Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech financings
Spanish biotech Ona announced a $86.6 million Series B round, ranking among the largest venture financings in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funds will expand Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical pipeline.

Pulmonary Tuberculosis: Developing Drugs for Treatment
The FDA has issued a final Level 1 guidance titled “Pulmonary Tuberculosis: Developing Drugs for Treatment,” replacing the draft released in December 2022. The guidance, docket FDA‑2013‑D‑1319, outlines clinical development expectations for new antibacterial agents targeting pulmonary TB. It provides sponsors with detailed recommendations on trial design, endpoints, and safety assessments. Stakeholders may submit comments on the guidance at any time through the FDA’s online portal or by mail.

The Peptide Split: How GLP-1s, Lutathera, Vosoritide, and Peptide Cancer Vaccines Are Quietly Rewriting Medicine While BPC-157, TB-500, MOTS-C, and...
Peptide therapeutics are diverging into two distinct trajectories. Clinically validated drugs such as GLP‑1 agonists, Lu‑177 radioligands, vosoritide, and emerging peptide cancer vaccines are delivering hard‑outcome data across cardiometabolic disease, oncology and rare disorders. In parallel, a booming wellness market...

Postapproval Pregnancy Safety Studies
The FDA has released its final Level 1 guidance on post‑approval pregnancy safety studies, updating the 2019 draft to provide detailed recommendations for designing investigations of drug and biologic exposure in pregnant women. The guidance outlines methodological standards, data sources, and...

What Is PDUFA—And Why Does It Matter for Biotech Innovators, FDA & Patients?
Congress will reauthorize the Prescription Drug User Fee Act (PDUFA) next year, renewing the FDA’s authority to collect fees from biotech firms for drug reviews. The user‑fee model supplies a stable budget that underpins faster, more predictable approval timelines—about ten...

The US FDA Grants IND Clearance to Harbour BioMed’s HBM7004 to Initiate P-I Trial in Advanced Solid Tumors
The U.S. Food and Drug Administration has granted IND clearance for Harbour BioMed’s bispecific antibody HBM7004, allowing a Phase I trial in patients with advanced solid tumors. HBM7004 links the tumor‑associated antigen B7H4 to CD3, redirecting T‑cells to the tumor...

Eisai, Biogen Face Delay to Subcutaneous Leqembi
The FDA has extended its review of Eisai and Biogen’s subcutaneous Leqembi Iqlik, moving the decision on its use as a starting dose to August 24. The agency requested a major amendment to the marketing application but has not raised safety...
Gilead Sets Blockbuster Bar for Yeztugo’s First Full Year on the Market
Gilead announced a $1 billion sales target for its twice‑yearly PrEP injection Yeztugo in 2026, up from a prior $800 million outlook. The drug generated $166 million in Q1, a 72% increase from the previous quarter, and 95% of U.S. patients enjoy $0...

FDA Approves the First PROTAC in History, a $1B siRNA Wave Hits Cardiometabolic Disease, and Cytokinetics Cracks Non-Obstructive HCM –...
The FDA granted its first-ever approval for a PROTAC drug, Arvinas’ Veppanu, targeting ESR1‑mutated metastatic breast cancer, marking a regulatory milestone for targeted protein degradation. In parallel, precision‑medicine siRNA deals surged: Madrigal paid $25 million upfront (up to $975 million in milestones)...

Ascendis Retreats to Rare Endocrinology Wheelhouse as Cancer Plans Falter
Ascendis Pharma announced it will halt development of onvapegleukin alfa, its IL‑2 β/γ oncology candidate, after a Phase 1/2 trial showed modest survival benefit in platinum‑resistant ovarian cancer. The company cited a strategic mismatch and will instead concentrate on its rare‑endocrinology franchise,...
Moderna Launches mRNA Hantavirus Vaccine Development in 2024
Did ya know… Moderna began working on an mRNA vaccine for hantavirus in 2024. $MRNA

Capricor Sues Nippon Shinyaku over Duchenne Drug 'Pricing Flaw' And Launch Prep
Capricorn Therapeutics has filed a lawsuit against Japan’s Nippon Shinyaku, alleging a breach of their licensing agreement for the Duchenne muscular dystrophy cell‑therapy candidate deramio‑cel. The complaint centers on a pricing flaw that Capricorn says Nippon Shinyaku misrepresented during launch...

