Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Field‑Programmable Biofunctional Films: From Assisted Fabrication to Integrated Diagnostic‐Therapeutic Devices
Field‑programmable biofunctional films (FPBFs) are thin‑film platforms that can be programmed to react to a range of physical fields—thermal, mechanical, electrical, optical, magnetic and acoustic. Recent advances in single‑ and multi‑field‑assisted fabrication have expanded their structural tunability and functional density, enabling simultaneous sensing, actuation and drug‑release capabilities. By embedding these responsive layers into medical devices, researchers are creating closed‑loop systems that can diagnose, treat and monitor patients in real time. The review positions FPBFs as a foundational technology for adaptive, precision healthcare.
Australia’s TGA Issues Interim Decision to Tighten Vitamin B6 Supplement Rules
The Therapeutic Goods Administration (TGA) released an interim decision to review and potentially tighten regulations on high‑dose Vitamin B6 (pyridoxine) supplements in Australia. The move follows rising safety concerns over peripheral neuropathy linked to prolonged excessive intake. The decision now...

FDA Expands Vyvgart Approval to All Adult Myasthenia Gravis Types
The U.S. FDA approved a label expansion for Argenx's Vyvgart (efgartigimod) and Vyvgart Hytrulo, now covering all four adult generalized myasthenia gravis (gMG) serotypes. The decision is based on the ADAPT SERON Phase 3 trial, which showed a 3.35‑point improvement in...
Engineered Nanoparticles Tune Cell Density to Accelerate Tissue Repair
Researchers led by Park, Im and Jeong have unveiled a nanoparticle platform that precisely modulates cell density, enhancing cell‑to‑cell and cell‑to‑substrate adhesion and accelerating tissue repair. The work, published in Nature Communications, demonstrates in vitro and in vivo efficacy and...
FDA Launches ‘Plausible Mechanism Pathway’ to Accelerate Ultra‑Rare Gene Therapies
The U.S. Food and Drug Administration announced a new “plausible mechanism pathway” that lets developers deliver patient‑specific gene therapies for ultra‑rare disorders without traditional large‑scale trials. The move promises faster access but has ignited a safety‑versus‑speed debate among ethicists and...
Evaluating the Effectiveness and Safety of Digitalis Glycosides in Treating Heart Failure
A JAMA‑published trial presented at the ESC Heart Failure 2026 Congress found that adding digitalis glycosides to standard therapy lowered the combined risk of cardiovascular death and first worsening heart‑failure event in patients with HFmrEF and HFrEF. The reduction was...

The DASbox® Mini Bioreactor System: Small Scale. Big Results
Eppendorf’s DASbox Mini Bioreactor System lets researchers run up to 24 parallel bioreactors in a footprint of just 7.5 cm per unit. The platform supports 60‑250 mL working volumes, ideal for cell‑screening, media optimization, and early‑stage process development. Integrated DASware software provides QbD‑focused...

The Adiponectin Paradox: Fat’s Secret Longevity Signal or a Bio-Marker of Decline?
Adiponectin, a hormone secreted by fat cells, is celebrated for its anti‑inflammatory and insulin‑sensitizing effects, yet epidemiological data reveal a paradox: while centenarians exhibit high levels, elevated adiponectin in most older adults correlates with higher mortality and frailty. Researchers attribute...

GI Partners Launches Rose BioSolutions After Acquiring Charles River Labs' CDMO and Cell Solutions
GI Partners has completed the purchase of Charles River Laboratories' Contract Development and Manufacturing Organization (CDMO) and Cell Solutions businesses, creating the standalone company Rose BioSolutions. The new firm will offer integrated cell and gene therapy services across four U.S....

Novo's Oral Wegovy Snags 65% of New U.S. Prescriptions, Challenging Lilly's Foundayo
Novo Nordisk reported 1.3 million oral Wegovy prescriptions in Q1, giving it 65% of new U.S. weight‑loss drug orders. Eli Lilly’s oral GLP‑1, Foundayo, has treated just over 20,000 patients, underscoring a fierce rivalry in a market projected to near $100 bn by...
Nanoparticle Formulation Erases 60% of Alzheimer Plaques and Restores Memory in Mice
A collaborative team from Spain and China demonstrated that a specially engineered nanoparticle can eliminate nearly 60% of amyloid‑beta plaques and fully restore memory in Alzheimer’s‑model mice in under an hour. The breakthrough, published in Nature Nanotechnology, targets the blood‑brain...

