Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Oculis Gains FDA SPA for PIONEER-1 Trial of Privosegtor in Optic Neuritis
Oculis Holding AG announced that the U.S. FDA has granted a Special Protocol Assessment for its PIONEER-1 Phase 3 trial of Privosegtor in optic neuritis. The SPA confirms the study design and primary endpoint, moving the company closer to a potential NDA submission for the first neuroprotective therapy in this rare vision disorder.

The BioPharm Brief: Breakthrough Signals in Cancer, Duchenne, and RNA Medicine
Aptevo Therapeutics reported an 87% clinical benefit rate and 81% remission in its Phase 1b AML RAINIER study, combining mipletamig with venetoclax and azacitidine. Entrada Therapeutics disclosed positive Phase 1/2 data for ENTR‑601‑44, showing functional gains and increased dystrophin in Duchenne patients...
FDA Approves Otarmeni, First Gene Therapy to Restore Hearing in OTOF‑Related Deafness
The U.S. Food and Drug Administration has cleared Regeneron’s Otarmeni, the first gene‑therapy drug to treat genetic hearing loss. The treatment targets OTOF‑related deafness, a condition that affects roughly 200,000 people globally, and promises to restore natural hearing when administered...

Gene Therapy Restores Walking After Paralysis
Researchers at the University Hospital Cologne used a designer cytokine, hyper‑interleukin‑6 (hIL‑6), delivered via an AAV2 viral vector into the motor cortex of mice with spinal‑cord contusions. The protein traveled transneuronally to brainstem serotonergic neurons, prompting intact fibers to sprout...

CMS to Provide Medicare Part D Beneficiaries with $50 Monthly Access to Certain GLP-1 Medications
The Centers for Medicare & Medicaid Services announced a short‑term demonstration called the Medicare GLP‑1 Bridge, offering eligible Part D beneficiaries access to selected GLP‑1 drugs for $50 per month. The benefit starts July 1 2026 and runs through Dec 31 2027. The program is...

DUET Trial: Low-Sodium Oxybate Significantly Consolidates Nighttime Sleep Architecture in Narcolepsy
The phase‑4 DUET trial showed that low‑sodium oxybate (LXB, Xywav) markedly consolidates nighttime sleep in narcolepsy type 1 and type 2 patients. Objective polysomnography recorded a 45‑minute increase in deep N3 sleep, 13 fewer stage transitions and three fewer awakenings per night....

Pharmaceutical Executive Daily: Angelini Pharma Acquires Catalyst Pharmaceuticals in $4.1 Billion Agreement
Angelini Pharma announced a definitive agreement to acquire Catalyst Pharmaceuticals for $31.50 per share in cash, representing a 21% premium to the unaffected closing price and a total equity value of roughly $4.1 billion. The transaction gives Angelini its first direct...

Human Antibodies Identified That Have Potential To Prevent and Treat Measles Virus
Scientists at the La Jolla Institute for Immunology have isolated the first human monoclonal antibodies that can neutralize measles virus. The antibodies, derived from a vaccinated donor, bind the virus' hemagglutinin and fusion proteins, blocking entry into cells. In a...

NACDS Submits Comments to FDA on Expanding Access to Nonprescription Drugs
The National Association of Chain Drug Stores (NACDS) submitted formal comments to the FDA supporting the agency’s initiative to expand over‑the‑counter (OTC) drug availability. NACDS highlighted the potential for greater convenience, preventive care, and timely treatment if prescription‑only therapies move...

AI Longevity Promises Overlook Tiny Real-World Gains
If you hear another AI celebrity telling you on stage that in the next 5 years we will double lifespan or eliminate all diseases - please show them this output from their own LLMs and ask them "How?". This simple...

Stem Cell-Derived Islet Therapies Target Type 1 Diabetes Challenges
Sana Biotechnology is leveraging hypoimmune cell engineering to create allogeneic, stem‑cell‑derived pancreatic islet‑like cells that can evade both adaptive and innate immune attacks. The company aims to deliver a single intramuscular injection that restores normal blood‑sugar control for type 1 diabetes...

Aptevo Reports Strong Remission Data in Frontline AML Trial as RAINIER Study Advances Toward Phase 2
Aptevo Therapeutics disclosed Phase 1b data from its RAINIER trial, showing mipletamig combined with venetoclax and azacitidine achieved an 87% clinical benefit rate and an 81% composite remission rate in 31 frontline AML patients. The regimen produced a 65% complete remission...
Targeting Ischemic Disease with DiaMedica CEO Rick Pauls — Episode 254
DiaMedica Therapeutics, led by CEO Rick Pauls, is advancing a recombinant KLK1 protein to treat ischemia‑driven diseases such as preeclampsia, fetal growth restriction, and acute ischemic stroke. The podcast episode highlights the company’s focus on restoring vascular blood flow and...

