Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Brolucizumab Superior in Preserving Visual Acuity in Proliferative Diabetic Retinopathy
A phase‑3 CONDOR trial of 689 adults with proliferative diabetic retinopathy (PDR) showed that intravitreal brolucizumab outperformed panretinal laser photocoagulation (PRP) in preserving visual acuity over 54 weeks. The brolucizumab arm gained a mean BCVA change of +0.2 letters versus a loss of –4.2 letters with PRP. Patients receiving brolucizumab also saw more eyes achieve no‑PDR (187 vs 65), a lower rate of center‑involved diabetic macular edema (31% vs 73%), and fewer ocular adverse events. The findings suggest a viable anti‑VEGF alternative to laser therapy for PDR.

How Specialty Drug Commercialization Differs in Canada From the US
Jessica Lovett, VP of Commercial Strategy at Innomar Strategies, outlined a 24‑month planning horizon for specialty drug launches in Canada, emphasizing early coordination with Health Canada. The roadmap moves from pre‑development regulatory and import considerations to a 12‑18‑month development and...

Clinical Trials Day
Clinical Trials Day on May 20 commemorates James Lind’s 1747 scurvy experiment, widely regarded as the first randomized clinical trial. The observance highlights the enduring role of trials in generating the safety and efficacy data that underpin FDA drug approvals. FDA’s...

BsUFA IV: Fiscal Years 2028-2032
The FDA announced the kickoff of the fourth Biosimilar User Fee Act (BsUFA IV) reauthorization, covering fiscal years 2028‑2032. A public meeting was held on December 3, 2025 to launch the process, following the 2022 law that reauthorized BsUFA II. BsUFA III expires in September 2027,...

In a First, Scientists Are Rewinding Human Cells Back to a ‘Youthful’ State. Is This the Dawn of Immortality?
Scientists are advancing partial cellular reprogramming to reverse age‑related decline while preserving cell identity. YouthBio Therapeutics is preparing a first‑in‑human trial of its brain‑targeted YB002 program for Alzheimer’s after receiving FDA feedback. Parallel efforts such as Life Biosciences’ ER‑100 aim...

Celcuity’s Gedatolisib Beats Novartis’ Piqray in Phase III Advanced Breast Cancer Trial
Celcuity announced that its PI3K‑mTOR inhibitor gedatolisib achieved a progression‑free survival benefit versus Novartis' alpelisib (Piqray) in a Phase III study of advanced breast cancer patients with PIK3CA mutations. The data could reshape treatment options for a disease that affects millions...

FDA Vaccine Studies Censored by Trump Admin After Finding Benefits of Shots
The Food and Drug Administration, under the Department of Health and Human Services, blocked the publication of two internal studies that demonstrated the safety and efficacy of COVID‑19 vaccines, and prevented two Shingrix (shingles vaccine) abstracts from being presented at...

Pfizer Posts $14.5 B Q1 Revenue, Reaffirms 2026 Guidance
Pfizer announced first‑quarter 2026 revenue of $14.5 billion, a 5% year‑over‑year increase, and a 22% rise in operational revenue from launched and acquired products. The company reaffirmed its full‑year 2026 financial guidance while highlighting progress in oncology and obesity pipelines.

Tofersen, a New Treatment for A.L.S., Reverses Symptoms for Some
Tofersen, the first FDA‑approved therapy targeting the SOD1 genetic form of ALS, is showing functional gains in a subset of patients. In a case highlighted by the New York Times, 58‑year‑old Amanda Sifford’s lung capacity rebounded from 48% to 86%...
LTZ Therapeutics Secures $38M to Advance Myeloid Engager Immunotherapy Pipeline
LTZ Therapeutics announced an oversubscribed $38 million financing round led by GL Ventures, bringing its total capital to roughly $130 million since 2022. The funds will propel the Universal Myeloid Cell Engager (U‑MCE) platform, supporting the Phase 1 trial of lead bispecific antibody...