MoonLake Immunotherapeutics Announces Positive Outcome From Its Final Pre-BLA Meeting with the U.S. FDA and Reports First Quarter 2026 Financial...
MoonLake Immunotherapeutics reported a positive outcome from its final pre‑BLA meeting with the U.S. FDA, securing alignment on the submission plan and label strategy for sonelokimab (SLK) in hidradenitis suppurativa (HS). The FDA accepted MIRA trial data showing a ~43%...

Gilead’s $11.5B Bet: What Two Companies Saw in One Week
Gilead Sciences disclosed $11.5 billion in acquisition charges for three deals, notably Ouro Medicines and Tubulis, on May 7, 2026. In the same week UCB paid $2.2 billion for Candid Therapeutics, a company with an almost identical dual‑target autoimmune platform. Both firms are betting...

Is Testosterone Therapy Safe and Effective? What We Know
A December FDA expert panel advocated expanding testosterone therapy beyond classic hypogonadism, branding it a multibillion‑dollar preventive‑care opportunity. Recent evidence, notably the 5,200‑patient TRAVERSE trial, found no rise in cardiovascular events among high‑risk men receiving therapeutic doses. However, high‑dose use—often...

Gene Editing Has the Science Figured Out and Now Needs an Entire Stack of New Business Models, Reimbursement Mechanics, and...
Gene editing has moved from scientific proof‑of‑concept to commercial reality, highlighted by Vertex's CASGEVY therapy earning $43 million in Q1 2026 and treating over 500 patients at a $2.2 million list price. While the science is solid, the therapy’s multi‑step delivery—spanning screening, cell...

Swiss Real‑World Study Confirms Psychedelic Therapy Cuts Severe Depression
Researchers at Geneva University Hospitals analyzed compassionate‑use data and found that single‑session psychedelic‑assisted psychotherapy with LSD or psilocybin produced rapid, strong reductions in severe depression and anxiety among treatment‑resistant adults. The findings, published in Psychiatry Research, provide the first real‑world...

How a Pill Approved 25 Years Ago Transformed Cancer Treatment
Gleevec, the brand name for imatinib, emerged from Dr. Brian Druker's vision of a targeted cancer therapy that switches off the BCR‑ABL enzyme driving chronic myeloid leukemia. After early‑stage trials showed 100% response with mild side effects, Novartis accelerated the...

ACROBiosystems Overhauls HEK293 Licensing to Cut R&D Delays
ACROBiosystems announced a global upgrade to its HEK293 functional cell‑line licensing, moving from a multi‑step approval process to a purchase‑based model. Effective May 9, 2026, the change grants researchers immediate usage rights for internal research, drug discovery and assay development, cutting compliance...

Common Mistakes Avoided With Expert CMC Regulatory Consulting
Pharmaceutical and biotech firms increasingly turn to expert CMC regulatory consulting to avoid costly missteps that can stall product approvals. Common pitfalls include incomplete documentation, weak manufacturing controls, inadequate stability testing, delayed regulatory planning, and mismatched global requirements. Consultants bring...
Sequential-Release Nanoparticles Eradicate Drug‑Resistant Tumors in Mice
A team led by Prof. Eijiro Miyako at Tohoku University, together with CNRS collaborators, reported on May 6, 2026 that porous amino‑acid nanoparticles delivering a P‑gp inhibitor followed by doxorubicin, plus near‑infrared photothermal heating, eliminated tumors and yielded 100% survival...

Zhejiang Wenda’s NHWD-870 Gains Breakthrough Therapy Designation in China for Rare NUT Carcinoma
Zhejiang Wenda Pharmaceutical’s oral BET inhibitor NHWD-870 has been granted Breakthrough Therapy Designation by China’s NMPA for advanced thoracic NUT carcinoma. The Phase II data showed a 45% objective response rate and median overall survival of 9.33 months, far exceeding the...

Is Novo Nordisk Turning the Page on CagriSema?
Novo Nordisk says its launch timeline for the dual‑agonist CagriSema remains unchanged despite scrapping a single‑chamber delivery device. The Phase 3 trial showed a 23% average weight loss over 84 weeks, impressive but still below Lilly’s tirzepatide at 25.5%. Meanwhile, Novo...

Drug Development Funnel: What I Learnt Building One From Scratch
A new D3.js funnel visualizes drug‑development attrition, showing that the headline billions‑dollar cost reflects cumulative failures rather than a single success. The plot reveals that pre‑clinical spending (~$430 M per approved drug) rivals the cost of late‑stage trials, and that only...

