Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

LANL: Scientists Map the Shape of RNA That Can Shut Down Genes
Los Alamos National Laboratory and an international team have mapped the three‑dimensional structure of the SINE B2 ribozyme, a self‑cleaving RNA that acts as a molecular switch in mammalian cells. By integrating X‑ray scattering, biochemical mutagenesis, and biophysical probing with simulations on the Chicoma supercomputer, they revealed how the cleavage site reorganizes the RNA’s architecture to silence genes. The work lays a foundation for AI‑driven modeling of RNA ensembles, feeding into the new “Beyond Alpha Fold” project. Understanding this RNA dynamics could accelerate therapeutic design targeting non‑coding regions of the genome.

Glint of Light in Therapy for Deadly ALS After Decades of Struggle
Researchers reported that tofersen, an antisense oligonucleotide targeting the SOD1 gene, dramatically slowed and even reversed disease progression in a subset of ALS patients with the rare SOD1 mutation. The phase‑III trial, published in JAMA Neurology, showed about a quarter...

We’ve Launched the Most AI‑discovered Longevity Drugs
This is cool. Not sure why you need so much money if you are making drugs faster and cheaper. But it is great - maybe I should be thinking about raising more to scale. At the end of the day,...

Smart Homes Integrated with Biotech Extend Lifespan
A bigger house is not going to give you significantly longer life. But a much smarter house integrated into the advanced biotechnology research and clinical ecosystem will. Very happy to see our chapter with CY Leung published in Springer Nature...
Inclisiran Linked to Lower MACE, AMI Rates in High-Risk ASCVD Population
Inclisiran, added to maximally tolerated statins, lowered 4‑point major adverse cardiovascular events (MACE) from 22.7% to 14.6% in a real‑world ASCVD cohort. The therapy also reduced acute myocardial infarction (5.0% vs 8.5%) and all‑cause hospitalizations (20.8% vs 28.8%) over one...
Billion-Dollar Ideas Lurk in Free PubMed Data
Martin Shkreli came on MFM a while back and told Shaan and I something interesting: PubMed has 40M+ biomedical papers. It's the government database of every medical innovation ever logged. And it's 100% free. He told us if you sit there and read long...

Taking a Look at the Toxicity Trade-Offs of EPCORE FL-1
The phase 3 EPCORE FL‑1 trial added AbbVie/Genmab’s bispecific antibody epcoritamab to the rituximab‑lenalidomide (R²) regimen for relapsed or refractory follicular lymphoma. The triplet produced a 79% reduction in progression‑or‑death risk and lifted 16‑month progression‑free survival to 85.5%, nearly doubling the complete...

Search for Regulatory References | Drugs
The FDA has compiled a searchable reference guide that aggregates key regulatory resources for drug and biologics developers. It includes links to guidance on the BPCI Act’s license provision, Abbreviated New Drug Applications (ANDA), CDER small‑business assistance, and submission address...
STAT+: Capricor Therapeutics Accuses Nippon Shinyaku of Slow-Walking Plans on Duchenne Drug
Capricor Therapeutics has filed a lawsuit in New Jersey alleging that Nippon Shinyaku and its U.S. arm, NS Pharma, are deliberately stalling the U.S. launch of deramiocel, its experimental Duchenne muscular dystrophy therapy. The complaint highlights a pricing formula error...

Mabwell IPO Highlights China’s Rising Biologics Powerhouse
Do you remember the company which sold their IL11 nanobody to Google Calico after it realized that after 13 years in the business they need a drug for aging? The company is called Mabwell and they just listed in Hong...

FDA Grants Seventh Approval Under the National Priority Voucher Pilot Program
The FDA approved Bizengri (zenocutuzumab‑zbco) for adults with advanced, unresectable or metastatic cholangiocarcinoma harboring NRG1 gene fusions, marking the seventh clearance under the Commissioner’s National Priority Voucher (CNPV) pilot. The approval follows a single‑arm trial of 19 patients that showed...