Latus Bio Secures $97 Million Series A to Scale Gene‑Therapy Access
Latus Bio closed a $97 million Series A financing, with a $43 million extension led by 8VC, to accelerate its AAV‑based gene‑therapy pipeline. The capital will fund IND filing for Huntington’s disease candidate LTS‑201 and a CLN2 trial, underscoring investor confidence in...

Cannabis Execs Anticipate Tax Benefits From Rescheduling
The DOJ and DEA reclassified medical cannabis as a Schedule III drug on April 23, ending its Schedule I status. This change eliminates the IRS 280E restriction for medical cannabis businesses, potentially cutting tax burdens by up to half. Industry leaders Curaleaf and Green...

Heavy-Chain BsAbs More Manufacturable than Light-Chains
Bispecific antibodies (BsAbs) have grown to 19 FDA approvals and roughly 250 candidates in development, but scaling their production remains a bottleneck. A recent study by Universidad Nacional Autónoma de México researchers compared six BsAb architectures and linked design to...

Researchers’ Spinout Focuses on Simplifying Viral Vector Purification
Researchers at North Carolina State University have spun out ChromaGenix to commercialize synthetic peptide ligands for affinity purification of viral vectors used in gene therapies. The peptide ligands are cheaper, more stable and less immunogenic than traditional protein ligands, cutting...

Moss Powering the Next Drug Frontier
Eleva is commercializing a moss‑based biomanufacturing platform that can produce complex glycoproteins difficult to express in traditional CHO or yeast systems. The German firm has advanced its first candidate, a recombinant alpha‑galactosidase for Fabry disease, into clinical trials and is...
Regulators Should Rely on Peers’ GMP Audits to Cut Inspection Burden
Biopharma manufacturing sites face an average of 2.68 GMP inspections per year, each lasting up to nine days, and preparation can take six months to a year. The International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) argues that regulators should...

First U.S. Patients Treated With Microrobotic Surgery For Alzheimer’s
A microrobotic surgery trial for Alzheimer’s disease began at Baptist Health in Jacksonville, treating the first patient with moderate disease. Medical Microinstruments Inc. (MMI) plans to enroll 15 participants and monitor them for a year, aiming to clear cervical lymph‑node...
Financings for May 6, 2026
UK‑based Cytospire Therapeutics announced a £61 million (≈$82.7 million) Series A round to accelerate its EGFR‑targeted T‑cell engager pipeline for solid tumours. The capital backs multiple pre‑clinical programs and reflects strong investor confidence in next‑generation immunotherapies. Bio Korea 2026 highlighted a shift in...

Other News to Note for May 6, 2026
Cytospire Therapeutics announced a £61 million (≈$82.7 million) Series A round to advance its next‑generation T‑cell engager platform targeting the epidermal growth factor receptor (EGFR) in solid tumours. The funding will accelerate pre‑clinical and early‑clinical programs aimed at delivering more selective immunotherapies. Meanwhile,...

IPLEDGE Hurdles Block some Inmates From Receiving Isotretinoin in Jails, Prisons
The FDA‑run iPLEDGE REMS program blocks many incarcerated patients from receiving isotretinoin because it requires pharmacies to register with an NCPDP number—a identifier jails and prisons do not possess. In addition, the system mandates a phone number or email for...

Eli Lilly Opens First Dedicated Genetic Medicine Facility
Eli Lilly inaugurated Lilly Lebanon Advanced Therapies, its first dedicated genetic‑medicine manufacturing site in Lebanon, Indiana, alongside a $4.5 billion capital infusion. The investment lifts Lilly’s Indiana spending to over $21 billion since 2020 and its U.S. commitments to more than $50 billion. The...

