registration requirements for OTC monograph facilities, payment timelines, and penalties...
FDA Blocks Proven Therapies, Permits Untested Peptides
So let me get this straight - this FDA 1) won’t approve life saving melanoma and Huntington’s disease drugs based on BS objections related to control groups that would not be ethical 2) blocks large study showing exquisite safety of...

The BioPharm Brief: Breakthrough Biologics and Long-Term Wins in IBD and Beyond
Viridian Therapeutics reported positive Phase III REVEAL‑2 data for veligrotug (VRDN‑001) in chronic thyroid eye disease, showing statistically significant reductions in proptosis and higher overall response rates. Eli Lilly presented four‑year long‑term results for mirikizumab (Omvoh) in ulcerative colitis, with a substantial...

ADMA Biologics Wins FDA Label Expansion for ASCENIV, Adding Pediatric Patients 2+ Years
ADMA Biologics announced that the FDA approved a supplemental BLA expanding ASCENIV’s indication from patients 12 years and older to include pediatric immune‑compromised patients two years of age and up. The move opens a new market segment for the company’s patented...

Pharmaceutical Executive Daily: BioNTech Reports 2026 First Quarter Results
Cellenkos secured FDA clearance to start a Phase 1b/2a trial of its off‑the‑shelf allogeneic T‑reg therapy CK0802 for steroid‑refractory graft‑versus‑host disease, with enrollment slated for late 2026. BioNTech reported first‑quarter 2026 revenue of $138 million, a 35% drop from the prior year,...

Questions and Answers on Current Good Manufacturing Practice Regulations | Production and Process Controls
The FDA released a detailed Q&A clarifying current Good Manufacturing Practice (CGMP) expectations for pharmaceutical manufacturers. It addresses equipment labeling, warehouse sampling of containers, media‑fill contamination sources, and the number of validation batches required for new products. The guidance also...

FDA Clears Investigational New Drug Application for Phase Ib/IIa Trial of CK0802 in Steroid-Refractory Graft-Versus-Host Disease
The U.S. Food and Drug Administration has cleared Cellenkos' investigational new drug application for CK0802, paving the way for a Phase Ib/IIa trial in patients with steroid‑refractory graft‑versus‑host disease (GVHD). The mid‑stage study will evaluate safety, tolerability and early efficacy, using...
First Patient Enrolled in Massive Heart Failure Trial
CVRx has begun enrolling patients in BENEFIT‑HF, a pivotal trial of its Barostim implant for heart failure. The study plans to enroll roughly 2,500 NYHA Class II‑III patients across the United States and Germany and will run through 2032. Participants must...

F.D.A. Blocked Publication of Research Finding Covid and Shingles Vaccines Were Safe
The U.S. Food and Drug Administration blocked publication of several agency‑conducted studies that found Covid‑19 and shingles vaccines to be safe. The withdrawn research, funded with public dollars, had been slated for peer‑review journals and a drug‑safety conference. FDA officials...

White House Estimates Trump’s Big Pharma Dealmaking Will Save Americans $529 Billion over the Next 10 Years
White House economists estimate that President Trump’s “most‑favored‑nation” drug‑pricing deals could save Americans $529 billion over the next ten years, with Medicaid projected to cut $64.3 billion in costs. The analysis, prepared by the Council of Economic Advisers, assumes 17 major pharmaceutical...

Austin Russian: How Fragmentation Delays Rare Disease Therapy Access
Austin Russian, SVP of Program Excellence at PANTHERx Rare, warned that fragmentation across prescribers, insurers, pharmacies and manufacturers slows patient access to orphan drugs. As more rare‑disease therapies reach the market, the lack of a single coordinating entity creates miscommunication...

Partial Reprogramming Concern Altos Labs Is Becoming Less Stealthy
Altos Labs, launched in 2022 with roughly $3 billion in private funding, is intensifying its public profile as it pursues partial cellular reprogramming to reverse organ aging. The company is racing alongside rivals such as Life Biosciences, which has just begun...

PharmaLogic Expands Radiopharmaceutical Footprint With New Atlanta Facility
PharmaLogic announced the opening of a new PET radiopharmaceutical manufacturing facility in Atlanta, Georgia, as part of its $250 million US expansion. The state‑of‑the‑art plant will increase production capacity, accelerate delivery to Southeast hospitals, and serve as an innovation hub for...

5th Circuit Reinstates In‑Person Mifepristone Rule, Prompting Supreme Court Stay
The 5th U.S. Circuit Court of Appeals reinstated an in‑person dispensing requirement for the abortion pill mifepristone, prompting Danco Laboratories to seek a stay from the Supreme Court, which granted a one‑week pause. The back‑and‑forth highlights how even long‑approved drugs...