Endometriosis Imaging Study Highlights 99mTc-Maraciclatide as Diagnostic and Monitoring Tool
Serac Healthcare and Oxford’s Nuffield Department have published Phase 2 results of the DETECT study, showing that the gamma‑emitting radiotracer 99mTc‑maraciclatide can non‑invasively locate endometriotic lesions, including superficial peritoneal disease. Imaging findings matched laparoscopy in 16 of 19 participants and identified disease...

AI‑enhanced Dog Breath Test Detects Cancers with 90%+ Accuracy
New MCED / cancer screening test: Dogs smell human breathing + Bayesian AI detects multiple cancers with 90.8% sensitivity & 91.3% specificity (AUC 0.962), even in early stages. Phase II India study (1,502 people, 7 cancer types)- strong potential for affordable...

FDA Reconsiders Ebvallo Cell Therapy After Surprise Rejection
Pierre Fabre Pharmaceuticals and Atara Biotherapeutics secured a meeting with the FDA that reversed the agency’s Jan. 2026 rejection of their EBV‑associated T‑cell therapy, Ebvallo. The regulator now deems the completed single‑arm trial sufficient for a resubmission, opening a path...

Court Rulings Threaten Nationwide Access to Abortion Pill Mifepristone
A 5th U.S. Circuit Court of Appeals decision that blocks the FDA’s 2023 rule allowing mail delivery of mifepristone was temporarily halted by the Supreme Court, leaving the future of the abortion pill’s nationwide availability unclear. Advocates warn the split...

A Light at the End of the Tunnel for Huntington’s Disease Treatment
Researchers at Florida Atlantic University have uncovered a cellular pathway that enables mutant huntingtin protein (mHTT) to travel between neurons via tunneling nanotubes (TNTs). Using LC‑MS/MS, they identified the intracellular pH sensor Slc4a7 as a critical membrane partner of the...

Odyssey Therapeutics Upsizes IPO to 15.5 Million Shares at $18, Raising $304 Million
Odyssey Therapeutics announced an upsized initial public offering of 15.5 million shares at $18 per share, granting underwriters a 30‑day option for an additional 2.33 million shares and completing a concurrent private placement of 1.39 million shares to a TPG Life Sciences affiliate....

Webinar Q&A Follow Up: Immunoassay Signal Amplification: Bold New Solutions for Existing ELISAs
Cavidi’s principal scientist Peter Stenlund explained how the BOLD signal‑amplification platform boosts ELISA sensitivity by lowering the lower limit of quantification while modestly reducing the upper limit. The technology relies on click‑chemistry conjugation of stable DBCO‑modified oligos, offering precise stoichiometry...

Streamline Data Capture and Automate EHR-to-EDC Data Transfer
Paradigm Health introduced eSource Casebook, an EHR‑integrated platform that extracts both structured and unstructured clinical data, auto‑populates electronic case report forms, and streams the information directly to sponsor EDC systems. The tool tackles the industry‑wide surge in manual data entry,...
MEDSIR Reports PHERGain and PHERGain-2 Trial Results for Breast Cancer
MEDSIR presented Phase II data from the PHERGain and PHERGain‑2 trials, exploring chemotherapy‑free strategies for early HER2‑positive breast cancer. PHERGain showed that PET‑guided use of trastuzumab and pertuzumab allowed roughly 30% of patients to omit chemotherapy while achieving nearly 90% five‑year...

Manufacturing Hangover Drives Daiichi Sankyo Into the Red
Daiichi Sankyo announced a fiscal‑year loss of ¥149.4 billion (about $950 million) despite a 12.5% revenue increase to ¥2,123 billion ($13.4 billion). The shortfall stems from costly third‑party manufacturing contracts for its antibody‑drug conjugates (ADCs) and an impairment of ¥19.3 billion ($123 million) after scrapping an...

