Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Hoth Therapeutics Gains First European Clearance for HT-001 Oncology Trial in Spain
Hoth Therapeutics announced that Spain’s health authority has granted authorization for its Phase 2a CLEER trial of HT-001, a therapy aimed at reducing EGFR‑inhibitor skin toxicities. Interim results show meaningful efficacy and safety, paving the way for broader European patient enrollment.

Bayer to Acquire Perfuse Therapeutics, Expanding Its Ophthalmology Portfolio
Bayer disclosed plans to acquire biotech firm Perfuse Therapeutics, aiming to strengthen its ophthalmology pipeline. The deal, announced through Bayer’s newsroom, comes without disclosed financial terms but signals a strategic push into eye‑care. The acquisition could accelerate Bayer’s development of...
Gilead’s Yeztugo Sees 9,000 Q1 Prescriptions Yet Faces Insurance Hurdles
Gilead Sciences' long‑acting HIV pre‑exposure prophylaxis, Yeztugo, logged more than 9,000 prescriptions in Q1 2024, but clinicians say uptake is uneven. Insurers’ reliance on medical‑benefit billing and a $14,000‑plus price tag create out‑of‑pocket costs that deter many patients.

Spinogenix Secures FDA Fast Track for ALS Drug Tazbentetol
Spinogenix announced that the U.S. FDA granted Fast Track designation to its oral ALS candidate tazbentetol, accelerating development after Phase 2a data showed 82% of patients stabilized or improved and a 76% slower decline versus historical controls.

Akebia Therapeutics Inc (AKBA) Q1 2026 Earnings Call Transcript
Akebia Therapeutics reported 2025 total revenue of $236.2 million, a 47% increase year‑over‑year, driven by the commercial launch of its oral HIF‑PH inhibitor Vafseo and higher sales of the phosphate binder Auryxia. Vafseo now reaches 290,000 dialysis patients across 24 organizations,...

Monte Rosa Therapeutics Inc (GLUE) Q1 2026 Earnings Call Transcript
Monte Rosa Therapeutics reported Q1 2026 worldwide revenue of $141.2 million, a 14.3% year‑over‑year increase, driven primarily by strong U.S. sales of its AtriClip and Cryosphere product lines. Adjusted EBITDA rose to $17 million, nearly doubling the prior‑year quarter, while gross margin...
Ironwood Pharmaceuticals Inc (IRWD) Q1 2026 Earnings Call Transcript
Ironwood Pharmaceuticals reported 2025 LINZESS U.S. net sales of $865 million, matching guidance despite a 6% price erosion tied to Medicare Part D changes. Prescription demand grew 11% for the second year, maintaining a 45% market share in the IBS‑C and chronic...

There Is No Vaccine for Deadly Hantavirus: What that Means for Future Outbreaks
An outbreak of Andes hantavirus on the cruise ship MV Hondius left three confirmed cases and three deaths, highlighting the deadly potential of the rodent‑borne virus. The World Health Organization confirmed the strain has no approved treatments or vaccines. Virologist...

Corvus Pharmaceuticals Inc (CRVS) Q1 2026 Earnings Call Transcript
Corvus Pharmaceuticals reported Q1 2026 results highlighting accelerated R&D spending and a strengthened balance sheet after a $200 million public offering, giving the company cash runway into the second quarter of 2028. Clinical data from the Phase I/Ib atopic dermatitis trial...

INmune Bio Inc (INMB) Q1 2026 Earnings Call Transcript
INmune Bio reported Q1 2026 results showing a cash balance of $21.4 million, sufficient to sustain operations through Q1 2027, and a narrowed net loss of $5.4 million. The company highlighted significant progress on its CORDStrom platform, including approval of the U.K. Pediatric Investigation...