Briumvi’s Optional Subq Gives It Market‑share Edge
On the $TGTX call the following was casually mentioned in response to a question about subq: "We feel actually very confident in our ability to deliver a quarterly product." It's important to understand that Briumvi will be the only product that...
Heroic Psilocybin Dose Dramatically Rewires Brain Connectivity
“Scientists gave people a “heroic” dose of psilocybin and then looked at their brain. Here’s what happened” https://t.co/M9FjCRGNNq

CoCoGraph AI Model Generates Molecules that Comply with Rules of Chemistry
Researchers at Universitat Rovira i Virgili have unveiled CoCoGraph, an AI diffusion model that generates synthetic molecules while strictly adhering to fundamental chemical rules. By progressively disordering and reconstructing real molecules, the system ensures valid bond counts and produces chemically...
80+ Therapeutic Peptides Approved Across Major Disease Areas
Did you know... over 80 therapeutic peptides have been approved to treat a wide array of diseases, ranging from infectious diseases, cardiovascular, dysmetabolic diseases, and cancer: https://t.co/kW4zgAcmE3
Scientists Remove Essential Amino Acid, Redefining Life's Code
All Life Uses 20 Amino Acids. Scientists Just Deleted One in Bacteria The synthetic bacteria push the limits of life and could open the door to designer proteins and new medicines https://t.co/mVZrQ0z7vz https://t.co/zFzSpnrk6h
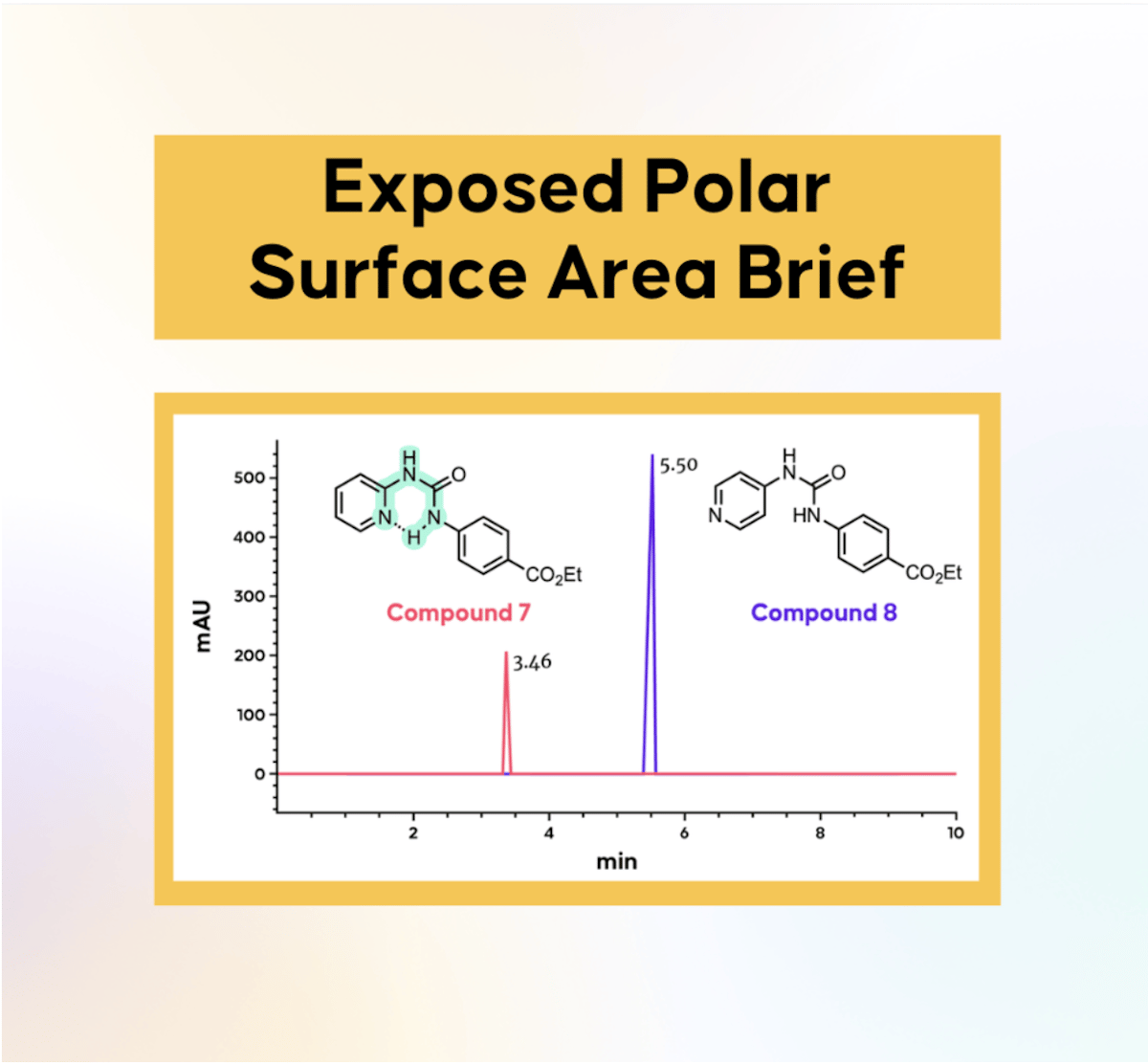
EPSA: A Useful Metric Across Chemical Space
The article highlights EPSA (Experimental Polarity Surface Area) as a robust metric for assessing molecular polarity across broad chemical space. Unlike traditional PSA, EPSA is derived from supercritical fluid chromatography, offering experimental insight into a compound’s three‑dimensional polarity profile. The...
Pill GLP‑1 Drugs Activate Amygdala Reward Circuit, Not Hypothalamus
Discovery of a new brain reward circuit in the amygdala for small molecule (pill) GLP-1 drug effect, unlike the injectables (hypothalamus), as shown in mice https://t.co/3lkfxROh5Z
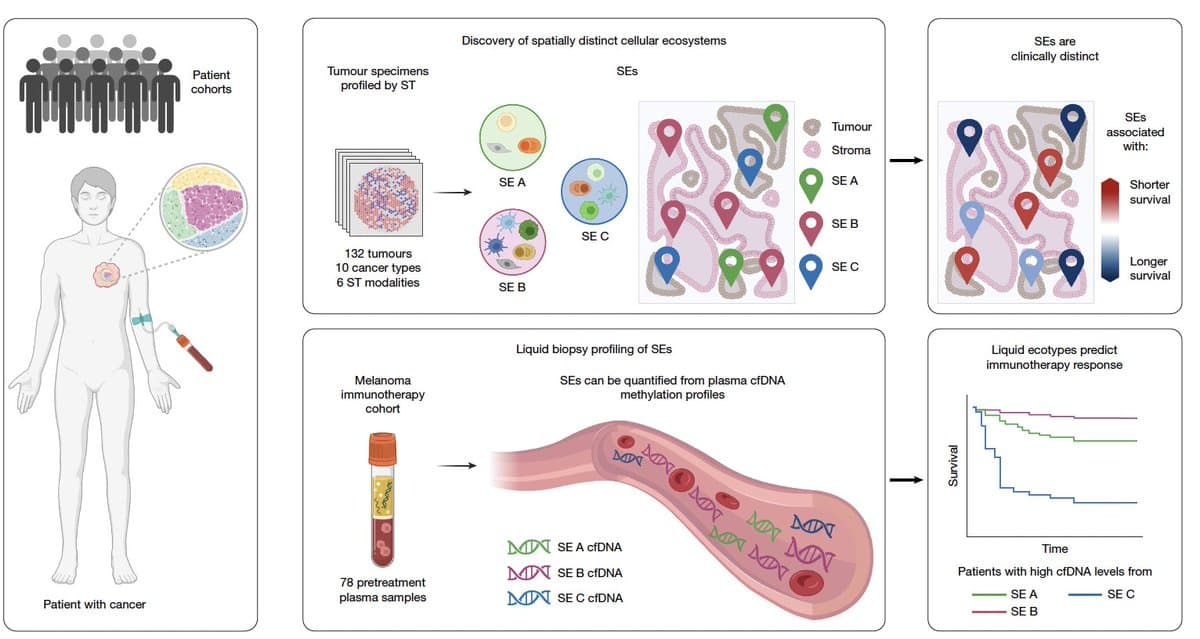
Blood Test Now Gauges Tumor Microenvironment Non‑invasively
We've known how important the tumor microenvironment is for cancer progression and treatment, but we never had a non-invasive blood test to assess it. Today, as reported @nature, one has been discovered https://t.co/rDaF5YUwTq