About CDER Small Business and Industry Assistance (SBIA)
The FDA’s Center for Drug Evaluation and Research (CDER) Small Business and Industry Assistance (SBIA) serves as a primary contact point for small pharmaceutical firms seeking regulatory guidance. SBIA provides direct phone and email support, a dedicated web portal, and...

Pharmaceutical Executive Daily: FDA Extends Review of Leqembi's sBLA
The FDA has extended the priority review of Eli Lilly's subcutaneous Leqembi by three months, delaying the agency's action date while requesting additional information. The once‑weekly injection is designed to replace the current infusion regimen for patients with early Alzheimer’s...

Political Shift Boosts Biotech, Trim Long Positions
Macro: political shift lifts biotech risk appetite. Key: report Trump to dismiss FDA chief drove Replimune +21%. Risk: headline volatility & regulatory uncertainty. Insight: trim longs. 📈 — Viktor Kopylov, PhD, CFA. More insights: t.me/si14Kopylov
Electromagnetic Field Activation of Gene Therapy as an Approach to Reprogramming
Researchers have engineered an electromagnetic‑field (EMF)‑responsive DNA element that remotely activates partial cellular reprogramming genes in mice. By cycling EMF exposure, the system triggers the Oct4‑Sox2‑Klf4 cassette without permanent gene integration, extending median lifespan to 108 weeks—about 70 human years....
Harbour BioMed Gains FDA Clearance for First-in-Human Study of B7H4xCD3 Bispecific Antibody HBM7004
Harbour BioMed announced FDA IND clearance to launch a Phase I first‑in‑human study of its bispecific antibody HBM7004, which targets B7H4 and CD3 in advanced solid tumors. The trial will assess safety, tolerability, pharmacokinetics and early anti‑tumor activity across multiple cancer...

FDA Rolls Out 1-Day Assessment Pilot in Bid to Refocus Inspection Resources
The FDA announced a pilot program that conducts one‑day inspectional assessments to better allocate its inspection resources. The initiative aims to streamline oversight by focusing on higher‑risk facilities while providing clearer expectations for manufacturers. Cooley’s regulatory partner Sonia Nath said...

Daiichi Sankyo Posts 'Extraordinary Loss' Of Nearly $1B
Daiichi Sankyo announced it is scrapping its planned antibody‑drug‑conjugate (ADC) manufacturing line, a move that triggered an extraordinary loss of 149.4 billion Japanese yen (approximately $950 million). The loss reflects a write‑down of capital expenditures and R&D investments tied to the abandoned...

Biosimilar Ramp-Up to Drive Growth for Biocon in FY27
Biocon is pivoting from a heavy‑investment phase to a profit‑driven model, banking on new biosimilar launches and scale‑up of recent products to fuel FY27 growth. Biosimilars already accounted for 60% of FY26 revenue, delivering 16% year‑on‑year top‑line growth and a...
Some Gene Therapies No Longer Require Clinical Trials, Thanks to New FDA Rule. Is This Safe, and Who Will It...
The FDA has introduced a "plausible mechanism pathway" that lets developers market experimental gene‑editing therapies for rare, monogenic disorders without completing traditional large‑scale clinical trials. The rule relies on prior safety data for the delivery platform and permits customization of...
Can Existing Flu Shots Help Protect Against Bird Flu?
Researchers from National Taiwan University and the University of South Florida analyzed 35 ferret studies spanning two decades and found that seasonal influenza vaccines containing the neuraminidase N1 protein reduced H5N1‑related mortality by roughly 73%. By contrast, vaccines without N1...

Karolinska Study Finds Daily Peanut Exposure Safely Treats 82% of Toddler Allergies
Researchers at Karolinska Institutet reported that 82% of toddlers who received daily oral peanut immunotherapy could safely eat three and a half peanuts after three years, compared with just 12% in a control group. The three‑year study of 75 children...