Zymeworks Inc (ZYME) Q1 2026 Earnings Call Transcript
Zymeworks reported a PDUFA deadline of August 25, 2026 for its zanidatumab HER2‑positive gastroesophageal adenocarcinoma (GEA) indication, with a supplemental BLA filed in China. The company stands to receive up to $265 million in milestone payments contingent on U.S. and Chinese...
Absci Corp (ABSI) Q1 2026 Earnings Call Transcript
Absci Corp (ABSI) reported Q1 2026 revenue of $200 K while R&D expenses rose to $19.3 M as the company advances its lead prolactin‑receptor antibody ABS‑201. The Phase 1/2a androgenic alopecia trial is on track, with safety data due next month and interim...

Harmony Biosciences Holdings Inc (HRMY) Q1 2026 Earnings Call Transcript
Harmony Biosciences reported record Q1 product revenue of $243.8 million, a 21% increase year‑over‑year, driven by sustained demand for its Wakix sleep‑wake therapy. The company added roughly 400 patients per quarter, reaching about 8,500 on Wakix, and reaffirmed 2026 revenue guidance...

Lexicon Pharmaceuticals Inc (LXRX) Q1 2026 Earnings Call Transcript
Lexicon Pharmaceuticals reported Q1 2026 revenue of $5.5 million, largely from a $4.3 million Novo Nordisk licensing deal and INPEFA sales. The company cut its quarterly net loss to $15.5 million and reduced annual operating expenses by $129.5 million, boosting cash to $125 million after...

Crinetics Pharmaceuticals Inc (CRNX) Q1 2026 Earnings Call Transcript
Crinetics reported $10.3 million net product revenue from Palsonify in Q1 2026, driven by 232 new patient enrollments and a prescriber base that grew to 263 unique physicians. Treatment‑naive patients rose to 15 % of enrollments, indicating expanding first‑line use. Reimbursement coverage...

Arrowhead Pharmaceuticals Inc (ARWR) Q2 2026 Earnings Call Transcript
Arrowhead Pharmaceuticals reported accelerating uptake of its newly launched triglyceride‑lowering therapy REDEMPLO, surpassing 400 prescriptions with a 40% surge in the last month. The company announced a price reduction to $45,000 per patient per year and secured payer policies that...

Scholar Rock Holding Corp (SRRK) Q1 2026 Earnings Call Transcript
Scholar Rock Holding Corp reported $368 million in cash and secured a $550 million debt facility, providing ample liquidity for 2026 initiatives. The FDA completed a reinspection of the Catalent Indiana site with no further requests, clearing the path for an imminent...

Caris Life Sciences Inc (CAI) Q1 2026 Earnings Call Transcript
Caris Life Sciences reported Q1 2026 revenue of $216 million, up 79% year‑over‑year, propelled by an 85% surge in molecular profiling revenue. Gross margin improved to 65% on a GAAP basis, reflecting a 61% rise in clinical average selling price and...
SIGA Technologies Inc (SIGA) Q1 2026 Earnings Call Transcript
SIGA Technologies reported Q1 2026 revenue of roughly $6 million, a pretax loss of $5 million and a cash balance of $146 million with no debt. The company announced a $13 million oral TPOXX order for an Asia‑Pacific customer in Q2 and sealed an...

Insmed Inc (INSM) Q1 2026 Earnings Call Transcript
Insmed reported that its newly launched Brinsupri generated $144.6 million in its first full quarter and now projects at least $1 billion in U.S. revenue for 2026. The company highlighted strong early adoption, with over 11,500 new patients in 2025, 90%+ payer...

Science Spotlight: New Prime Editing Tools for Large DNA Insertions
BioCentury’s website employs a tiered cookie framework that classifies cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Strictly necessary cookies power core services such as authentication, registration, and user‑preference management, while functional cookies enhance site personalization. Marketing and...

An Essential Framework to Navigating Technical Challenges in Contemporary OSD Development
Oral solid dosage (OSD) development faces mounting technical hurdles as biopharma pipelines become richer in poorly soluble compounds, with 70‑90% of new small‑molecule candidates classified as BCS Class II or IV. Early‑risk identification—through comprehensive physicochemical profiling, excipient compatibility testing, and Quality...