TriSalus (TLSI) Initiates Patient Enrollment for PREDICTT Liver Tumor Clinical Trial
TriSalus Life Sciences announced on May 4 that patient enrollment has begun for the PREDICTT trial, a prospective study of its Pressure‑Enabled Drug Delivery (PEDD) platform in liver tumors. The investigator‑led trial at MD Anderson Cancer Center will enroll about 20...
AtaiBeckley (ATAI) Reports Positive Phase 2a Results for Social Anxiety Treatment EMP-01
AtaiBeckley announced expanded Phase 2a results for EMP‑01, an oral R‑MDMA formulation targeting Social Anxiety Disorder. The study showed a 38% reduction in patient‑reported symptoms and a 32% drop in real‑world avoidance behaviors by Day 43, with 49% of participants classified as...

Partner Therapeutics’ Bizengri Gains FDA Approval and Fast‑Track Voucher for Rare Bile‑Duct Cancer
Partner Therapeutics announced that its antibody Bizengri (zenocutuzumab‑zbco) has been approved by the U.S. FDA for adults with advanced NRG1‑fusion cholangiocarcinoma. The agency also granted a fast‑track voucher, signaling regulatory confidence in the drug’s potential to address an unmet oncology...

Ozempic Mainly Reduces Lean Mass, Not Skeletal Muscle
How much “muscle” are people actually losing on Ozempic and other GLP-1 drugs? The first thing to understand: Lean mass is not the same thing as skeletal muscle mass. That distinction matters.

FDA Extends Review of Eisai/Biogen Subcutaneous Leqembi to Aug 2026
Eisai and Biogen announced that the FDA has added three months to its review of a supplemental biologics license application for weekly subcutaneous Leqembi IQLIK, moving the action date to Aug. 24, 2026. The agency cited a major amendment and a...

BridgeBio Q1 2026 Earnings Show Atruvio Revenue Surge to $180.6M
BridgeBio posted Q1 2026 revenue of $194.5 million, powered by Atruvio net product revenue of $180.6 million—a 24% sequential rise and 392% year‑over‑year increase. The company also announced a $500 million share‑repurchase program and a cash balance of $940.2 million, underscoring a stronger financial...

Real World Outcomes Support the Benefits of Psychedelic Therapy for Severe Depression
A Swiss compassionate‑use program evaluated psychedelic‑assisted psychotherapy using either 100 µg LSD or 25 mg psilocybin in adults with treatment‑resistant depression or anxiety. More than a third of participants reported at least a 50% reduction in depressive symptoms within three months, and...

Upcoming Meeting to Shape Gerotherapeutic Regulatory Frameworks
Affecting the Aging Trajectory: Regulatory Constructs for Gerotherapeutic Drug, Biologic & Device Development 👉 “On May 27, 2026, the Reagan-Udall Foundation for the FDA, in collaboration with @ARPA_H and the @xprize Foundation, will convene a hybrid public meeting on exploring...
Glucose Nanoparticles Help CBD Cross the Blood-Brain Barrier
Researchers have engineered glucose‑coated polymer nanoparticles that dramatically improve cannabidiol (CBD) delivery across the blood‑brain barrier. The particles use a PEG‑PHB core to solubilize CBD and a surface glucose layer to hijack GLUT‑1 transport, while reactive‑oxygen‑species triggers release in inflamed...

Guardant Health Q1 Revenue Surges 48% YoY to $302M, Fastest Growth in Five Years
Guardant Health posted Q1 2026 revenue of $302 million, a 48% year‑over‑year increase and the strongest growth rate in five years. The biotech’s oncology and screening divisions drove the surge, prompting the company to lift its full‑year revenue outlook to $1.30‑$1.32 billion.

Magnesium Nanocoating Promises Safer, Self‑Absorbing Medical Implants
Scientists from the University of the Sunshine Coast, China’s First People’s Hospital and the University of Tokyo unveiled a magnesium‑based nanocoating that adds antibacterial activity and improves biocomibility of degradable implants. The breakthrough could eliminate the need for removal surgery,...

Ginkgo Bioworks Q1 Shows 49% Revenue Drop but Strong B2B Platform Demand
Ginkgo Bioworks posted Q1 2026 revenue of $19 million, down 49% year‑over‑year, but emphasized growing demand for its synthetic‑biology platforms from industrial customers. The company’s Nebula autonomous labs, new cloud‑based sales channels, and a $47 million federal contract illustrate accelerating B2B adoption.