Venom and Hot Peppers Offer a Key to Killing Resistant Bacteria
Researchers at Mexico’s UNAM have created three new antibiotics from scorpion venom and habanero pepper compounds. Two benzoquinone molecules from Diplocentrus melici venom show activity against tuberculosis, Staphylococcus aureus and Acinetobacter baumannii, while a defensin peptide from Capsicum chinense targets...

ALX Oncology Presents P-I/II Trial Data on Evorpacept Combination in Metastatic Breast Cancer (mBC) at ESMO Breast Cancer’26
ALX Oncology reported exploratory Phase Ib/II data on evorpacept combined with Ziihera in 24 heavily pre‑treated HER2‑positive metastatic breast cancer patients, all of whom had previously received Enhertu. Overall, the regimen achieved a 33% confirmed overall response rate (cORR) with...

Cera and uMed Announce New Partnership
Cera, Europe’s largest HealthTech provider, has partnered with uMed’s automated living registry platform to bring clinical trial recruitment into patients’ homes. The collaboration leverages Cera’s network of over 2.5 million monthly home‑care visits and uMed’s regulated digital consent platform to reach...
Beacon Therapeutics Reports P-II (DAWN) Trial Data on Laru-Zova in X-Linked Retinitis Pigmentosa (XLRP)
Beacon Therapeutics presented 12‑month data from its Phase‑2 DAWN trial of laruparetigene zovaparvovec (laru‑zova) in patients with X‑linked retinitis pigmentosa (XLRP) who previously received an AAV‑RPGR gene therapy. The study showed sustained gains in low‑luminance visual acuity (LLVA) and microperimetry mean...

Dual‑LAO Accelerates Robust Binding Free Energy Calculations
New paper published @CommsChem with the @qubit_pharma team led by @Narjes_Ansari: "Dual-LAO for calculating fast and robust relative binding free energies of simple and complex transformations" (#OpenAccess) #compchem #drugdesign @FefeAviat J. Hénin L. Lagardère https://t.co/ktdX8AWMBo
Semaglutide Race: Torrent Grabs 38% Market Share
Torrent Pharma has seized a 38% share of India’s generic semaglutide market within a month of launch, generating about ₹17 crore ($2 million) in April sales. Generic semaglutide revenue reached ₹44 crore ($5.3 million), while total market sales—including Novo Nordisk’s innovator—hit roughly ₹89 crore ($10.7 million)....

FDA’s Expedited Drug Reviews Are Hailed in some Quarters but Other Approval Practices Are Problematic
In July 2025 the FDA launched the Commissioner’s National Priority Voucher (CNPV) pilot to slash drug review times from six‑to‑eight months down to one‑or‑two months, issuing 17 vouchers for therapies ranging from cancer to cholesterol drugs. Commissioner Marty Makary later...

Trump’s New Drug Advertising Proposals Fall Short on Public Health and the Constitution
The Trump administration’s FY 2027 budget proposes two contrasting measures: a sensible requirement that compounding pharmacies prominently disclose that their products have never been evaluated by the FDA, and a broader effort to give the FDA new authority to deem FDA‑approved...

Study Finds Rapamycin May Undermine Exercise Gains in Older Adults
Researchers led by Dr. Brad Stanfield reported that older adults taking a low weekly dose of rapamycin gained less muscle strength and physical function from a structured exercise program than those on placebo. The findings suggest the popular anti‑aging drug...
Revolution Medicines' Daraxonrasib Doubles Survival in Advanced Pancreatic Cancer
Revolution Medicines announced that its experimental KRAS inhibitor daraxonrasib doubled overall survival for patients with metastatic pancreatic cancer in a Phase 3 trial. The FDA has granted expanded‑access permission, allowing the drug to be used outside trials while regulators review the...

Roche to Acquire PathAI for Up to $1.05 B, Accelerating AI‑Driven Diagnostics
Roche announced a definitive agreement to buy U.S. digital‑pathology specialist PathAI for up to $1.05 billion, with $750 million paid upfront and up to $300 million in milestone payments. The deal, expected to close in the second half of 2026, aims to fuse...

