Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Lilly, Gilead Lead Pharma’s M&A Boom
Pharma M&A activity surged in early 2026, with 24 deals announced by April and upfront payments topping $64 billion—almost triple the $24.5 billion recorded in the same period last year. The boom is driven by companies racing to refill pipelines before a projected $300 billion loss from patent expirations by 2030, and by cash generated from blockbuster products like Eli Lilly’s GLP‑1 drugs. Oncology attracted the most capital, followed by immune‑modulating and CNS therapies. Major players such as Gilead, Lilly, Merck and Sun Pharma led the spending spree.
High Hs‑CRP, Not LDL, Drives Statin Benefit
"But my cholesterol is normal." JUPITER trial (NEJM): 18,000 people, normal LDL, high hs-CRP. Statin → cardiac events ↓44%, heart attacks ↓54%, strokes ↓48%. 322,000-person UK Biobank: hs-CRP outranked LDL. Full episode 🎙 https://youtu.be/FBLB1CQGBPM

The Rise of Trispecific Antibodies: Biopharma’s Next Big Bet After Bispecifics
Trispecific antibodies are emerging as the next wave of multispecific therapeutics, extending the success of bispecifics by simultaneously engaging three targets. More than 100 candidates are now in clinical trials, with major players such as Pfizer, Sanofi, AbbVie and Johnson...

Treatment-Resistant IBD May Benefit From New Combo Antibody Therapy
Phase 2b DUET‑Crohn’s and DUET‑UC trials, funded by Johnson & Johnson, tested the fixed‑dose co‑antibody JNJ‑4804 (guselkumab + golimumab) in patients whose IBD had failed prior advanced therapies. In ulcerative colitis, JNJ‑4804 matched guselkumab’s efficacy and outperformed golimumab, while in Crohn’s disease the highest dose...

HHS Unveils MAHA Action Plan to Slash Psychiatric Overprescribing
On May 4, 2026, HHS Secretary Robert F. Kennedy Jr. introduced the MAHA Action Plan, a federal strategy aimed at curbing the overprescription of psychiatric medications, especially among children. The plan mandates informed consent, shared decision‑making and a shift toward...

Bayer Reports P-III (REVEAL) Trial Data on Iodine 124 Evuzamitide to Diagnose Cardiac Amyloidosis
Bayer announced that its investigational PET/CT radiotracer I‑124 evuzamitide met the primary sensitivity and specificity endpoints in the Phase III REVEAL trial of 170 adults with suspected cardiac amyloidosis. The study compared the tracer to standard clinical diagnosis and achieved the...

Silence Therapeutics Highlights Recent Business Achievements and Reports First Quarter 2026 Financial Results
Silence Therapeutics reported Q1 2026 results, ending the quarter with $70.1 million in cash and a net loss of $15 million, a sharp improvement from the $28.5 million loss a year earlier. The company highlighted progress on its lead siRNA candidate divesiran, with Phase 2...

With Gilead’s Reasonableness Standard, Side Effects May Vary
The California Supreme Court heard oral arguments in the Gilead Tenofovir case, where plaintiffs seek to impose a novel “duty to commercialize a safer alternative” on drug makers. The proposed duty would require manufacturers to act reasonably in bringing a...

Unlocking Lithium’s Hidden Effects on Alzheimer’s Disease at the Cellular Level
A University of Eastern Finland team mapped lithium chloride’s cellular actions in Alzheimer’s models, showing it reduces Tau hyperphosphorylation at several key sites and reshapes kinase and Rho GTPase signaling. Phosphoproteomic analysis revealed lithium’s impact extends beyond the primary GSK‑3β...

RESEARCH: NITAZOXANIDE in Head and Neck Squamous Cell Carcinoma - 2025 Paper From China
A 2025 Chinese study published in Springer Nature demonstrates that nitazoxanide, an FDA‑approved antiparasitic, exhibits potent anti‑tumor activity against head and neck squamous cell carcinoma (HNSCC). Using integrated single‑cell RNA sequencing and spatial transcriptomics, researchers identified that the drug down‑regulates...