Safety Debate Heats Up Over Stem Cell Longevity Treatments
Leading researchers and clinic founders are confronting the safety of stem‑cell therapies marketed for longevity, with experts warning that not all products are genuine stem cells and that regulatory oversight remains limited. The debate underscores a booming market driven by...
FDA Sets 2027 PDUFA Date for Taletrectinib in ROS1‑Positive Lung Cancer
The FDA has accepted Nuvation Bio’s supplemental new drug application for taletrectinib in ROS1‑positive non‑small cell lung cancer and scheduled a PDUFA decision for Jan. 4, 2027. The filing adds 10 months of phase‑2 data showing high response rates and durable disease...

Short‑Term Pain, Long‑Term Gain: TMDX Still Bullish
Just wrapped up a 45 minute call with $TMDX management… lots of good stuff.. obviously there’s some short term pain from margin compression due to bigger investments (R&D, trials, international expansion, etc) but my conviction has not changed and I...

Japan’s FY26 Price Revision Expands G1 Repricing and Drives Price Reduction
Japan’s FY26 National Health Insurance drug price revision took effect in April 2026, lowering average prices by 4.02% across about 15,800 products. The revision expands the G1 repricing rule to all off‑patent medicines and biologics with biosimilar competition, driving price...

Medical Marijuana Removed From Schedule I – Moving Closer to Broadcast and Online Advertising but Concerns Still Remain
The Justice Department and DEA moved FDA‑approved medical marijuana and state‑licensed medical cannabis from Schedule I to Schedule III, creating a limited federal pathway for distribution. Recreational cannabis remains Schedule I, so its advertising stays prohibited. The change allows federally registered dispensaries to...

Bayer Announces Positive Data for Investigational PET Radiotracer
On May 7, 2026 Bayer announced that its investigational PET radiotracer I‑124 evuzamitide achieved positive topline results in the Phase III REVEAL study, meeting primary sensitivity and specificity endpoints for diagnosing cardiac amyloidosis. The multicenter trial evaluated 170 adults at 19 U.S....
A Monocyte‐Targeted Nanoplatform for Phagocytosis Activation and Ferroptosis Inhibition in Intracerebral Hemorrhage
Researchers have engineered a monocyte‑targeted nanoplatform (mPDA@DFO‑CpG‑N1) to accelerate hematoma clearance after intracerebral hemorrhage (ICH). The system combines a high‑affinity aptamer for selective monocyte delivery, a TLR9 agonist that overrides CD47‑SIRPα inhibition, and the iron chelator deferoxamine to block ferroptosis....

FDA Delays Leqembi Decision; Artiva Raises $300M; Pharvaris Prices Offering
The FDA has postponed its decision on the subcutaneous formulation of Leqembi, moving the target date to August 24, delaying a potential launch for Eisai and Biogen’s early‑Alzheimer’s therapy. The shift adds uncertainty to the Alzheimer market, where Leqembi could become...

Kanvas Biosciences Receives New Funding to Develop First-In-Category Microbiome Therapeutic for Improving Maternal, Newborn and Child Health
Kanvas Biosciences announced a new investment from the Gates Foundation to develop the world’s first fully synthetic microbiome therapeutic targeting maternal environmental enteric dysfunction (EED). The product will use a complex, multi‑strain consortium designed with AI‑driven discovery and spatial microbiome...