Why Not Do Random Testing in Randomized Trials Designed to Measure Risk of Infection?
The post argues that randomized trials measuring infection risk, such as the recent Moderna flu study, rely on symptom‑driven testing rather than random testing of all participants. This selective approach inflates reported efficacy by omitting mild or asymptomatic cases and...

Armata Pharmaceuticals Wins FDA Fast Track for AP‑SA02 Bacteriophage Therapy
Armata Pharmaceuticals announced that the FDA granted Fast Track designation to its AP‑SA02 bacteriophage candidate for complicated Staphylococcus aureus bacteremia. The status promises more frequent agency interaction and a rolling Biologics License Application review, positioning the drug for accelerated approval...
Screening for Photoreceptor Survival
Researchers used human retinal organoids to screen compounds that affect cone survival under glucose starvation. They identified two kinase inhibitors, CS‑KI‑1 and CS‑KI‑2, targeting CK1 and MAPK11, that protected cones and rods in vitro and in a mouse model of...

Amplia Therapeutics Launches New Narmafotinib Ovarian Cancer Study with ANZGOG
Amplia Therapeutics announced the launch of the PRROSE investigator‑initiated trial, testing its FGFR inhibitor narfotininb together with carboplatin and paclitaxel in high‑grade serous ovarian cancer patients who responded poorly to initial platinum therapy. The study will enroll 15‑20 participants, prioritize...
Finger‑prick Blood Test Accurately Detects Active TB, Predicts Risk
A finger-prick blood test measuring immune response can accurately detect active tuberculosis and moderately predict future disease in household contacts, offering potential for more targeted TB screening and prevention. tuberculosis
Spermidine Halts Liver Fibrosis by Cell Signal Remodeling
Researchers have demonstrated that spermidine, a naturally occurring polyamine, can halt the progression of liver fibrosis by reprogramming cellular signaling pathways. In mouse models, spermidine treatment reduced collagen deposition and restored normal liver architecture within eight weeks. The study identified...
Two Mechanisms Vie to Deliver First Hypoxic Ischemic Encephalopathy Drug
The article is BioCentury’s cookie policy, outlining five cookie categories—strictly necessary, functional, marketing, advertising, and analytics—and describing their purposes for site operation, personalization, outreach, and data collection. It explains how each type works, the data it handles, and the impact...

Astellas Touts Data From Early Test of Stem Cell-Derived Eye Therapy
Astellas Pharma announced early-stage data from its stem cell‑derived retinal therapy, aimed at treating age‑related macular degeneration (AMD). In a small cohort receiving the highest dose, patients showed statistically significant gains in best‑corrected visual acuity and no serious safety signals....

Supreme Court Keeps Abortion Pill Mail Access in Place for Now
The U.S. Supreme Court has left in place lower‑court rulings that allow the abortion pill mifepristone to be mailed while the case proceeds. By refusing emergency relief, the Court maintains the current federal framework for medication abortion. The decision highlights...