A New Kind of CRISPR Could Treat Viral Infection and Cancer by Shredding Sick Cells' DNA
Researchers have engineered a novel CRISPR protein, Cas12a2, that acts as a molecular shredder, destroying DNA in cells that express a specific RNA trigger. In vitro, Cas12a2 cut the growth of KRAS‑mutant lung‑cancer cells by 50% and eliminated over 90%...

Waiv Enters Collaboration with Daiichi Sankyo to Deliver AI-Derived Biomarkers for ADC Program
Waiv, the Paris‑based AI precision‑testing firm formerly known as Owkin Dx, has partnered with Daiichi Sankyo to lead digital pathology biomarker discovery for an antibody‑drug conjugate (ADC) program. The collaboration will apply Waiv’s end‑to‑end computational pathology platform to early‑phase trial data,...
IV Ketamine Shows Rapid Benefits for Suicide Risk, Depression in Major Depressive Episodes
A new meta‑analysis of 26 randomized trials involving 1,166 patients shows that intravenous ketamine rapidly alleviates both suicidal ideation and depressive symptoms in major depressive episodes. Single infusions cut suicidal scores by an SMD of –0.69 within 24 hours and depressive...

Simulations Plus and NVIDIA Collaborate to Scale GPU-Accelerated, AI-Assisted Modeling Workflows
Simulations Plus announced a technical collaboration with NVIDIA to embed GPU‑accelerated and AI‑assisted modeling into drug‑development workflows. The partnership will re‑engineer Simulations Plus’ PBPK, PK/PD and QSP engines for NVIDIA GPUs, delivering up to a 75% reduction in simulation runtimes...

Chicago Trip Reveals GLP-1 Adoption Still in Infancy
being in Chicago for a few days has made me realize how insanely early we are in the GLP-1 adoption cycle

Most Oncology Brands Lose Before Launch Day — New ZoomRx Analysis of 40+ Drugs Quantifies the Awareness Gap That Predicts...
ZoomRx’s 2026 "Laggards and Leaders" analysis of 44 oncology brands shows that pre‑launch physician awareness drives commercial success. Brands entering the market with about 75% aided awareness outperform those starting at 55%, a 25‑point gap that persists through four years....

Gene Therapy Field Focused on FDA Shifts in Rome
Even at a meeting in Rome, FDA shifts are top of mind for gene therapy field https://t.co/J9WXJqzqTY via @statnews

Understanding Supply Chain Constraints to Enable Growth for Cell & Gene Therapies
Cell and gene therapies are poised for growth, yet only 3 % of the 4,000+ candidates in development secure FDA or EMA approval. Prices ranging from $100,000 to $3 million per dose lack a unified reimbursement framework, creating payer uncertainty worldwide. Logistics...

The VESALIUS-CV Trial
In the VESALIUS-CV trial, researchers evaluated the impact of adding the PCSK9 inhibitor evolocumab to standard lipid‑lowering therapy in patients at high cardiovascular risk who had never experienced a myocardial infarction or stroke. Over a median follow‑up of 3.2 years,...
Cyclin E1 and CCNE1 Shift in Ovarian Cancer Post-PARP
The British Journal of Cancer study shows high‑grade serous ovarian carcinoma tumors develop resistance to PARP inhibitors by up‑regulating Cyclin E1 and amplifying the CCNE1 gene. Researchers compared pre‑ and post‑treatment samples using FISH and IHC, finding a significant rise in...