AI-Guided Labs Are Approaching Full Autonomy
Ginkgo Bioworks is building fully autonomous biopharma labs that combine 70 robots, 90 lab devices and AI‑driven software in an 18,000‑square‑foot Boston facility. Scientists can submit dozens of unique protocols each day, which the system executes without human intervention. In...
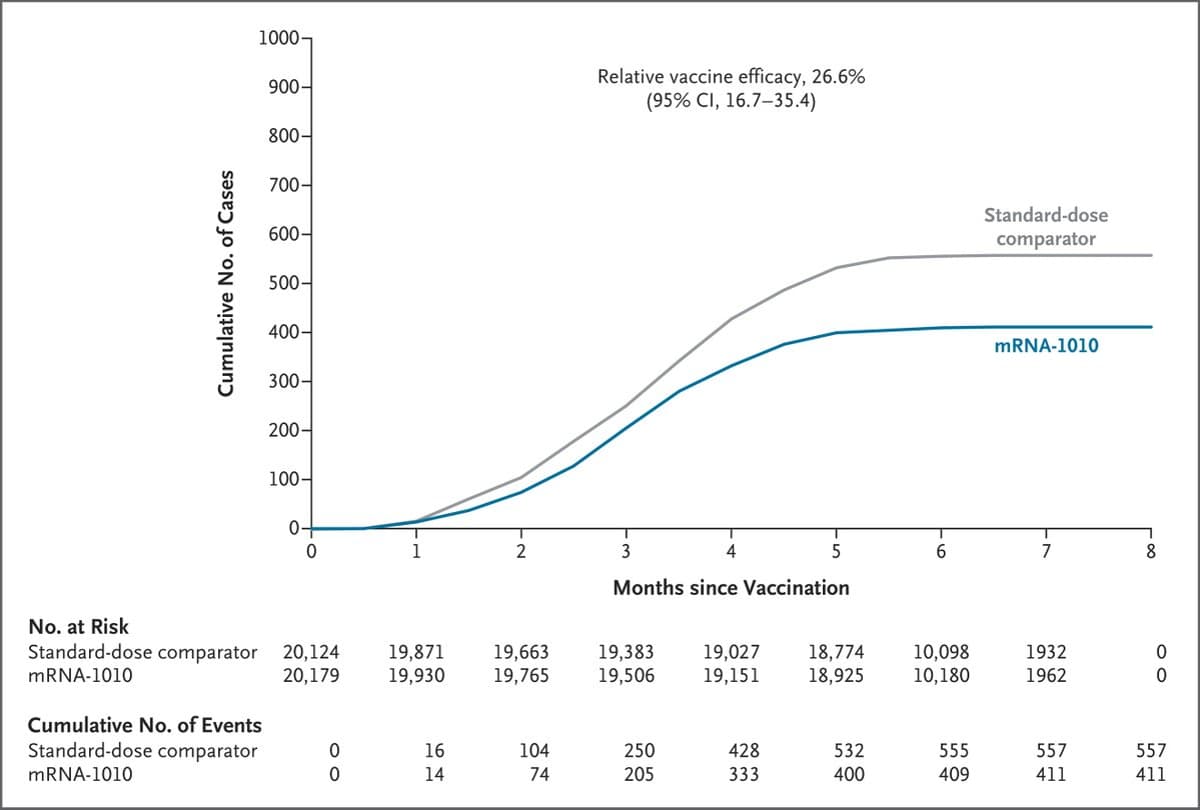
NEJM Publishes Moderna Flu Data After FDA Refusal
A Friday footnote: The New England Journal of Medicine has published the Moderna influenza vaccine data -- the same that led the FDA's CBER division to refuse to review the vaccine under Vinay Prasad. (That decision, called a refuse-to-file or...