Novel Nanoparticle Therapy Using Manganese Could Improve Cancer Treatment
Researchers at the University of Michigan and MD Anderson have engineered a manganese‑based nanoparticle named CRYSTAL that activates the cGAS‑STING immune pathway without provoking systemic inflammation. Preclinical studies across several tumor models, including advanced triple‑negative breast cancer, demonstrated robust, sustained...
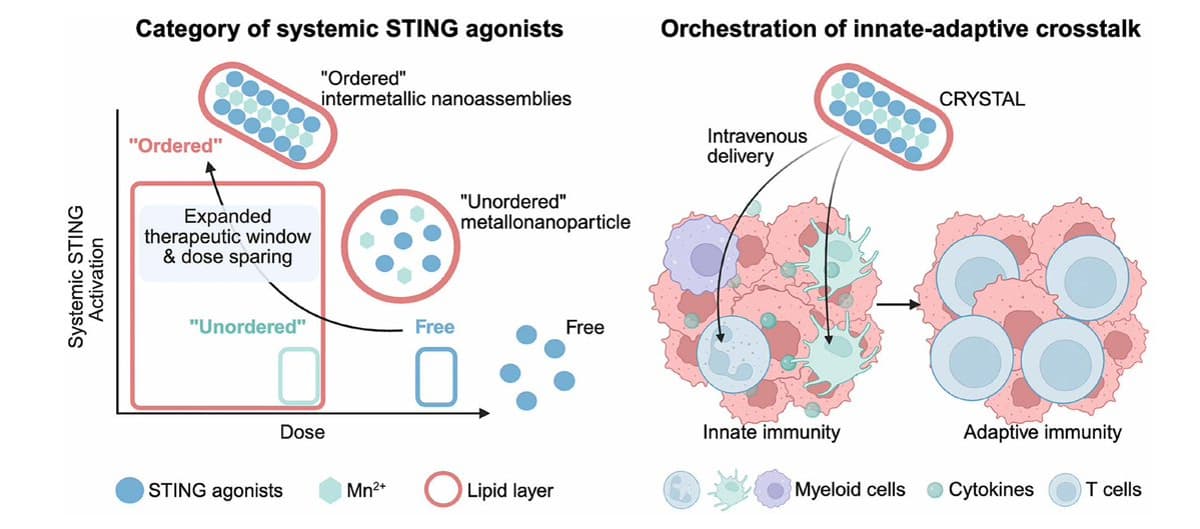
STING Pathway Emerges as New Cancer Immunotherapy Frontier
The number of new potential ways to rev up the immune response to cancer keeps expanding. Today it's STING-ing it. @ScienceMagazine https://t.co/p9g0Mmc8kG https://t.co/LbKfODQkO1

CAPR Sues NS Pharma over Launch Delays, Pricing Disputes
$CAPR is suing NS Pharma, its deramiocel commercial partner. Capricor alleges NS Pharma is not doing enough to prepare for commercial launch + some pricing mishegas. 8K https://t.co/7iFyLRhKsq
G-Link CAR-T Delivery Platform Showcased at ASGCT
Vyriad unveiled its G‑Link CAR‑T delivery platform at the ASGCT meeting, showcasing a modular protein adapter that repurposes existing lentiviral vectors for in‑vivo use. The technology promises to cut development timelines by eliminating extensive vector redesign and to boost transduction...

FDA Approval of DOR/ISL Expands HIV Treatment Options Beyond INSTIs: Amy Colson, MD, MPH
The FDA has approved Idvysno, a two‑drug regimen of doravirine and islatravir (DOR/ISL), marking the first HIV therapy that omits both integrase strand transfer inhibitors (INSTIs) and tenofovir. Clinical trials showed non‑inferior viral suppression at 48 weeks compared with standard...

Rare Disease News, Events & Reports
The FDA’s Center for Drug Evaluation and Research (CDER) continues to expand its Rare Disease Innovation Hub and Accelerating Rare Disease Cures (ARC) program through a steady stream of workshops, public meetings, and reports from 2022 to 2026. Recent highlights...
Single Psilocybin Dose Triggers Month-Long Brain Changes and Mood Boosts
Researchers at the University of California‑San Francisco and Imperial College London reported that a single 25 mg dose of psilocybin produces measurable increases in brain entropy and white‑matter integrity that persist for at least a month, while participants report heightened insight...

With Legal Briefs in, Supreme Court Weighs Telehealth Access for the Abortion Pill
The Supreme Court issued a one‑week stay on the 5th Circuit’s May 1 ruling that would have ended telemedicine prescribing of the abortion pill mifepristone nationwide. The pause keeps the FDA’s 2023 rule—allowing mail‑order and pharmacy dispensing—alive until at least May 11,...

Medicare Launches $50 Copay Pilot for GLP‑1 Weight‑Loss Drugs Starting July 2026
Medicare announced a short‑term pilot that will cover GLP‑1 weight‑loss drugs—including Wegovy, Zepbound and Foundayo—for a flat $50 monthly copay beginning July 1, 2026. The program, run through Dec. 31, 2027, aims to broaden access for beneficiaries with obesity‑related health...