CRISPR Base Editing Provides Irreversible Microbial Biocontainment
A CRISPR-dCas9 base editing system now enables irreversible shutdown of essential genes in engineered microbes, offering a robust biocontainment strategy that minimizes escape and enhances biosafety for industrial and therapeutic applications. biotechnology

AlphaFold Predicted Billions of Proteins in Just One Month
Still incredible that the DeepMind documentary has footage of exact moment Demis is told that AlphaFold can “easily” predict all known (1-2B) protein sequences “in a month” and he says to do it. Then, it shows the moment AlphaFold is...
The Emerging Cancer Treatment That’s Exciting Scientists: ‘We’ve Just Scratched the Surface on What’s Possible’
CAR T-cell therapy, a genetically engineered immunotherapy, is gaining attention after Australian actor Sam Neill announced remission from stage‑three cancer following a clinical trial. Australia has approved four CAR T products since 2018, all targeting blood cancers, while researchers push the...
AI's Real-World Impact in Pharma and Medicine
Just out-latest @timmermanreport post: AI in Practice: on the important, difficult work of getting AI to deliver-in pharma, medicine, & beyond. https://t.co/fuwuxubhrb Cited: @Loftus, @oziadias, @DeanKateBaicker, @EricTopol, @patricksmalone, @arjunmanrai, @zakkohane, @WSJBooks

CRISPR Safeguard Changes How Engineered Microbes Can Be Controlled
Researchers have developed an irreversible CRISPR‑dCas9 base‑editing biocontainment system that disables essential genes in engineered microbes without causing DNA double‑strand breaks. By targeting the start codons of multiple essential genes, the platform permanently halts cell viability and dramatically lowers escape...

Modern Tech's Black Boxes: GLP‑1s and AI
Listening to Ezra Klein & Julie Belluz talk about the black box that obscures our understanding of GLP-1s, you can't help thinking of the black box that obscures our understanding of AI. Feels like the modern condition: magical technologies in...

Rznomics Secures FDA RMAT Designation for RZ-001 Liver Cancer Therapy
South Korean biotech Rznomics announced that the U.S. FDA granted Regenerative Medicine Advanced Therapy (RMAT) designation to its RNA‑editing candidate RZ‑001 for hepatocellular carcinoma. The designation, based on promising Phase 1b/2a data, speeds regulatory interaction and opens priority review pathways.

Medicare Launches $50‑per‑month GLP‑1 Pilot, Reshaping Weight‑loss Drug Coverage
Medicare will offer a $50 monthly copayment for select GLP‑1 weight‑loss drugs in a pilot program beginning July 2026. The short‑term bridge, running through Dec. 2027, targets beneficiaries with BMI 27+ and related health conditions, potentially shifting cost dynamics for insurers...