FDA Approvals for Aging Therapies: Updated Insights
My 20min talk on FDA approvals of aging therapies & more coming soon from the Dec 2025 Longevity Summit at the Buck Institute is now on YouTube: https://t.co/L024jUz9iT It's an updated version of the one I gave at Vitalist Bay May'25. I'll be...

Viridian Data Lift Prospects for Thyroid Eye Disease Drug
Viridian Therapeutics announced that its subcutaneous drug elegrobart met primary endpoints in a Phase 3 trial for chronic thyroid eye disease, showing 50%‑54% response rates versus 15% for placebo. The once‑monthly regimen also improved double vision in 61% of patients, while...

FDA Commissioner Defends Agency's Drug Approval Decisions After Wave of Backlash
FDA Commissioner Marty Makary faced intense media and political backlash after the agency rejected Replimune’s melanoma drug candidate. In a CNBC interview, Makary emphasized that three independent FDA panels reached the same safety conclusion and rejected accusations of bias toward...
Ascidian Completes Adult Dose Escalation, Opens Pediatric Enrollment in STELLAR Gene Therapy Trial
Ascidian Therapeutics announced the completion of the adult dose‑escalation portion of its Phase 1/2 STELLAR trial for the RNA‑editing gene therapy ACDN-01 and opened enrollment to pediatric patients 12 years and older. The move adds a 10‑patient adult cohort and launches...
Bio Korea 2026 Kicks Off with Spotlight on Oligonucleotides
In early May 2026, several biotech firms announced pivotal milestones. Axsome Therapeutics received FDA clearance for Auvelity (AXS‑05) to treat agitation in Alzheimer’s disease, a condition affecting up to 75% of patients. Sonire Therapeutics began its U.S. Sunrise II trial of...

Pfizer Scrubs Trillium Pipeline, CD47 Concept Proves Bust
Sometimes in biotech you can get lucky and win even though you were dead wrong. $PFE just dumped the rest of the Trillium ( $TRIL) pipeline - it all came to naught. I made out nicely when PFE bought TRIL,...
Trump Plan Cuts Drug Costs 6% but Still Double Abroad
WH says Trump's drug plan will reduce drug spending by about 6%, still leave us paying twice as much as people Canada and Europe.

Second Life for Gene Therapy; Takeda Phase 2/3 Win; UK Cancer Biotech's $83M
A Cleveland‑based biotech announced a revamped gene‑therapy platform that could give a previously stalled program a second chance, while Takeda disclosed positive Phase 2/3 results for its oncology candidate. Across the Atlantic, a UK cancer‑focused biotech raised roughly $83 million to accelerate...

Brain IGF-1 Shields Male Mice From Age‑Related Decline
Central IGF-1 protects against features of cognitive and sensorimotor decline with aging in male mice https://t.co/7U25aLdLFv
Awaiting Faster
Looking forward to the implementation of quicker/less bureaucratic path to first in human and only one pivotal study required for approval. Question is when these changes will happen and will all FDA offices/divisions support.
ADAPT OCULUS Trial Shows Promising Results in Treatment Efficacy for Ocular MG: Carolina Barnett-Tapia, MD, PhD
The ADAPT OCULUS Phase III trial evaluated efgartigimod alfa (VYVGART) in patients with ocular myasthenia gravis (oMG). In a double‑blind, placebo‑controlled arm, participants receiving the drug showed statistically significant reductions in ptosis and diplopia versus placebo. An open‑label extension confirmed continued...

BioNTech's Remarkable Vaccine Supply Chain: A Bittersweet Legacy
For those of us (OK, mainly me) obsessively tracking the details of BioNTech's *INCREDIBLE* vaccine supply chain buildout during COVID-19, this piece is bittersweet. What BioNTech achieved for humanity in 2020-21 is simply an incredible story. https://t.co/H0YcTszLjr
Viridian Data Boosts Thyroid Eye Disease Drug Prospects
Viridian data lift prospects for thyroid eye disease drug https://t.co/fjfkebQ99D by @Lilah_Alvarado $VRDN + 28% $AMGN

Prolific Machines Sets Monoclonal Antibody (mAb) Manufacturing Record with Light-Controlled Platform
Prolific Machines, an SOSV portfolio company, announced a record 21 g/L monoclonal antibody titer in a 15‑day intensified fed‑batch CHO run using its light‑controlled optogenetic platform. The photomolecular system lets operators toggle gene expression in real time with light, offering reversible,...