Sanofi Pulls Teplizumab From FDA’s Makary Review Over Political Interference
Sanofi has formally requested that the FDA withdraw its type‑1 diabetes therapy teplizumab from Commissioner Marty Makary’s accelerated review pathway after an acting CDER director, a political appointee, stepped into the scientific review. The move follows a missed April 21 decision...

Eli Lilly’s Mounjaro Becomes World’s Top-Selling Drug
Eli Lilly’s tirzepatide‑based drug Mounjaro has overtaken Merck’s Keytruda as the world’s top‑selling medication, posting $8.7 billion in Q1 2026. Combined with weight‑loss drug Zepbound, the tirzepatide platform generated $36.5 billion in 2025, outpacing Keytruda’s $31.6 billion annual sales. The shift reflects growing demand for...

Mayo Clinic and Stanford Launch First Blood Test to Map Tumor Microenvironment
Mayo Clinic and Stanford Medicine announced a new blood test that maps the tumor microenvironment, enabling more accurate prediction of immunotherapy response across 17 cancer types. The test uses spatial transcriptomics and AI to identify nine cellular neighborhoods from cell‑free...

NIH-Funded Study Suggests that Testosterone Suppresses Brain Tumor Growth in Males
A NIH‑funded study by Cleveland Clinic researchers found that loss of male hormones, especially testosterone, accelerates glioblastoma growth in mouse models by triggering inflammation and the hypothalamus‑pituitary‑adrenal (HPA) stress axis. Supplemental testosterone was associated with a 38% lower risk of...

Magic Mushroom Compound Shows Promise Against Cocaine Addiction
A randomized, double‑blind trial of psilocybin in 40 cocaine‑dependent adults, published in JAMA Network Open, found that 30% of participants receiving a single dose were completely abstinent after 180 days, compared with none in the placebo arm, and remaining users...

Eli Lilly Raises $9 Billion in Record Investment‑Grade Bond Offering to Finance Acquisitions
Eli Lilly & Co. sold $9 billion of investment‑grade bonds in an eight‑tranche offering, the biggest issuance in the company's history. The proceeds are earmarked for a series of planned acquisitions, underscoring the pharma giant's push to expand its pipeline through...
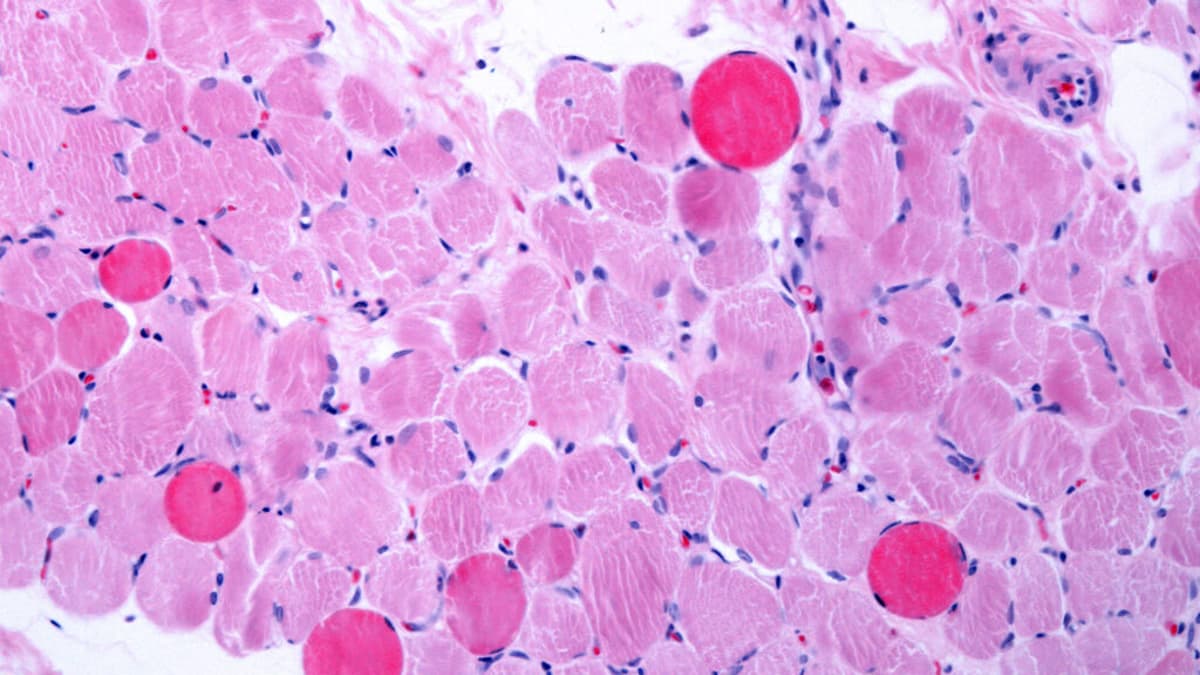
STAT+: Next-Gen Duchenne Drug From Entrada Disappoints
Entrada Therapeutics reported that its next‑generation exon‑skipping drug for Duchenne muscular dystrophy failed to achieve its primary efficacy endpoints in an early‑stage trial. The study showed only a modest rise in dystrophin levels, far below the thresholds set by the...