Vistagen Announces Completion of Last Patient Visit in PALISADE-4 Phase 3 Public Speaking Challenge Study of Fasedienol for Acute Treatment...
Vistagen announced that the final participant has finished the randomized, double‑blind portion of its Phase 3 PALISADE‑4 trial evaluating fasedienol nasal spray for acute social anxiety disorder. The company expects to release topline efficacy and safety data in the second quarter...
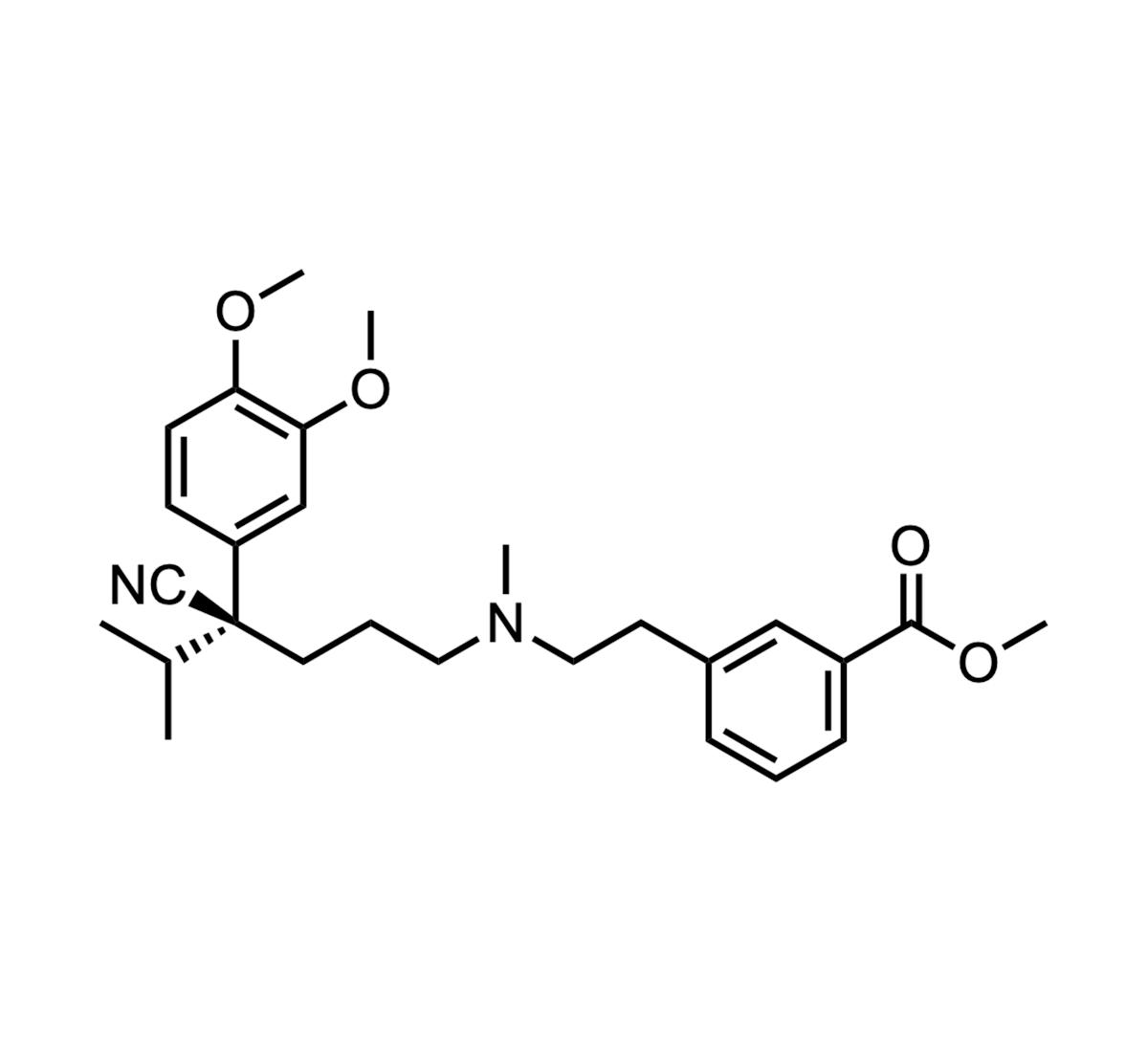
Etripamil
Etripamil (Cardamyst®) received FDA approval as a rapid‑acting, intranasal L‑type calcium channel blocker for converting acute paroxysmal supraventricular tachycardia (PSVT) episodes to sinus rhythm in adults. The drug leverages an ester‑sensitive phenylalkylamine scaffold to achieve fast onset and a short...

