Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Aspen Neuroscience Begins First Personalized Brain‑Repair Trial for Parkinson’s
Aspen Neuroscience announced the start of its ASPIRO Phase 1/2a trial, the first personalized brain‑repair therapy for Parkinson’s disease. Eight patients received autologous dopamine‑producing cells, reporting roughly two extra hours of “Good ON” time per day. The trial marks a shift from symptom‑management to cellular replacement in neurodegeneration.

Foundayo’s Liver Failure Blip Weighs Down Lilly Shares but Analysts Unconcerned
A single hepatic failure case linked to Eli Lilly’s new weight‑loss pill Foundayo appeared in the FDA’s adverse‑event database, prompting a brief sell‑off that pushed the stock down about 3% before rebounding to close up 0.48%. Lilly quickly investigated and...

CareFusion 213, LLC - 722729 - 04/30/2026
The FDA issued Warning Letter 320‑26‑72 to CareFusion 213, LLC, a BD subsidiary, citing extensive CGMP violations at its El Paso sterile drug facility. Inspectors documented over 2,500 customer complaints involving foreign particles, missing components, and compromised seals, and found the company’s investigations,...

Active Cosmetics Manufacturing Inc. - 722408 - 04/22/2026
The FDA issued a warning letter to Active Cosmetics Manufacturing Inc. after an October 2025 inspection uncovered multiple Current Good Manufacturing Practice (CGMP) violations. The firm failed to conduct thorough investigations of out‑of‑specification microbial results, used an inadequately validated rapid...

Intelligent Remedies, Inc. - 681941 - 01/23/2026
The FDA issued a warning letter (CMS #681941) to Intelligent Remedies, Inc., alleging that ten of its products—including Pryenda, Athrombosyn, and VIRAsol—are marketed with therapeutic claims that classify them as new drugs. The agency says the claims violate the Federal Food,...

Respilon Production S.R.O. - 719705 - 04/20/2026
The U.S. Food and Drug Administration issued Warning Letter 320‑26‑69 to Respilon Production S.R.O., a Czech over‑the‑counter drug manufacturer, citing multiple Current Good Manufacturing Practice (CGMP) violations. The FDA found failures in identity testing of components, lack of stability studies,...

Ray’s Vitamins - 726694 - 04/24/2026
The FDA issued a warning letter to Ray’s Vitamins after discovering that its product “Yeicob Ácido Hialurónico” contains undeclared diclofenac and dexamethasone, classifying it as an unapproved new drug and a misbranded drug. The agency cited violations of sections 301(d), 505(a) and...

Twist Bioscience Q2 2026 Loss Widens as Synthetic DNA Demand Slows
Twist Bioscience Corp posted a second‑quarter 2026 net loss of $44.0 million, or $0.71 per share, compared with a $39.3 million loss a year ago. Revenue rose 19.3% to $110.7 million, but the widening loss highlights pressure on the synthetic‑DNA market as demand...