Entrada Shares Sink After Duchenne Drug Falls Short of Expectations
Entrada Therapeutics reported that its experimental Duchenne drug ENTR-601-44 generated only a 2.36‑point increase in dystrophin, far below the 10‑11% rise analysts had modeled. The shortfall sent the company’s shares tumbling more than 50% in pre‑market trading. By contrast, Avidity...

Angelini Fortifies Neurology Portfolio with $4.1B Buyout of Catalyst
Italian drugmaker Angelini Pharma announced a $4.1 billion acquisition of U.S.-based Catalyst Pharmaceuticals, adding its trio of FDA‑approved treatments for rare neurological disorders to Angelini’s portfolio. The deal brings Ingrezza, Firdapse and a third specialty drug under Angelini’s control, instantly expanding...

Roche to Acquire PathAI for ~$1.05B
Roche has signed a definitive agreement to acquire AI‑driven diagnostics firm PathAI for an estimated $1.05 billion, comprising $750 million upfront and up to $300 million in milestones. The deal, slated to close in the second half of 2026, builds on a partnership...

The Goalposts Shifted in Berlin, Your Trial Timeline Didn’t
The European Society for Medical Oncology (ESMO) Breast 2026 conference in Berlin showcased a wave of late‑stage breast cancer data that is reshaping therapeutic benchmarks. Several trials reported higher pathologic complete response rates and novel biomarker‑driven combinations, tightening the competitive...

Scientists Discover Why Ozempic Works Better for some People
GLP‑1 drugs like Ozempic improve blood sugar and weight loss, but response varies. A Japanese study of 92 type‑2 diabetes patients found that those who overeat in response to external cues (appearance or smell) achieved greater weight loss and glucose...

Encoded Therapeutics Doses First Patient in Pivotal ETX101 Trial for Dravet Syndrome
Encoded Therapeutics has dosed the first patient in the pivotal ENDEAVOR Part 2 study of its AAV9‑based gene regulation therapy ETX101 for SCN1A‑positive Dravet syndrome, while also opening an expansion cohort for older children. The move advances the program into late‑stage...

Will Wegovy Tablets Spark a “Turnaround” At Novo Nordisk?
Novo Nordisk’s newly launched Wegovy oral tablet is delivering a rapid market surge, with over two million U.S. prescriptions and first‑quarter sales of about $350 million, outpacing expectations. Weekly new prescriptions reached roughly 200,000 by mid‑April, marking the fastest uptake for...

Pharma Pulse: GLP-1 Momentum Builds While Lilly Expands Genetic Medicine Manufacturing
Eli Lilly opened its first dedicated genetic‑medicine manufacturing plant in Lebanon, Indiana, expanding U.S. capacity for advanced gene‑editing and RNA therapies. GLP‑1 drugs accounted for eight percent of all prescriptions filled in March 2026, highlighting their growing role beyond diabetes. Bayer...

Strengthening the Generic Drugs Supply Chain
The API Innovation Center published a whitepaper titled “From Fragility to Resilience,” highlighting weaknesses in the U.S. generic‑drug supply chain and proposing data‑driven solutions. The report, authored by VP Natalie de Graaf, identifies the most vulnerable drug classes and quantifies supply‑chain...