Novel Psychedelic Compound 25C-NBF Shows Rapid Antidepressant Effects without Addictive Traits
Researchers reported that the synthetic psychedelic 25C‑NBF rapidly induces dendritic growth and reverses depressive behaviors in male rodents after a single dose. The compound binds tightly to the serotonin 5‑HT2A receptor while showing minimal activity at 5‑HT2B, reducing heart‑valve risk....

GLP‑1 Use Surges, Unlocking $100B Anti‑obesity Market
i think roughly 40% of Americans are significantly overweight so still plenty of upside for GLPs. $LLY

STAT+: Novo Nordisk’s Less-Bad News on Its Wegovy Pill Boosts Earnings and Share Price
Novo Nordisk reported first‑quarter results that showed a modest improvement in its outlook for the obesity drug Wegovy. The company narrowed its adjusted sales decline forecast to 4%‑12% for the year, up from a prior 5%‑13% range, and aligned its...

STAT+: Pharmalittle: We’re Reading About a PhRMA Ad Campaign Aimed at 340B, Sales of Novo’s Obesity Pill, and More
PhRMA, the leading pharmaceutical lobby, has launched a seven‑figure TV advertising campaign aimed at the 340B Drug Pricing Program, accusing hospitals of misusing the discount to benefit themselves rather than low‑income patients. Meanwhile, FDA data show the rejection rate for...

Federal Funding Boosts Psychedelic Therapies, Yet Integration Support Lags
Federal agencies have announced new funding to accelerate psychedelic therapies, but practitioner Sergio Lialin warns that preparation and integration infrastructure has not kept pace, threatening long‑term benefits for patients.
Cytokinetics Announces Positive Phase 3 Results for Aficamten in Non‑Obstructive HCM
Cytokinetics disclosed that its pivotal Phase 3 ACACIA-HCM trial of aficamten met both primary endpoints—improved Kansas City Cardiomyopathy Questionnaire scores and higher maximal exercise capacity—versus placebo. The data, presented on May 5, position the cardiac myosin inhibitor as a potential first‑in‑class therapy...
Medicare Launches $50 GLP‑1 Bridge Pilot for Weight‑Loss Drugs
The Centers for Medicare & Medicaid Services announced a two‑year GLP‑1 Bridge pilot that will let eligible beneficiaries obtain Wegovy, Zepbound or Foundayo for a $50 monthly copay starting July 1, 2026. The program aims to lower cost barriers for...

Harvard-Backed Trials Still Face Skeptical Disagreement
Have a team at Harvard Medical School, and drug pipeline in human trials, and scientific papers dating back to 2008 challenging that assumption No - I don’t agree 🤣

BIOTECanada Responds to CDA-AMC Guidance for Incorporating Impacts on Informal Caregivers and Productivity Outcomes in Economic Evaluations.
BIOTECanada, together with Innovative Medicines Canada, submitted a formal response to the Canadian Drug Agency’s draft guidance on incorporating informal caregiver health‑related quality of life and productivity outcomes into economic evaluations. The association recommends expanding the societal perspective to all...

Understanding Drug Recalls: What to Know and What to Do
The FDA oversees drug recalls, publishing announcements on FDA.gov and maintaining a searchable database of all recalled products. Recalls are classified into three severity levels—Class I (life‑threatening), Class II (temporary health effects), and Class III (minor labeling or packaging issues). Consumers are advised...
FDA Accepts Opus Genetics’ OPGx‑LCA5 Gene Therapy Into Rare Disease Evidence Program
Opus Genetics announced that its investigational LCA5 gene therapy, OPGx‑LCA5, has been accepted into the FDA’s Rare Disease Evidence Principles (RDEP) program. The acceptance aligns the company with early‑stage regulatory collaboration for a Phase 3 trial targeting Leber congenital amaurosis type 5,...