Sun Pharma to Acquire Organon for $12 Billion, Its Largest U.S. Deal Yet
Sun Pharmaceutical Industries Ltd. announced a $12 billion all‑cash acquisition of New Jersey‑based Organon & Co., the biggest U.S. outbound deal for an Indian drugmaker this year. The transaction pushes Sun Pharma into the specialty‑medicine arena and tests its ability to...
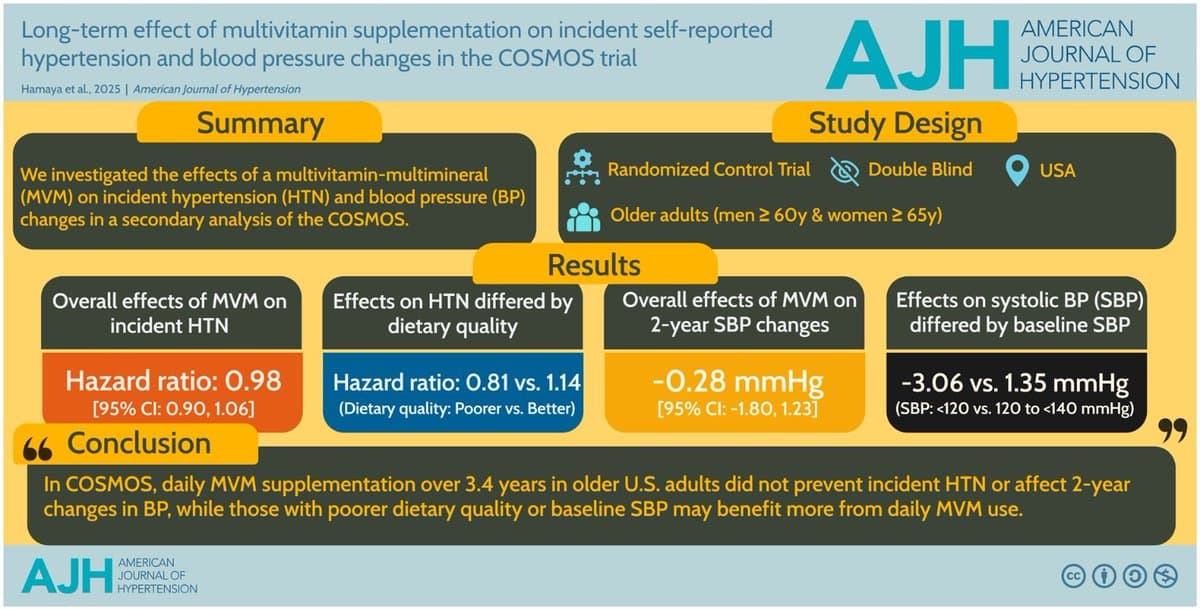
Multivitamins Show No Overall BP Benefit, Help Low‑Diet Individuals
Long-Term Effect of Multivitamin Supplementation on Incident Self-Reported Hypertension and Blood Pressure Changes in the COSMOS Trial "MVM supplementation versus placebo did not reduce hypertension incidence or lower BP overall. Exploratory analyses showed greater reduction in hypertension risk and BP changes...

Insulet Launches Pivotal Trial of Fully Closed‑Loop Insulin System for Type 2 Diabetes
Insulet Corp. has begun a pivotal clinical trial, called Evolve, enrolling up to 350 adults with Type 2 diabetes across 40 U.S. sites. The study tests the company’s first fully closed‑loop insulin delivery system, a move intended to broaden automated insulin...

STAT+: Pharmalittle: We’re Reading About RFK Jr. Targeting Antidepressants, J&J Pushing an IBD Drug, and More
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. announced a new HHS initiative aimed at curbing the widespread prescription of selective serotonin reuptake inhibitors (SSRIs), which currently treat roughly 16.7% of American adults. The plan introduces clinician training,...
Phase 3 FUZION Data Show Guselkumab Benefit in Perianal Fistulizing Crohn Disease
Late‑breaking Phase 3 FUZION data presented at DDW 2026 show that guselkumab significantly improves combined fistula remission in adults with perianal fistulizing Crohn disease. At 24 weeks, remission rates were 28.3% with 100 mg every eight weeks and 27.0% with 200 mg every four weeks,...
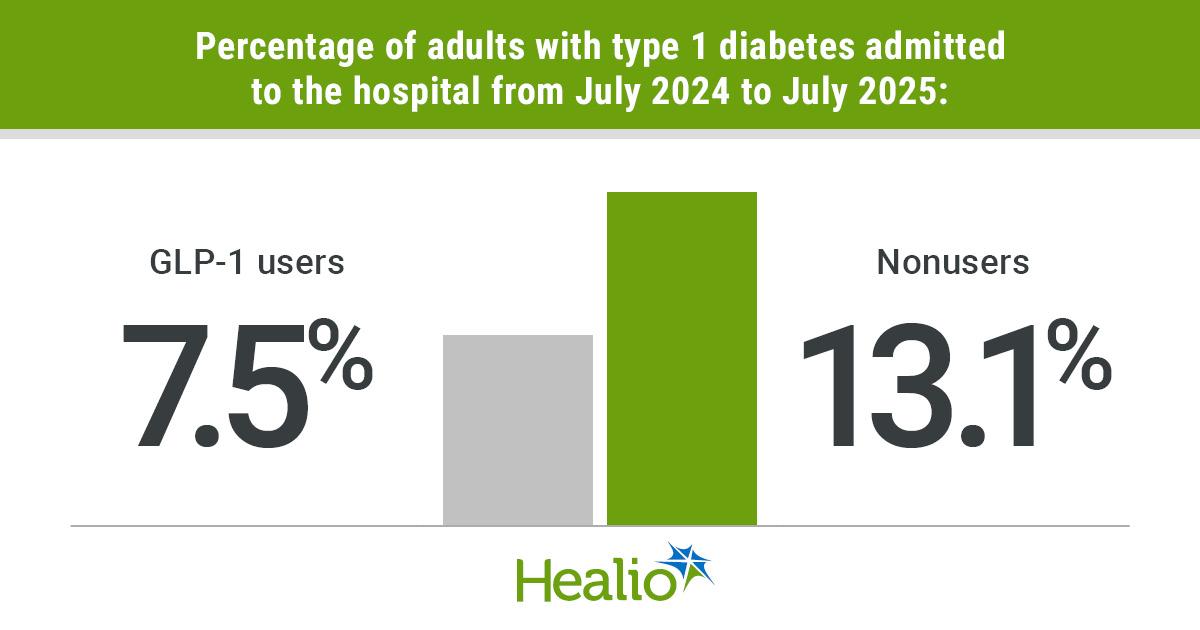
GLP-1s May Not Raise DKA, Pancreatitis Risk in Type 1 Diabetes
A single‑center study of 7,377 adults with type 1 diabetes found that none of the 255 patients using GLP‑1 receptor agonists were hospitalized for diabetic ketoacidosis (DKA) or pancreatitis over a one‑year period. Overall hospital admission rates were significantly lower for...