What Elements Is the Conversation About Bringing Pharma Manufacturing Home Missing?
President Trump’s 100% tariff on branded drugs aims to force pharmaceutical reshoring, but industry leaders warn the focus on APIs overlooks a critical upstream layer—key starting materials (KSMs). Charlie Lyon of the API Innovation Center notes that 98% of U.S....

The Vaccine Safety Signal the Media Still Won’t Read
A peer‑reviewed study published in Vaccine (Sept 2022) re‑analyzed Pfizer and Moderna mRNA COVID‑19 trial data and found excess serious adverse events—10.1 per 10,000 for Pfizer and 15.1 per 10,000 for Moderna—yielding harm‑to‑benefit ratios of roughly 4.4 : 1 and 2.4 : 1 respectively. The...

What the FDA's Latest Proposal Means for Lilly, Novo, and Hims
The FDA has proposed removing tirzepatide and semaglutide from the 503B bulk‑drug list, effectively ending compounding of these high‑demand GLP‑1 therapies. Eli Lilly, whose tirzepatide‑based products posted 56% YoY revenue growth and a 28% full‑year 2026 outlook, and Novo Nordisk, facing a...

Trump Promised Cheaper Drugs. Some Prices Dropped. Many Others Shot Up.
President Trump’s second term has produced a series of high‑profile drug‑price initiatives, including the TrumpRx discount portal, voluntary "most‑favored‑nation" agreements with manufacturers, and accelerated biosimilar approvals. While a handful of cash‑pay patients see steep cuts on fertility drugs and GLP‑1...

Angelini Pharma to Acquire Catalyst Pharmaceuticals for ~$4.1B
Angelini Pharma announced a definitive agreement to acquire Catalyst Pharmaceuticals for $31.5 per share, valuing the deal at roughly $4.1 billion. The transaction, expected to close in the third quarter of 2026, marks Angelini’s first direct entry into the U.S. pharmaceutical...

What Impact Could Protein Stabilisation Have for Cancer Patients?
In this episode of the DDW Podcast, CEO Anthony Johnson and CSO Matthew Fyfe discuss OutRun Therapeutics' novel protein‑stabilisation platform, which inhibits specific E3 ubiquitin ligases to prevent degradation of disease‑relevant proteins. Their lead program targets the E6AP ligase to...

Halozyme and Oruka Sign Agreement for Hypercon Technology
Halozyme’s Hypercon technology has entered an exclusive global licensing agreement with Oruka Therapeutics to develop ORKA-001, a biologic targeting the IL‑23p19 subunit for psoriasis and related inflammatory diseases. The deal provides Halozyme with an upfront cash payment, future milestone fees,...

Global Drugmakers Eye More China Biotech Deals After Record Year
Global pharmaceutical firms are intensifying their pursuit of Chinese biotech partners after 2025 set a record $135.7 billion in out‑licensing deals, nearly three times the 2024 total. Executives from Johnson & Johnson and Merck highlighted plans to expand on‑the‑ground resources and...
Fibrosis, IPF and the Search for Better Therapies
The BioSpace "Denatured" podcast episode spotlights the persistent challenges of idiopathic pulmonary fibrosis (IPF) and broader fibrotic diseases. Host Jennifer C. Smith‑Parker interviews Georg Vo Beiske, CEO of Tribune Therapeutics, and Jonas Hallén, CMO of Calluna Pharma, to dissect current treatment gaps and...
Macrophage‐Inspired Nanomedicines: Harnessing Innate Biology for Precision Therapy
The review outlines macrophage‑inspired nanomedicines that exploit three delivery platforms—whole macrophage carriers, macrophage‑membrane‑coated nanoparticles, and macrophage‑derived exosomes. By leveraging the cells’ natural homing, barrier‑penetrating and immunomodulatory traits, these systems aim to improve targeting of cancers, atherosclerosis and other inflammatory diseases....