Asembia AXS26: How Drug Innovation Could Strain Access Systems
In a follow‑up interview at the Asembia AXS26 summit, Aradigm Health CEO Will Shrank warned that emerging therapies priced above $1 million per patient could overwhelm existing market‑access and financing frameworks. He highlighted upcoming treatments for type 1 diabetes, wet macular degeneration...

Moderna CEO Labels Spike Protein “Garbage” In New Vaccine
Whoa. 🫣 This is incredible. Stephen Hoge, president of Moderna, gave an interview on April 17 to the "why should I trust you?" podcast in which he referred to the spike protein — famously in all the Covid vaccines —...

Ingevity Corporation (NGVT) Q1 2026 Earnings Call Transcript
Ingevity Corporation posted a strong first‑quarter 2026 performance, delivering $210 million in revenue, an 8% year‑over‑year increase, and adjusted EBITDA of $45 million, expanding its margin to 12%. The company unveiled a new high‑performance silica product line aimed at electric‑vehicle battery applications,...

FDA Turmoil Casts Shadow Over Gene‑Therapy Conference in Rome
At the Alliance for Regenerative Medicine’s meeting in Rome, industry leaders grappled with heightened U.S. regulatory uncertainty after the FDA’s cell‑and‑gene therapy rejection rate doubled since 2024 and the agency’s top gene‑therapy regulator, Vinay Prasad, left. Recent approvals for rare‑disease...

TerraPower Isotopes Breaks Ground on $450 Million Actinium‑225 Plant in Philadelphia
TerraPower Isotopes broke ground on a $450 million, 250,000‑square‑foot Actinium‑225 manufacturing plant in Philadelphia, a move that will increase global supply twentyfold. The flagship Bellwether Laboratory aims to support next‑generation alpha‑particle cancer treatments and create roughly 225 permanent jobs.

Feds Expand Pharmacy Benefit Program
The U.S. Department of Labor announced an expansion of pharmacy‑benefit reforms under the Federal Employees’ Compensation Act, extending cost‑saving measures to workers covered by the Black Lung, Longshore and Harbor, and Energy Employees compensation programs. The initiative builds on 2018...

Just ‘Stay Alive for the Next 10 Years’ – Anti-Ageing Drugs Are Coming, Says Billionaire Investor
Billionaire investor Jim Mellon told attendees at Spear’s 500 Live that the first truly anti‑ageing drugs could hit the market within a decade. He highlighted clinical‑stage senolytics, partial genetic reprogramming and repurposed weight‑loss medicines as the leading candidates to halt...

Eli Lilly Adds $4.5 B to Indiana Plant Portfolio, Launches First Genetic Medicine Facility
Eli Lilly announced a $4.5 billion expansion across two Lebanon, Indiana sites and opened Lilly Lebanon Advanced Therapies, its first dedicated genetic‑medicine manufacturing plant. The move lifts the company’s cumulative Indiana capital commitments since 2020 to more than $21 billion, underscoring a...

Parallel 3D Bioprinting Builds Tissue Model Arrays in Minutes
Researchers have introduced a slippery‑liquid‑infused porous surface (SLIPS) droplet microarray that enables parallel digital light processing (DLP) bioprinting of hydrogel tissue models. By removing physical walls and using hydrophilic spots on a superhydrophobic background, the system prints dozens to hundreds...
EnGene's Shares Crash on Updated Pivotal Bladder Cancer Data
EnGene Therapeutics reported Phase 2 data for its experimental bladder‑cancer therapy that revealed a modest 5% objective response rate and safety signals in more than half of patients. The results triggered an 80% plunge in the Canadian biotech’s stock, wiping...

AI Poised to Spark Trillion‑dollar Biotech Revolution
Last day @synbiobeta and had a fascinating discussion with my friend @andrewhessel about the SynBio space. His insight was that SynBio and Biotech feels very much like the AI space pre LLM acceleration. AI will accelerate Biotech and Genetic Agency. Today's biotech...

Blackstone Puts $250M Into Anagram to Tackle Cystic Fibrosis Complication
Blackstone Life Sciences announced a $250 million investment in Anagram, a biotech developing a novel therapy for a cystic fibrosis complication. The capital, drawn from one of the industry’s largest private life‑science funds, will accelerate Anagram’s late‑stage clinical trials, manufacturing scale‑up,...

Anesthetic and Analgesic Drug Products Advisory Committee Charter
The FDA has established the Anesthetic and Analgesic Drug Products Advisory Committee to provide expert advice on the safety and efficacy of marketed and investigational analgesics, including abuse‑deterrent opioids and anesthetic products. The charter, approved April 23, 2026, outlines a...