Disc Medicine Reports First Quarter 2026 Financial Results and Provides Business Update
Disc Medicine reported Q1 2026 results, highlighting the completion of enrollment in its Phase 3 APOLLO trial of bitopertin for erythropoietic protoporphyria, with topline data expected in Q4 2026. The company also announced that Phase 2 data for DISC‑0974 in myelofibrosis‑related anemia will be...

Genetic Variant Determines Individual GLP‑1 Drug Response
As a medical school professor, I get asked why GLP-1 drugs work miracles for some and barely budge for others. A new Nature paper from 23andMe gives part of the answer: your genes. Researchers ran a genome-wide association study in 27,885...

Odyssey Therapeutics (ODTX) IPO Deck
Odyssey Therapeutics, a clinical‑stage biopharma specializing in precision medicines for autoimmune and inflammatory disorders, unveiled its initial public offering deck in May 2026. The company aims to raise capital to advance its Phase 2‑tested pipeline, which targets conditions such as rheumatoid...
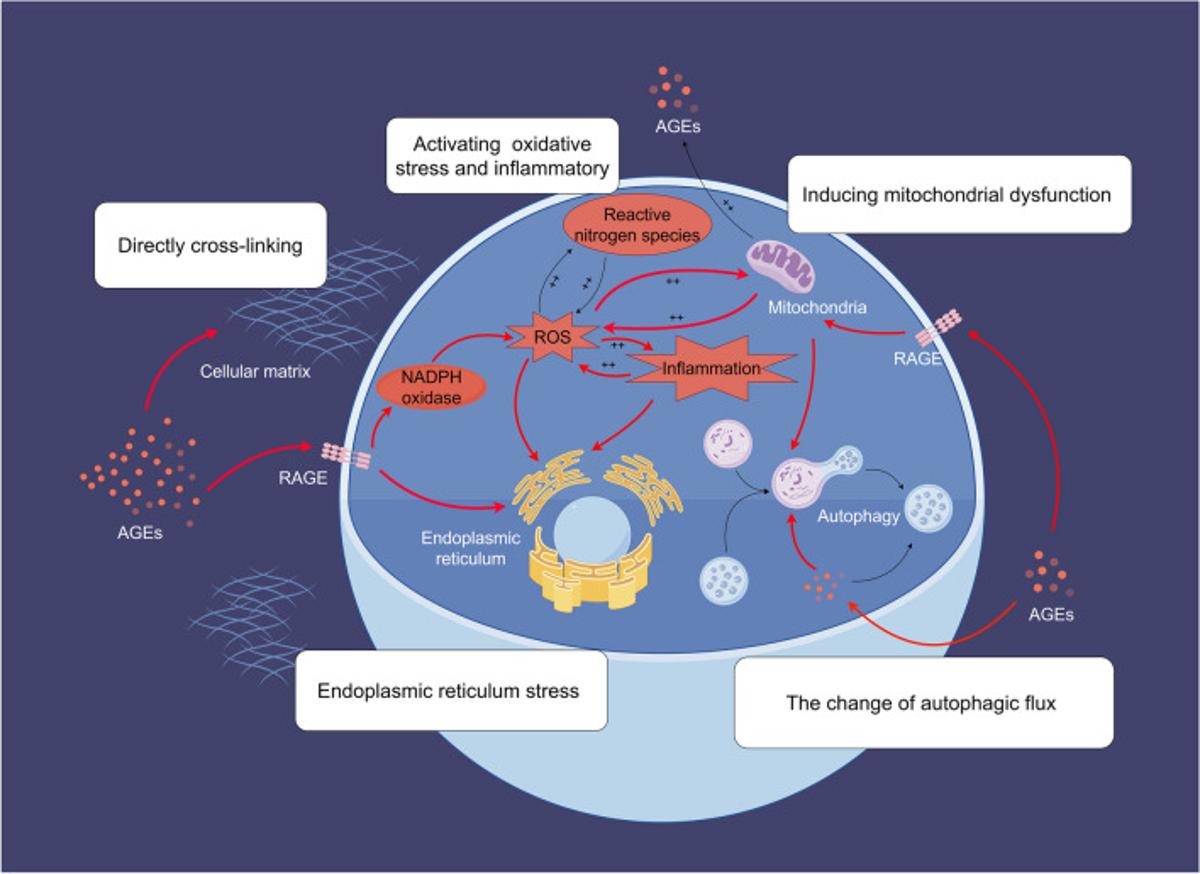
RAGE Gene Therapy Cuts Inflammation, Not Lifespan
It's a gene therapy that targets the RAGE pathway (Receptor for Advanced Glycation End-Products) that mediates inflammation and oxidative stress signaling. So far, the mice data doesn't show significant longevity benefits and mostly reduced inflammation and vascular stiffness and kidney damage. On...