Artera Secures FDA Clearance for AI-Based Breast Cancer Risk Tool
The U.S. Food and Drug Administration granted clearance to ArteraAI Breast, an AI‑driven digital pathology tool that combines digitized histopathology slides with clinical data to generate a risk score for distant metastasis in early‑stage hormone‑receptor‑positive/HER2‑negative breast cancer. The system stratifies...

Trends From the Trenches: The Capability Jump of AI and Its Impact
AI’s rapid capability jump is reshaping computational biology, especially bioinformatics pipelines such as RNA‑seq, single‑cell and spatial transcriptomics. While software firms report most code now AI‑assisted, biotech teams still write code manually, creating a clear adoption gap. Benchmarks—capability, task and...
Oncology Care Ignores Patient Survival vs QoL Preferences
"Patient preference for extending survival or maintaining QoL was not associated with up-front treatment modifications or downstream outcomes, suggesting a possible lack of responsiveness of the current oncology care delivery system to patient preference." https://t.co/0RXf4h2dr3 In: Outcomes of Older Adults With Advanced...

WA Biotech Duo Takes Aim at Diabetes Beyond Ozempic
Australian biotech duo ProGenis Pharmaceuticals and Syngenis Labs are developing RNA‑based antisense therapies that target insulin resistance, a root cause of type‑2 diabetes, to complement GLP‑1 drugs. Syngenis is building Australia’s first GMP‑grade synthetic DNA/RNA manufacturing facility, expected to be...

Japan's TMS-007 Shows Promise for Stroke Treatment
@ej23ny @BiotechXGuru @biotech_cash @Ceazar_Black Anyone here look at Japan Biotech? I'm far from an expert, but I think 4891.T (TMS) is worth looking into. Its a nano cap with dilutive equity warrants (and a smallish cash runway) but their main...

The Latest News in Vaccine Obstruction
Large-scale safety studies of COVID‑19 and shingles vaccines, analyzing millions of records, found rare serious side effects, but the FDA blocked their publication citing unsupported conclusions. The agency also refused to file Moderna’s mRNA flu‑COVID combo vaccine, despite European approval...

PYC Therapeutics Advances PKD Program with Phase 1b Multiple Ascending Dose Study Initiation
PYC Therapeutics has dosed the first patient in a Phase 1b multiple‑ascending‑dose (MAD) study of its PKD candidate PYC‑003, targeting safety, tolerability and early efficacy signals such as urinary PC1 protein, total kidney volume and eGFR. Results from the earlier Phase 1a...

Unfunded $24B Could Have Delivered 100‑Day Prototype Vaccines
Every so often I think about how, in 2022, for $24B we could had "prototype vaccines ready for each of the 26 known viral families that cause human disease" so they can be deployed in 100 days if there was...

Angelini Pharma Acquires Catalyst, Boosts US Rare‑Disease Portfolio
Angelini Pharma buys Catalyst Pharmaceuticals and its rare disease drugs for $4.1B The deal will expand the Italian pharma company’s work in the U.S. https://t.co/BwO6uGKvrT
The Peptide Problem: Hype Is Outrunning the Evidence
Health Canada has warned Canadians against buying and injecting unauthorized peptide products such as BPC‑157, CJC‑1295, ipamorelin, TB‑500 and retatrutide, and has already seized several shipments. Influencers on Instagram and TikTok are promoting these compounds for anti‑aging, weight loss and...