Biotech Poised to Create Universal Anti‑venom Breakthrough
Day 1 @SynBioBeta first panel on building a universal anti-venom "We haven't innovated in 125yrs, since injecting horses with venom worked okay" We're now on the cusp of a universal anti-venom thanks to modern biotech https://t.co/wNbPwXjHPi
Orexin Therapies Unlock New Horizons Beyond Narcolepsy
Orexin drugs for narcolepsy and more. Listen to Richard Pops @popsalks discuss the emerging opportunity, and his 35-year-career, on The Long Run. Sponsored by @AlphaSenseInc & Dash Bio. https://t.co/Qr6oJ8AwzB

FDA Flags eBay Listing over GLP-1 Claims in Rare Warning Letter
The FDA sent a rare warning letter to an eBay seller for making GLP‑1 and blood‑sugar support claims, marking only the fourth eBay‑related enforcement in recent years. Unlike typical cases, the agency did not purchase or test the product, relying...

Emcure Doubling Down on Biosimilars Pipeline: MD Satish Mehta
Emcure Pharmaceuticals is expanding its biosimilars pipeline as a wave of biologic patent expiries creates market tailwinds. The company currently sells six biosimilars through its Gennova Biopharmaceuticals subsidiary and will now seek external partners to bring additional products to India...
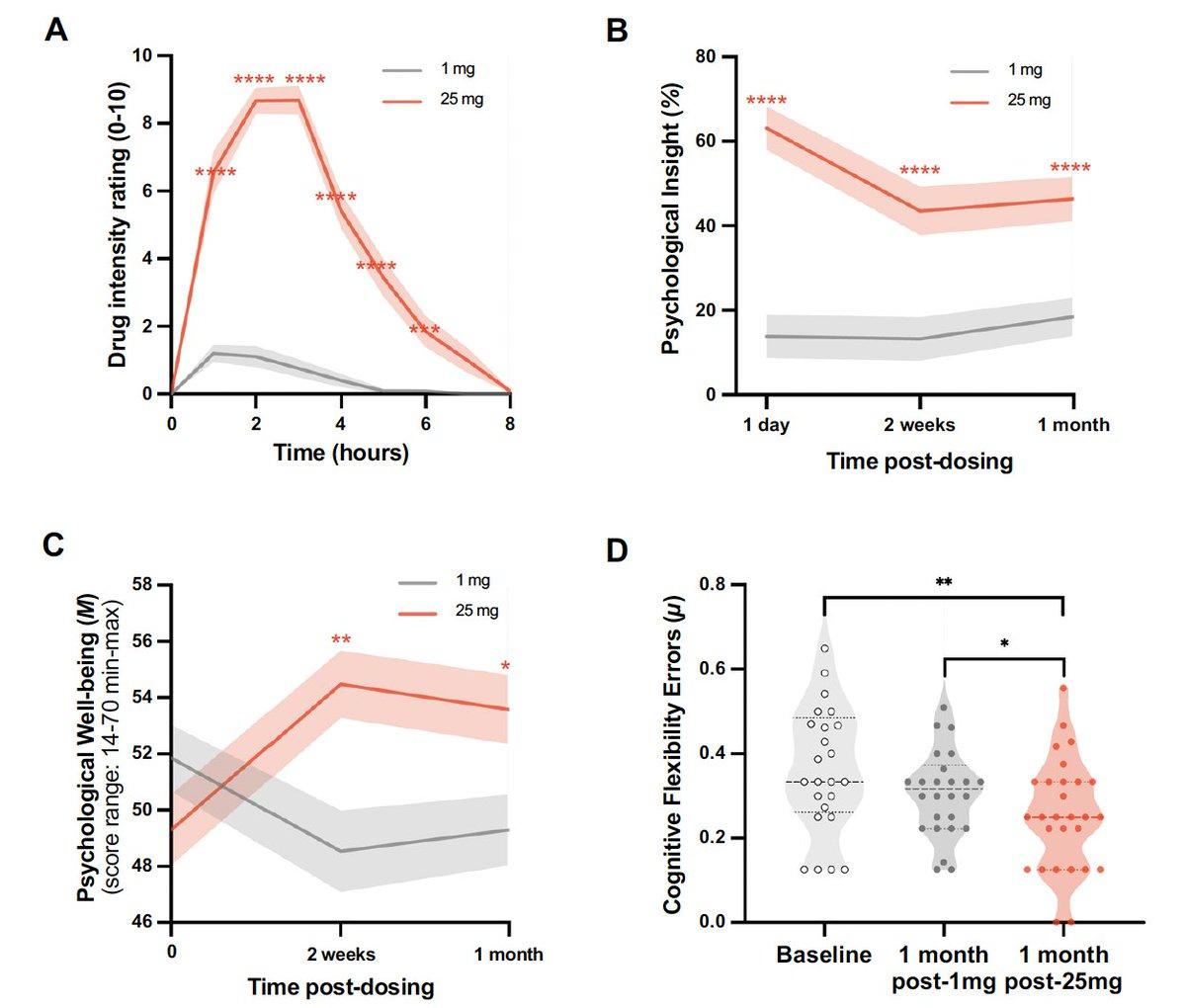
Single 25 Mg Psilocybin Dose Triggers Month-Long Brain Changes
A single 25 mg dose of psilocybin leads to brain structural changes that were seen at 1 month. From a cross-over study of 28 healthy volunteers, no prior psychedelic, who also were also assessed after 1 mg. Behavioral results in...