Trump Appeal on Vaccines Validates Connecticut's New Immunization Law
The Trump administration appealed a federal court order that blocked its effort to strip vaccine recommendations for flu, rotavirus, hepatitis, meningitis and RSV, a move championed by Robert F. Kennedy Jr. Connecticut responded by passing a law that anchors the...

Pointed Ironies: SERD Wars, ADC Hype, and What Really Works in Breast Cancer
The FDA rejected camizestrant at the ODAC meeting, while approving vepdegestrant a day later. AstraZeneca’s vepdegestrant leverages ctDNA to detect ESR1 mutations early, allowing a treatment switch while patients remain on a CDK4/6 inhibitor backbone. In contrast, Arvinas pursued a...

Do We Really Need Smarter AI to Cure Cancer?
The debate over AI’s role in curing cancer intensifies as Emilia Javorsky argues that over‑investment in AGI/ASI distracts from practical AI tools and data infrastructure needed for personalized oncology. She stresses cancer’s heterogeneity, urging focus on early detection, trial acceleration,...
Minerva Neurosciences Provides First Quarter 2026 Financial Results and Business Updates
Minerva Neurosciences announced the start of its global confirmatory Phase 3 trial of roluperidone for negative symptoms of schizophrenia, enrolling about 380 patients across 40 sites, with the first patient screened in March 2026. The company reported a GAAP net loss...

The Technology Behind the Story: 3D Skin Grafts and the World of Scarpetta
The article highlights how 3D‑printed skin grafts are transitioning from laboratory experiments to clinical tools for complex wounds. Researchers at Columbia University have demonstrated patient‑specific, three‑dimensional grafts that fit irregular body parts like a glove, reducing surgery time and improving...

Myqorzo ACACIA Study Achieves Dual Primary Endpoint Success
$CYTK Here we go! Myqorzo ACACIA study results in nHCM --> BOTH co-primary endpoints hit with statistical significance. There may be debate about magnitude of improvements for Myqorzo via KCCQ-CSS and peak V02. But, if you were dreaming for a...

STAT+: Cytokinetics Drug Myqorzo Meets Twin Efficacy Goals in Study of Genetic Heart Disease
Cytokinetics announced that its Phase 3 ACACIA trial met both primary efficacy endpoints for Myqorzo in patients with non‑obstructive hypertrophic cardiomyopathy, showing significant symptom relief and improved cardiovascular fitness. Myqorzo is already approved for the obstructive form of HCM, and this...

Patents Hide Life‑Saving Drug Clues Await Discovery
What if the next life-saving drug is buried in a patent diagram? In early drug discovery, progress can hinge on a single decision. The evidence exists, but some of the most valuable insights stay invisible to conventional search, especially inside...