Catalyst Poised as Florida’s First Multi‑Billion Biotech Exit
Is Catalyst (based in Coral Gables) the first Florida biotech to have a multi billion exit?
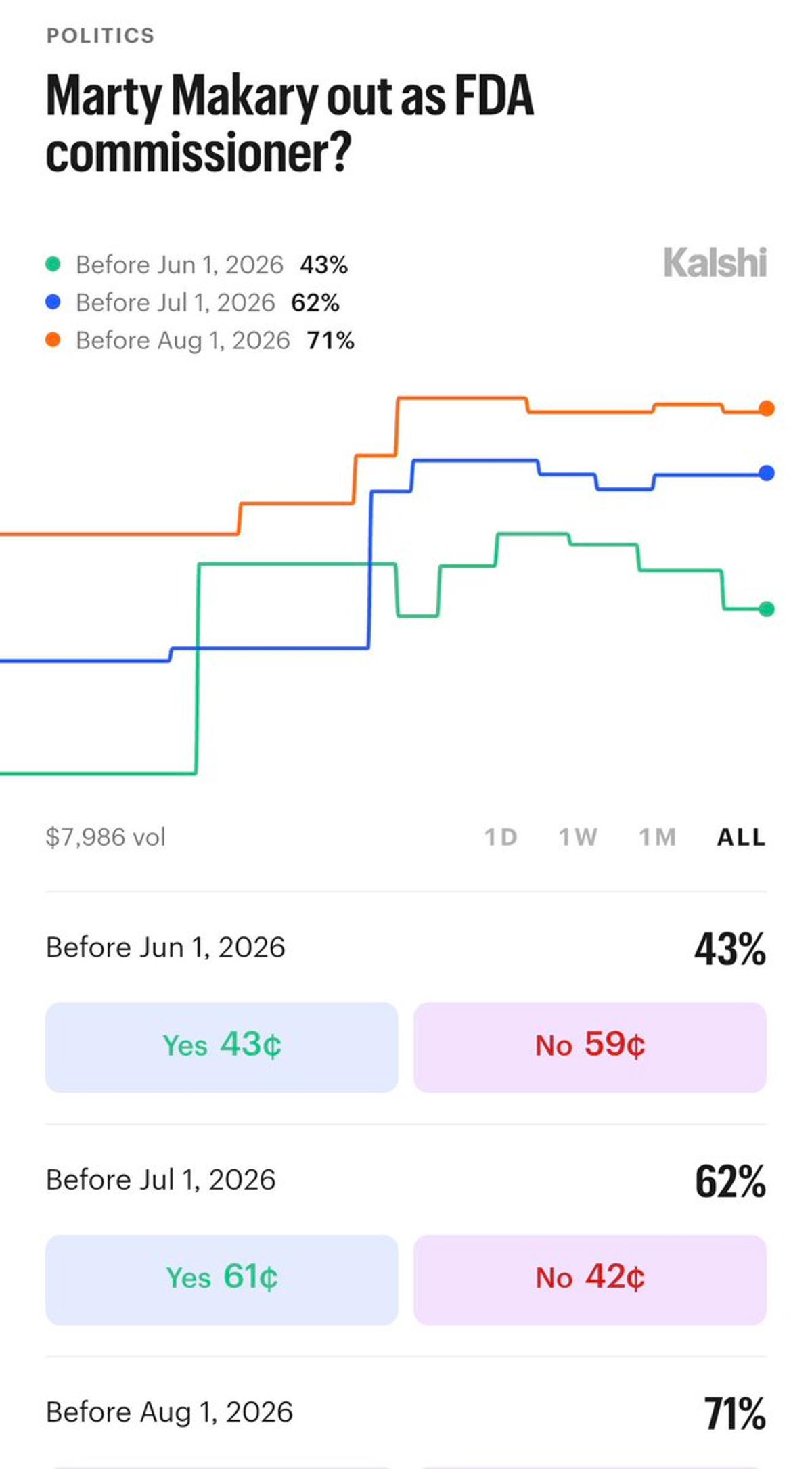
Kalshi Offers Odds on FDA Commissioner Makary’s Tenure
Kalshi now has odds on Commissioner Makary’s term at FDA. Very thin liquidity and wide spreads so I wouldn’t put much credence into these numbers currently but still interesting. https://t.co/rMdpTWB5ES

Did the CDC Improperly Block a Study Showing the COVID Vaccines Were Effective?
The CDC’s acting director, Jay Bhattacharya, delayed and ultimately blocked a COVID‑19 vaccine‑effectiveness paper from being published in the agency’s flagship MMWR journal. The author of the Substack post argues the decision was justified, citing the study’s reliance on a test‑negative...

Targeted Therapies Resurge; some Phase 3 Trials Risk Obsolescence
With a rapid fire raft of trial updates coming out of #esmobreast26, one thing stood out... targeted molecules are back in fashion and a few companies might be left holding the baby if their phase 3 trials have already started: https://t.co/jlKDLsecVZ...