First Human Trials Aim to Rejuvenate Immune System by Regenerating the Thymus
The TRIIM‑X trial has begun enrolling participants to test a thymus‑regeneration therapy that could reverse immune aging. Researchers hope the intervention will restore T‑cell production and improve resilience against infection and cancer, marking a shift from measuring immune decline to...

Nanobiotix Secures FDA Acceptance of Protocol Amendment to Accelerate Phase 3 NANORAY-312 Trial
Nanobiotix announced that the U.S. FDA has accepted a protocol amendment for its Phase 3 NANORAY‑312 trial, eliminating the interim analysis and moving to a modified final analysis with fewer events. The change is designed to shorten the study timeline for...

Merck Completes $5.3 B Purchase of Terns, Adding Oral CML Drug TERN‑701
Merck closed a cash tender offer for Terns Pharmaceuticals at $53 a share, valuing the deal at roughly $5.3 billion. The acquisition brings TERN‑701, an oral BCR‑ABL1 inhibitor with FDA Breakthrough Therapy Designation, into Merck’s oncology pipeline and will be recorded...

Caris Life Sciences Launches AI Test to Predict Early and Late Breast Cancer Recurrence
Caris Life Sciences introduced Caris MI Clarity, the first AI-driven test that predicts both early and late distant recurrence in postmenopausal HR‑positive/HER2‑negative, node‑negative breast cancer at diagnosis. Results are returned within three business days, promising faster, more comprehensive risk stratification...

CVS Caremark Updates Formularies to Prioritize Biosimilars, Targeting $0 Out‑of‑Pocket for Members
CVS Caremark announced July 1, 2026 updates to its most common commercial template formularies that will prefer FDA‑approved interchangeable biosimilars over select brand drugs, including Stelara. The changes are designed to deliver $0 out‑of‑pocket costs for many members while preserving clinical quality....

The BioPharm Brief: Precision, Immunotherapy, Expansion
Madrigal Pharmaceuticals is licensing Arrowhead’s RNA‑interference candidate ARO‑PNPLA3 to broaden its precision‑medicine pipeline for metabolic dysfunction‑associated steatohepatitis (MASH). LTZ Therapeutics secured $38 million to push forward myeloid‑engager immunotherapies that activate innate immune cells against cancer. Amgen and Eli Lilly announced major U.S....

IBM, Cleveland Clinic, RIKEN Simulate Record 12,635‑Atom Protein on Quantum Computers
IBM, Cleveland Clinic and Japan’s RIKEN have jointly simulated a 12,635‑atom protein using quantum‑centric supercomputing, a 40‑fold increase over their previous benchmark and a 210‑times accuracy improvement. The work demonstrates that quantum processors can now tackle biologically relevant molecular systems...

AI End‑to‑End Models Will Accelerate BioPharma Innovation
AI + Biopharma @synbiobeta Fireside w/ @AnthropicAI , Mithrl, & @obviousvc Insights: - Anthropic aren't going to make their own science models, they'll be training the same base model everyone uses - Base Science models already well served - Anthropic wants to help...
FDA Clears Regeneron's Otarmeni, First Gene Therapy for Hearing Loss
The U.S. Food and Drug Administration has approved Otarmeni, Regeneron's gene‑therapy drug for OTOF‑related hearing loss, marking the first FDA‑cleared treatment that restores auditory function. The therapy, free for U.S. patients, could affect up to 200,000 people worldwide with this...

Dems Probe Withholding Of Vaccine Safety Studies At CDC, FDA
Democrats on the House Energy & Commerce Committee are pressing HHS Secretary Robert F. Kennedy Jr. for an explanation after a CDC study that found COVID‑19 vaccines cut hospitalizations during the latest respiratory virus season was not released. The committee...

MFN Drug Pricing Proposal Raises Questions Around Access, Innovation, and Commercial Coverage
The White House’s most‑favored‑nation (MFN) drug pricing framework projects roughly $600 billion in savings over the next decade, tying U.S. prices to the second‑lowest net price among eight high‑income reference countries. The plan distinguishes prospective launches, which could slash net prices...