Agents Without Moats Will Falter in Fierce Market
Pointed ironies in the SERD wars, plus ADC hype, and what really works in breast cancer... How agents with no moat, no differentiation, and no clear advantage will struggle in a tough competitive market: https://t.co/2Efcy5ItNM https://t.co/FBDM2eDJd4

HHS Unveils MAHA Action Plan to Slash Antidepressant Overprescribing
HHS Secretary Robert F. Kennedy Jr. announced a federal MAHA Action Plan aimed at curbing the overprescribing of psychiatric medications, with a focus on antidepressants for children. The initiative promises new guidelines, education campaigns, and a study linking antidepressants to...
Slow Alzheimer’s Diagnoses ‘Mean UK Patients Missing Out on Experimental Treatments’
Alzheimer's Research UK warns that delayed or imprecise diagnoses are keeping UK patients out of a surge of experimental drug trials. While global trials hit a record 192 this year, fewer than 1,000 UK participants are enrolled in phase‑3 studies....
NHS to Roll Out ‘1-Minute’ Immunotherapy Jab to Tens of Thousands with Cancer
The NHS is introducing a sub‑cutaneous form of pembrolizumab (Keytruda) that can be administered in just one minute, replacing the traditional two‑hour IV infusion. The rapid jab is approved for 14 cancer types, including lung, breast, head‑and‑neck, and cervical cancers,...

Boehringer’s Next‑Gen GLP‑1 Survodutide Cuts Weight 16% in Phase 3 Trial
Boehringer Ingelheim announced that its experimental GLP‑1/glucagon dual‑agonist survodutide produced an average 16.6% body‑weight reduction in a 725‑person Phase 3 study, far surpassing placebo. The result positions survodutide as a potential next‑generation tool for biohackers focused on metabolic optimization.

Vertex Q1 2026 Revenue Hits $2.99B, CF Sales Drive Growth and UAE Gene Therapy Deal
Vertex Pharmaceuticals posted Q1 2026 total product revenue of $2.99 billion, an 8% year‑over‑year rise led by its cystic fibrosis franchise. The company also highlighted a landmark collaboration with Abu Dhabi’s Stem Cells Centre to deliver the first CRISPR‑Cas9 gene therapy...

Inhibiting ID1 and ID3 Reduces Lung Fibrosis
Researchers identified that the transcription factors ID1 and ID3 are markedly elevated in fibroblasts from idiopathic pulmonary fibrosis (IPF) patients. Using both small‑molecule inhibitors and gene‑silencing techniques, they suppressed these proteins in mouse models, which halted fibroblast activation and triggered...
U of T Engineers Create Polymer Bristle Coating that Blocks Proteins and Germs on Medical Surfaces
University of Toronto engineers have developed a silicone‑based polymer bristle coating that prevents protein adhesion, a key step in bacterial colonisation. The non‑toxic surface could replace harsh disinfectants in hospitals, reducing chemical exposure and the risk of resistant strains.

IBM, Cleveland Clinic & RIKEN Simulate 12,635‑Atom Protein, Quantum Milestone
Scientists from IBM, Cleveland Clinic and Japan's RIKEN have used IBM’s 156‑qubit Heron processors together with the Fugaku and Miyabi‑G supercomputers to simulate a protein complex of 12,635 atoms – the biggest biologically relevant molecule ever tackled with quantum hardware....

Full Asset Acquisitions Avoid Margin Drag of Royalties
Seedy is not long enough $ABVX, however I’m old enough to remember when $ABBV spent $21B to buy half of ibrutinib.
U.S. Courts Are Once Again Litigating Abortion Pill’s Distribution By Mail
U.S. courts are once again debating the mail distribution of the abortion pill mifepristone. The Fifth Circuit Court of Appeals ruled that the drug can only be dispensed in person at clinics, overturning the FDA’s 2021 rule that allowed mail...

Lannett Company and Sunshine Lake Pharma Report US FDA Approval of Langlara (Biosimilar, Lantus)
The U.S. Food and Drug Administration has granted interchangeable biosimilar status to Langlara, a copy of Sanofi’s Lantus insulin glargine. Langlara is approved for adults and children with type 1 diabetes and for adults with type 2 diabetes, based on data showing...