ParcelBio Unveils Programmable mRNA Platform Backed by $13M Financing
ParcelBio announced a $13 million seed round led by Breyer Capital, with participation from General Catalyst, Y Combinator and other investors. The funding will accelerate its proprietary Amplified and Prolonged Expression mRNA (APEXm™) platform, which claims to deliver markedly higher and longer‑lasting...
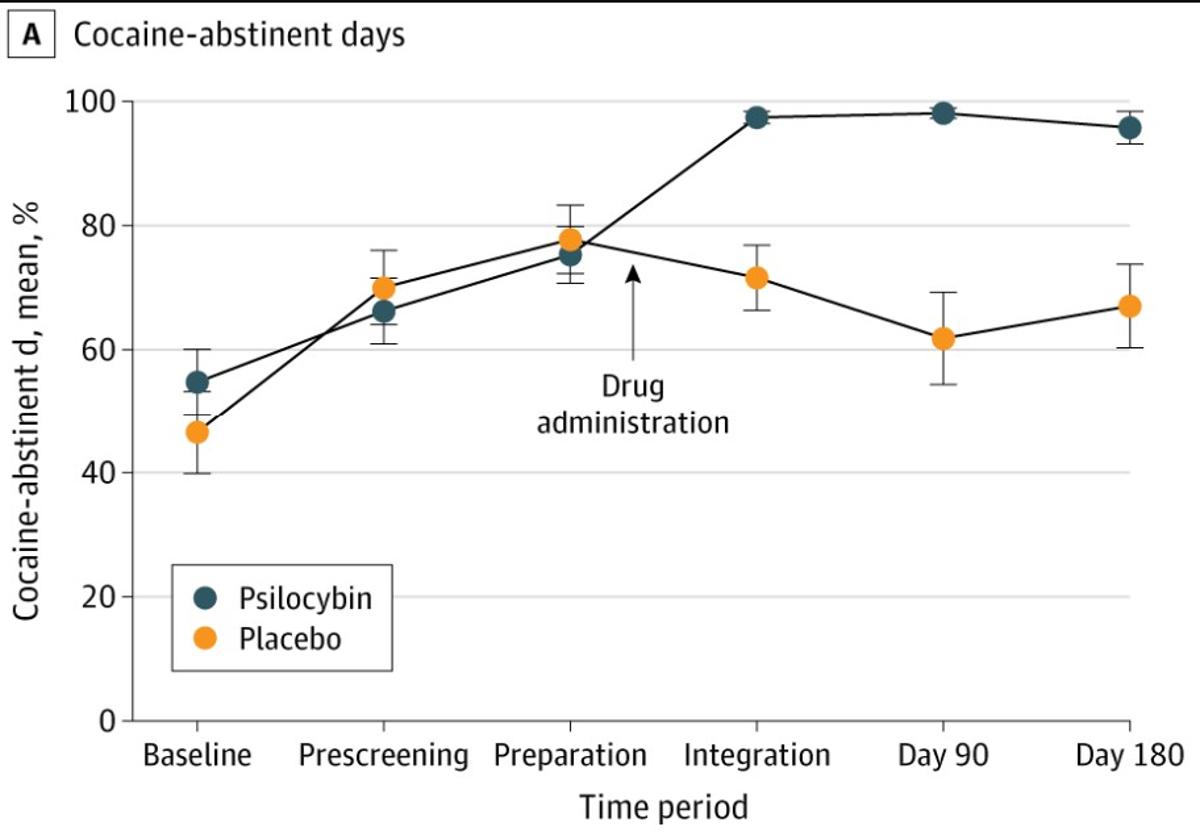
Psilocybin Shows Promise After Decades of Failed Cocaine Treatments
For 50 years the National Institute on Drug Abuse has spent hundreds of millions of dollars to find an effective cocaine addiction medication. After 100+ molecules & many hundreds of studies nothing has been FDA approved. It's the Holy Grail...