India’s Village Biotech Hub Unveils First Indigenous Bioreactor, Boosting Rural Innovation
Dyna Biotech, based in Phursungi village on Pune’s outskirts, launched India’s first indigenous bioreactor, marking a shift of high‑tech biotech into rural areas. The facility also rolled out a waste‑to‑energy system that claims five‑fold efficiency gains, positioning the hub as...
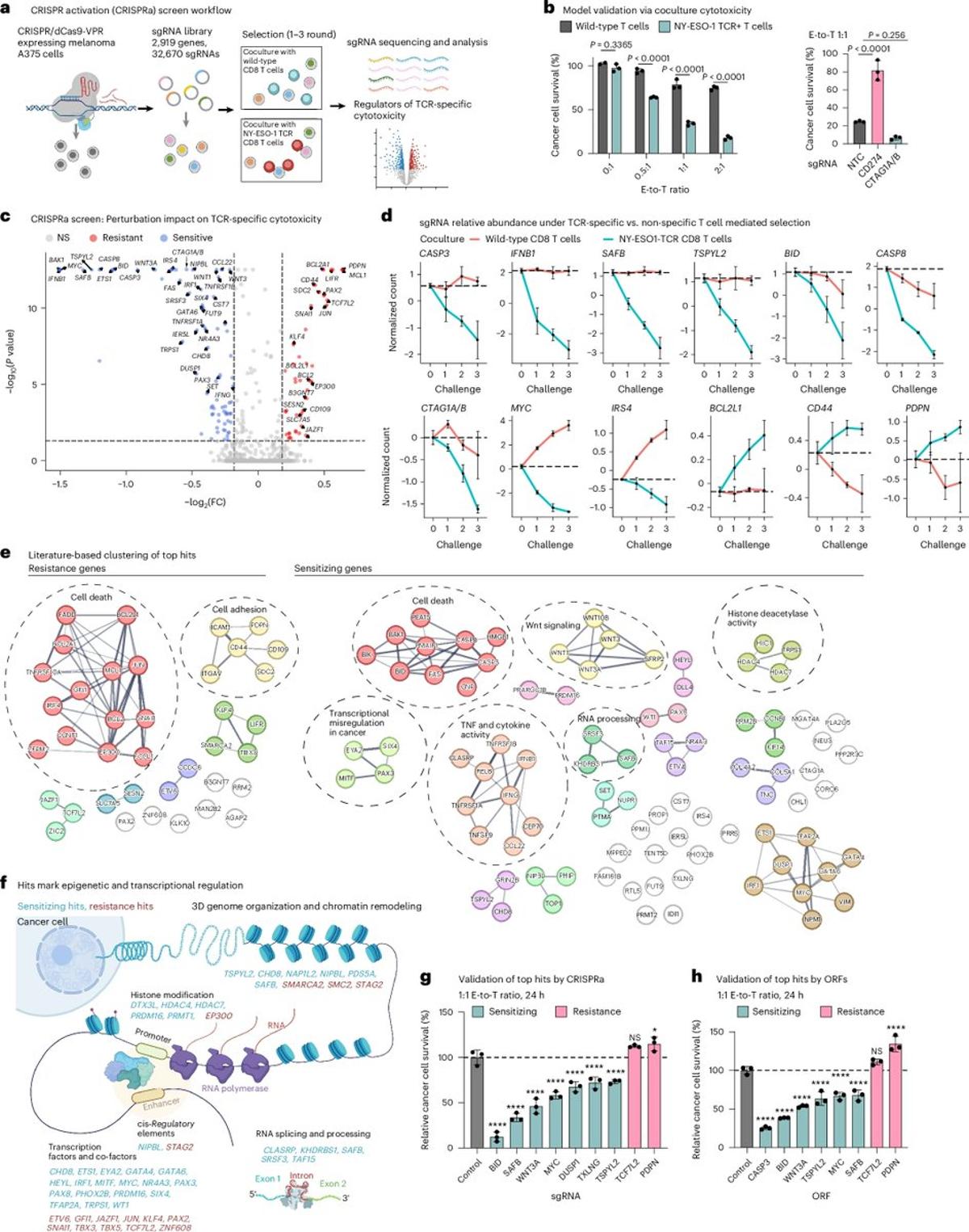
CRISPR Screens Reveal RNA Targets that Boost T‑cell Killing
High-content CRISPR activation screens identify synthetically lethal RNA-based mechanisms to sensitize cancer cells to targeted T cell cytotoxicity https://t.co/gi3hEvoi9V https://t.co/njFRebLRVo

Isomorphic Labs Eyes $2B Raise, Biotech’s Second‑largest
Great scoop from @RebeccaTorrenc5 & @mhbergen on Isomorphic Labs in "advanced discussions to raise more than $2 billion in a new round of funding" led by Thrive Capital Would be second-largest VC raise ever in biotech, only behind $3B for...

RTMS Cuts Smoking by 11 Cigarettes Daily by Boosting Willpower, Study Finds
Researchers at the MUSC Hollings Cancer Center demonstrated that high‑frequency repetitive transcranial magnetic stimulation (rTMS) of the dorsolateral prefrontal cortex lowered cigarette consumption by an average of 11 cigarettes per day, outperforming reward‑targeting and sham treatments. The findings suggest a...
Argenx Secures FDA Nod to Expand VYVGART to All Adult gMG Patients
argenx announced that the U.S. FDA has approved VYVGART and VYVGART Hytrulo for use in every adult with generalized myasthenia gravis (gMG), regardless of antibody status. The decision follows the Phase 3 ADAPT SERON trial, which demonstrated rapid, sustained symptom improvement across...

McKesson Posts 18% EPS Rise, Boosts FY2026 Guidance on Strong Pharma Demand
McKesson Corp. announced adjusted earnings of $39.11 per diluted share for fiscal 2026, an 18% jump, and raised its FY2027 guidance to $43.80‑$44.60 per share. The health‑care distributor credited specialty drug demand, AI‑driven cost cuts, and a $5.1 billion shareholder return...
Mabwell Secures FDA IND Clearance for 9MW5211 Antibody, Paving Way for IBD Trials
Mabwell (688062.SH, 02493.HK) announced that the FDA has cleared its investigational new drug application for 9MW5211, a first‑in‑class antibody targeting pathogenic immune cells in inflammatory bowel disease. The clearance enables the company to begin human trials and adds momentum to...