Mirum Pharmaceuticals Reports P-IIb (VISTAS) Trial Data on Volixibat in Primary Sclerosing Cholangitis (PSC) Patients with Cholestatic Pruritus
Mirum Pharmaceuticals announced that its Phase IIb VISTAS trial met the primary endpoint, showing volixibat (20 mg BID) reduced cholestatic pruritus in primary sclerosing cholangitis (PSC) patients by 2.72 points on the Adult ItchRO scale versus a -1.08 point change with...

Canagliflozin - Another Top Longevity Drug
The class of sodium‑glucose cotransporter‑2 (SGLT2) inhibitors has moved from glucose‑lowering pills to a cornerstone of cardiometabolic care. Large‑scale trials and real‑world registries consistently show that agents such as canagliflozin, dapagliflozin and empagliflozin cut heart‑failure admissions, slow chronic kidney disease...

STAT+: Johnson & Johnson Advances IBD Therapy, Despite Trial Miss
Johnson & Johnson's combination therapy JNJ-4804, pairing Tremfya and Simponi, failed to meet the primary clinical remission endpoint in two Phase 2b trials for ulcerative colitis and Crohn's disease, though it outperformed each drug alone. The DUET study results were...
Sertraline Manufacturer Recalls Antidepressant Batch After UK Packaging Mix-Up
Amarox, a subsidiary of India’s Hetero Group, is recalling a batch of 100 mg sertraline tablets after a packaging error placed citalopram strips in the same cartons. The UK Medicines and Healthcare products Regulatory Agency (MHRA) issued the recall following a...

The FDA Wants to Make Many Popular Prescription Drugs OTC—A Great Idea. Here’s Why It’s Unlikely to Happen
FDA Commissioner Marty Makary argues that most prescription drugs should be reclassified as over‑the‑counter unless safety concerns exist. The article highlights how prescription status inflates prices by routing purchases through insurance, citing lower cash prices for OTC versions of Claritin,...

Quantum Computing Moves Closer to Drug Discovery with Enzyme Study
A quantum‑computing team has successfully modeled the active site of a key enzyme, demonstrating that quantum simulations can capture chemical reactions with unprecedented accuracy. The study, conducted on a 127‑qubit superconducting processor, reproduced experimental binding energies within a few kilojoules...

Telomeres: History, Health and Hallmarks of Aging
Bill Andrews, a co‑discoverer of human telomerase, argues that telomere shortening is the primary limiter of human lifespan and that systemic activation of telomerase can reverse biological aging. He promotes small‑molecule activators such as TAM‑818 and botanical blends like Telo‑Vital,...

Comparing Antibiotic Outcomes in Preterm Infants
A recent multicenter cohort study examined how different antibiotic regimens affect outcomes in preterm infants born before 32 weeks. Researchers compared broad‑spectrum empiric therapy with a targeted, shorter‑duration approach, tracking mortality, necrotizing enterocolitis (NEC), late‑onset sepsis, and antimicrobial resistance. The...
Disruptions Threaten Progress in Inclusive Clinical Trials and Health Equity
JMIR Publications highlighted recent disruptions to inclusive clinical trials after a 2025 White House executive order targeting DEI programs. The order led to the termination and delayed funding of NIH‑funded studies, such as Dr. Mohottige’s kidney disease trial, affecting 383...

TGA Updates Australian Manufacturing Licences with New Approvals and Regulatory Actions
The Therapeutic Goods Administration (TGA) released its latest licensing decisions, granting 14 new manufacturing licences for therapeutic goods while suspending two and revoking eight existing licences. The approvals cover a diverse set of entities, including pharmaceutical firms, biotech innovators, logistics...
Takeda's TAK-881 Shows PK Parity with HYQVIA in Pivotal PID Trial
Takeda announced today that its investigational subcutaneous immunoglobulin, TAK-881, met the primary pharmacokinetic endpoint in a pivotal Phase 2/3 trial, showing comparable exposure to HYQVIA while offering reduced infusion volume and flexible dosing for primary immunodeficiency disease patients.

FDA Clears Incyte's Jakafi XR Extended‑Release for MF, PV and GVHD
The U.S. Food and Drug Administration approved Incyte's Jakafi XR extended‑release tablets for adults with intermediate‑ or high‑risk myelofibrosis, polycythemia vera resistant to hydroxyurea, and for patients 12 years and older with steroid‑refractory acute or chronic graft‑versus‑host disease. The once‑daily formulation...