Patents Aren't the Magic Key to Drug Development
The $3 billion number is hokey for a number of reasons, but the point is that there is not some magical sauce that allows for drugs to be developed when financed by patent monopolies, but not by upfront funding.

Merck Advances Scalable Manufacturing for Oral PCSK9 Therapy
Merck has published a landmark study in Science describing a scalable biocatalytic synthesis platform for its investigational oral PCSK9 inhibitor, enlicitide decanoate. The enzyme‑driven process enables selective peptide fragment formation, coupling, macrocyclization and uses crystallization‑based purification, aiming to reduce reliance...
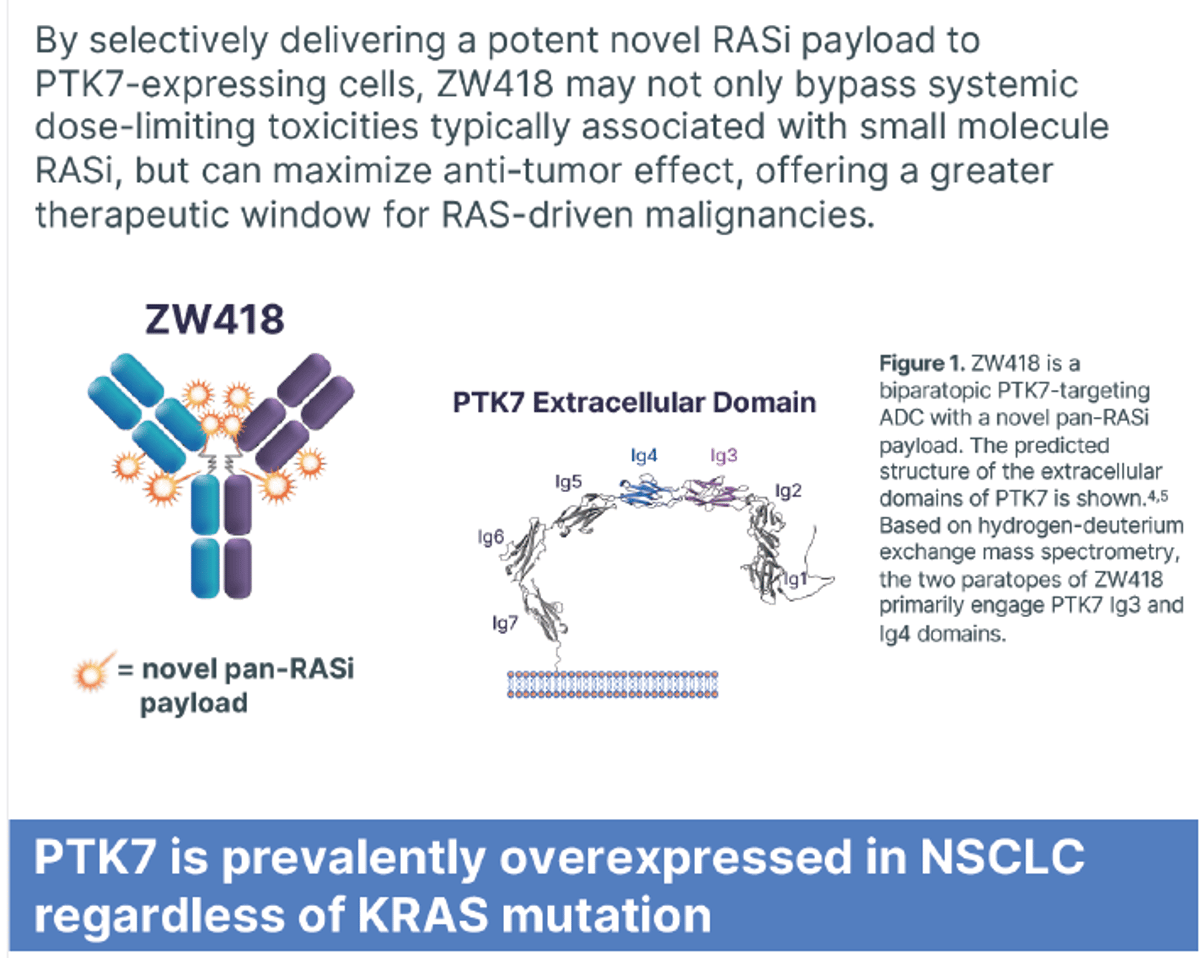
ZYME ADC
Catching up with $ZYME pan-RAS inhibitor payload ADCs from #AACR26. Seems the setting for each one is tumours that are RAS-mutated as well as expressing the target antigen (PTK7, Ly6E or Claudin18.2). https://t.co/hljX5soZQi