Bracco Launches BubbleGen™ Early Access Program for Microbubble-Based Cell Selection and Activation at ISCT
Bracco Imaging announced an Early Access Program for its new BubbleGen™ technology, which uses buoyant microbubbles to isolate and activate specific cell subtypes. The platform offers a one‑step, magnetic‑residue‑free alternative to traditional bead‑based cell separation, initially demonstrated with CD3⁺ T‑cell selection...
New Pharmaceutical Reform Alliance Poll: Americans Agree on One Thing – Rein in Big Pharma
The Pharmaceutical Reform Alliance released a new national poll of 1,524 registered voters showing overwhelming bipartisan support for prescription‑drug pricing reform. Eighty‑nine percent of respondents favor reform, while 68% say drug prices have risen and 94% blame pharmaceutical companies for...

NImmune Biopharma Announces Presentations at Digestive Disease Week 2026 Supporting a Differentiated Profile and Superior Efficacy of Oral, Once-Daily NIM-1324...
NImmune Biopharma presented Phase 1 data for its oral LANCL2 drug NIM‑1324 at Digestive Disease Week, showing safety, tolerability, target engagement and superior efficacy versus existing IBD therapies. The study met all primary and secondary endpoints with no dose‑limiting toxicities and...
Hepta Reveals Blood-Based Epigenetic Signatures of GLP-1 Response, Enabling Precision Medicine in Obesity and MASH
Hepta unveiled a blood‑based cfDNA methylation assay at Digestive Disease Week 2026 that can identify patients who will lose at least 10% of body weight on semaglutide before the first dose. The SAMARA trial showed baseline epigenetic signatures distinguished responders...
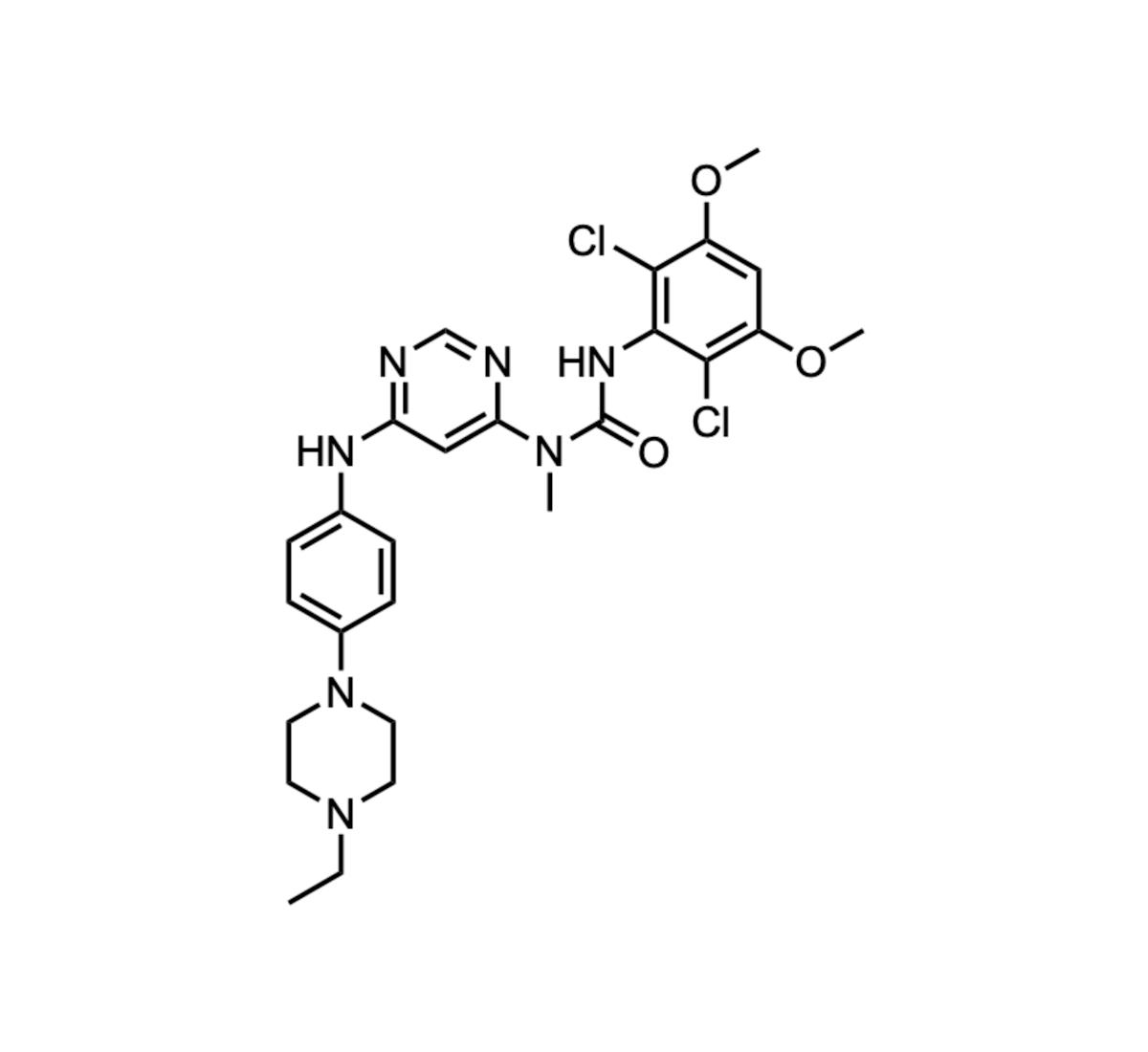
Infigratinib
Infigratinib, a pan‑FGFR inhibitor previously approved for cholangiocarcinoma, is being repurposed to treat achondroplasia. After its FDA accelerated approval was rescinded in 2024 due to enrollment challenges, BridgeBio reported that the Phase 3 PROPEL 3 trial met its primary endpoint in February 2026....