Self-Organizing ‘Pencil Beam’ Laser Could Help Scientists Design Brain-Targeted Therapies
MIT researchers discovered that under high power, chaotic laser light in a multimode fiber can spontaneously self‑organize into a tightly focused “pencil beam.” Using this beam, they captured three‑dimensional images of a human blood‑brain barrier model 25 times faster than...

DNA-Guided CRISPR Flips Gene Editing Script, Opening a New Path for Precise Diagnosis and Antivirals
Researchers at Hong Kong University of Science and Technology have created the first DNA‑guided CRISPR‑Cas12a system that can programmatically target and cleave RNA. The new platform, called SLEUTH, combines the DNA‑guided enzyme with isothermal amplification to achieve attomolar‑level detection of...
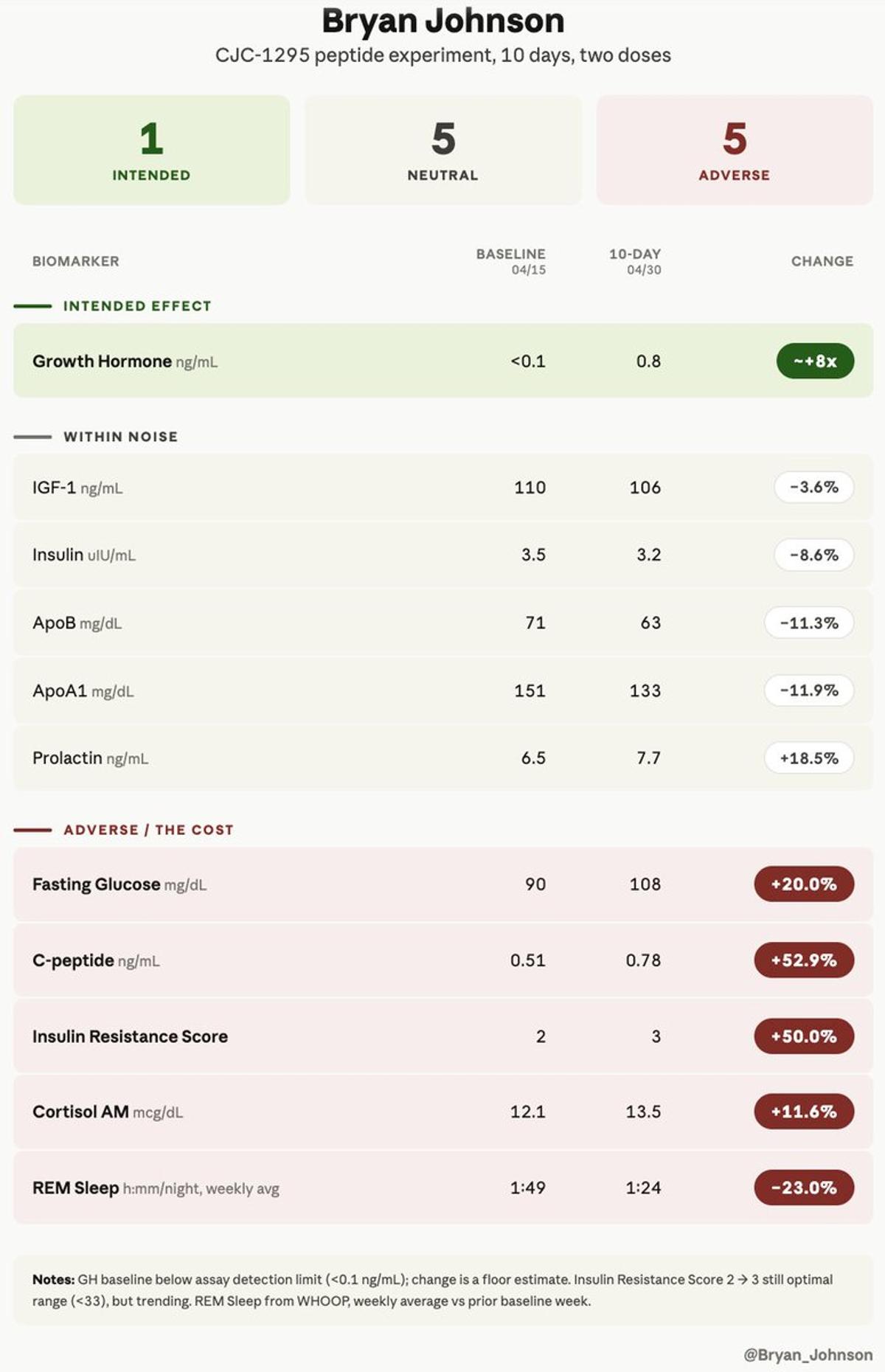
Peptides Boost Hormones, Yet Trigger Serious Metabolic Side Effects
People mistakenly believe peptides are only good. Peptides can be bad, too. They can cause adverse effects. Some dangerous. I did a peptide experiment and measured its effects in my body. The results are complicated. I tried a peptide called CJC-1295....