PTCT Jumps 17% on Strong Drug Uptake
$PTCT up 17% on results, really liked that one after writing up this week, nice value with strong uptake in its new drug, and those Jan. calls kept accumulating https://t.co/HEe8G2Sjdx

Bristol Myers Squibb Receives European Commission Approval of Sotyktu (Deucravacitinib) for the Treatment of Active Psoriatic Arthritis in Adults
Bristol Myers Squibb announced that the European Commission has approved Sotyktu (deucravacitinib) for adults with active psoriatic arthritis, either as monotherapy or with methotrexate. The approval follows positive Phase 3 POETYK PsA‑1 and POETYK PsA‑2 trials, which demonstrated statistically significant improvements in ACR20...

Communicating Benefit and Risk Information
The FDA’s Division of Drug Information highlighted its dual‑track approach to benefit‑risk communication: direct patient‑oriented counseling and sponsor‑directed guidance for manufacturers. Since 1999, the agency has mandated Medication Guides for drugs with serious adverse‑effect potential, and it enforces balanced risk...

Drug Promotion
The FDA oversees all promotional materials for prescription drugs, ranging from TV spots to website content, while the FTC handles over‑the‑counter products. Companies cannot obtain pre‑approval, but must submit their ads to the agency at the time they go public,...

Angelini Pharma to Acquire Catalyst Pharmaceuticals for $4.1 Billion
Italian drugmaker Angelini Pharma agreed to purchase Florida‑based Catalyst Pharmaceuticals for roughly $4.1 billion in cash. The transaction values Catalyst at $31.50 a share, a 28% premium, and gives Angelini a foothold in the U.S. rare‑disease market and neurology segment.

STAT+: Pharmalittle: We’re Reading About an FDA Gender Rule Change, Its Program for One-Day Inspections, and More
The U.S. Food and Drug Administration announced a proposed rule to strip references to “gender” from its regulations, aligning with a 2025 Trump administration executive order that stresses biological sex. While the agency claims the change won’t affect industry practice,...

Capricor Sues Partner Nippon Shinyaku in DMD Therapy Spat
Capricor Therapeutics has filed a lawsuit against its Japanese partner Nippon Shinyaku and its U.S. subsidiary NS Pharma, alleging a fundamental pricing flaw in their distribution agreement for the Duchenne muscular dystrophy (DMD) cell therapy deramiocel. The biotech claims the...

ASGCT Honors Mohamed Abou‑el‑Enein as Outstanding New Investigator
Mohamed Abou‑el‑Enein, MD, PhD, received the American Society of Gene and Cell Therapy’s 2026 Outstanding New Investigator Award and his lab earned the Best of Molecular Therapy Award. His team’s high‑dimensional spectral flow cytometry platform maps CAR‑T cell states, pinpointing...

Clostridioides Difficile Infection: Developing Drugs for Treatment, Reduction of Recurrence, and Prevention
The U.S. Food and Drug Administration has issued a final Level 1 guidance titled “Clostridioides difficile Infection: Developing Drugs for Treatment, Reduction of Recurrence, or Prevention.” The document replaces the October 2022 draft and provides detailed recommendations for clinical trial design, endpoints,...

GLP‑1 Breakthrough Costs $500/Month, Limiting Access
After catching my breath (unusual for GLP-1 story to discuss their costs approvingly), I suspect both that @continetti (not unusually) is likely directionally right (& I share enthusiasm for free market) - but - at $500/mo for transformative drug -...