Connected by Design: How AI and Automation Are Transforming Drug Discovery at BMS
Bristol Myers Squibb (BMS) is shifting from isolated AI tools to an integrated, learning‑driven ecosystem that connects data, models, and automation across discovery and development. The company highlights its shared data backbone, AI co‑scientists, and lab‑in‑the‑loop automation as foundational layers,...

STAT+: Are Analysts Too Quick to Gloss over Lilly’s Liver Case?
Analysts are being criticized for downplaying a recent liver safety issue at Eli Lilly, raising concerns about market oversight. Meanwhile, biotech earnings showed mixed results, with Vertex shelving an mRNA cystic fibrosis candidate and Pfizer and Alkermes delivering near‑consensus Q1 numbers....

Viridian Reports Positive Phase III REVEAL-2 Data for Elegrobart in Chronic Thyroid Eye Disease
Viridian Therapeutics announced that its subcutaneous IGF‑1R antibody elegrobart met the primary endpoint in the phase 3 REVEAL‑2 trial for chronic thyroid eye disease (TED). Patients receiving the drug every four or eight weeks showed 50‑54% proptosis responder rates and 55‑61%...

5 Key April FDA Approvals Signal Momentum Across Rare, Chronic Diseases
April 2026 saw five FDA approvals spanning HIV, type 1 diabetes, chronic spontaneous urticaria, genetic hearing loss, and systemic lupus erythematosus. Merck’s Idvysno introduced the first tenofovir‑free, non‑INSTI two‑drug HIV regimen, while Sanofi’s teplizumab received clearance for children as young as...

Cytospire Secures $83M to Develop Novel T‑Cell Engager
Cytospire hauls in $83M for a new type of T cell engager https://t.co/19BkL5JiHk by @gwendolynawu #biotech #startups

Key Biosimilars Events of April 2026
In April 2026 the biosimilar landscape saw a wave of regulatory approvals and strategic deals. The European Commission cleared Poherdy (pertuzumab) and Tuyory (tocilizumab), while Health Canada and China’s NMPA approved multiple denosumab biosimilars and granted IND clearances for Henlius’...