What’s New in Clinical Trial Innovation
The FDA’s Center for Clinical Trial Innovation (C3TI) publishes a newsletter that highlights new developments, opportunities, and initiatives in clinical‑trial innovation. Readers can subscribe via a public.govdelivery.com form to receive updates directly in their inbox. The site also provides free...

Hamilton Unveils Integrated Glucose Sensor, Marks Decade Milestone
Hamilton Company has been coming to @SynBioBeta for years. This year, they showed up with something new. Tyler Schweder from Hamilton's process analytics team walked Gary George through their full upstream workflow: pH, dissolved oxygen, viable cell density, total cell...
MRNA Flu Shot Cuts Illness in 50+, Raises Reactogenicity
Prasad wanted high-dose flu vaccine comparator - hence the original Refusal-to-File. FDA then agreed to a split application for over-65's and under, and accepted the revised application.
The Human Genome Encodes for a New Category of Molecule
Scientists have identified a previously unrecognized class of molecules encoded within the human genome, arising from short open reading frames once deemed non‑coding. These micro‑proteins, often called micropeptides, exhibit distinct biochemical activity and appear to regulate cellular pathways. Early experimental...

Even a Dog Loves Scaling Virtual Biology Panel
Not all our #SynBioBeta2026 attendees walk on 2 legs 🐶 Taco here found the "From Cells to Patients: Solving the Scale Mismatch in Virtual Biology" Main Stage Panel particularly interesting. https://t.co/6xMHnozSqd

First‑in‑class Molecular Glue Shows Breakthrough Pancreatic Cancer Results
We're seeing major advances vs pancreatic cancer. Today @NEJM data for daraxonrasib @RevMedicines https://t.co/IinceclK6w Context for this first-in-class molecular glue https://t.co/IinceclK6w https://t.co/odhmXQqzMF
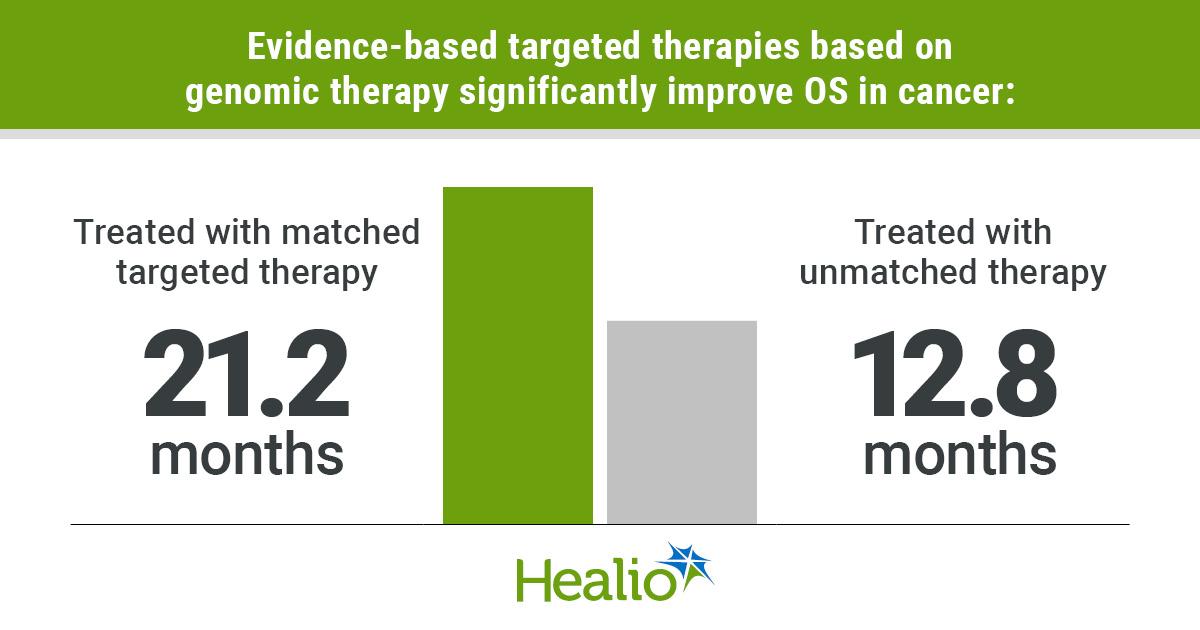
Treating Cancer Based on Mutation Alone Does Not Improve Survival
A large Australian study of 3,383 advanced‑cancer patients found that targeted therapies approved for a specific mutation within the same tumor type improved overall survival by roughly 40%, while using a drug solely because of a shared mutation across different...

Quick Turnaround: Good Longevity Trial News Arrives
I post about working around longevity clinical trials and an hour later @j_n_justice tells me good news about longevity clinical trials... 😍

Trump Drug Strategy Clashes With New Funding Policies
Trump administration’s drug strategy is at odds with recent actions on funding, policy https://t.co/UDiuVnEMDZ via @statnews

Metabolic Stability in Peptide Therapeutics
Peptide therapeutics are gaining traction but remain hampered by poor metabolic stability, limited permeability, and rapid clearance. The article outlines four primary metabolic pathways—hydrolysis, oxidation, reduction, and conjugation—and examines the hurdles of oral delivery, in‑vitro tools, and experimental workflows used...

AstraZeneca’s Camizestrant Hit by FDA Advisory Committee Vote While Truqap Moves Ahead
FDA’s Oncologic Drugs Advisory Committee issued a split verdict on AstraZeneca’s oncology pipeline this week. The committee voted 3‑to‑6 against the benefit‑risk profile of camizestrant combined with a CDK4/6 inhibitor for ESR1‑mutated advanced breast cancer, despite a 56% progression‑free survival...

OECD Blog Item Explores Why Biotech Start-Ups Lag in Europe
The OECD’s May 6 blog highlights that Europe continues to fall behind the United States and Asia in biotech start‑up formation, patenting, and venture‑capital funding. It points to the European Commission’s December 2025 Biotech Act, which introduces regulatory sandboxes, pre‑submission consultations, a...

Bayer Sees Opportunity With $300M Acquisition of Eye Drug Biotech
Bayer announced a $300 million upfront acquisition of Perfuse Therapeutics, a clinical‑stage biotech developing PER‑001, an endothelin‑receptor blocker delivered via a six‑month intravitreal implant. Phase 2 trials showed significant vision improvement in glaucoma and diabetic retinopathy, positioning the drug as a potential...

Pharmaceutical Executive Daily: Zentalis Doses First Patient with Azenosertib in Phase III Trial
Zentalis Pharmaceuticals announced the first patient dosing in the Phase III Aspenova trial of Azenosertib, an oral WEE1 inhibitor for Cyclin E1‑positive, platinum‑resistant ovarian cancer. The study is run with the GOG Foundation, the European Network of Gynecological Oncology Trials (EN‑GOT), and...