Entrada Stock Falls on Duchenne Data; Wegovy Expands Access
Entrada Therapeutics reported topline results from its Duchenne muscular dystrophy (DMD) cohort of six patients, showing no meaningful functional improvement. The disappointing data sent Entrada's shares down roughly 15% in after‑hours trading. Meanwhile, Novo Nordisk announced expanded payer coverage for...
Insmed Tanks On Its Most Important Launch; Why Analysts Remain Bullish
Insmed’s newly launched lung drug Brinsupri posted first‑quarter sales of $208 million, beating sell‑side forecasts but falling short of the $230 million buy‑side target. The miss triggered a near‑17% plunge in the stock, sending shares to $114.25, their lowest level since August....

The FDA's One-Day Inspection Pilot Is Already Running
The FDA launched a one‑day inspection pilot announced by Commissioner Marty Makary, with roughly 46 screening assessments completed since April across food, biologics, medical devices and clinical research sites. Most assessments resulted in a No Action Indicated outcome, though a...

FDA Reverses Course on Atara, Pierre Fabre’s Twice-Rejected Cell Therapy After Prasad’s Exit
Atara Biotherapeutics and Pierre Fabre’s EBV‑positive PTLD cell therapy Ebvallo received a regulatory U‑turn after FDA CBER director Vinay Prasad stepped down. The agency now says a single‑arm study with an appropriate historical control can satisfy the “adequate and well‑controlled” requirement, allowing...
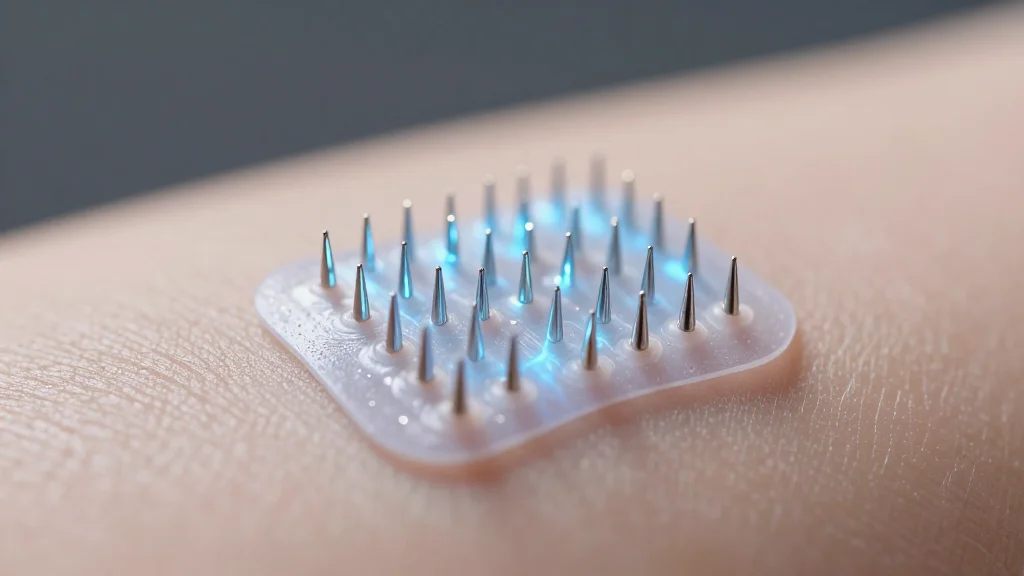
Electric‐Eel‐Inspired Ionic Power Source Microneedles With Self‐Reporting Structural Colors for Wound Healing
Researchers have engineered ionic power source microneedles (IPSMs) that combine electric‑eel‑inspired ion transport with chameleon‑like structural colors for wound care. The three‑layer device creates an internal K⁺‑driven electric field, delivering electrical stimulation that accelerates tissue repair. Integrated silver nanoparticles provide...

Transforming CTMS: An Operating Layer for Real-Time Trial Execution
Clinical trial management systems (CTMS) are evolving from static record‑keeping tools into an operating layer that adds real‑time, AI‑driven reasoning to coordinate complex, multi‑system studies. The article highlights that Phase III trials now span a median of more than ten countries,...

Funding Roundup: Kanvas Biosciences, LTZ Therapeutics Draw Investor Interest for Next-Generation Cancer Immunotherapies
Kanvas Biosciences and LTZ Therapeutics announced a combined $86 million financing round to accelerate next‑generation cancer immunotherapies. Kanvas closed a $48 million Series A to push its microbiome‑based platform and the lead candidate KAN‑001 toward clinical trials. LTZ secured $38 million to expand its...

Let's Do This: Two Concrete Steps You Can Take to Fight Back Against Recent Terrible Court Decisions
The 5th Circuit Court of Appeals temporarily blocked a nationwide ban on mailing the abortion pill mifepristone, while the Supreme Court weakened Section 2 of the Voting Rights Act, prompting redistricting efforts in Alabama, Louisiana and Tennessee. In Georgia, three state...

Amgen Adds $300M to Puerto Rico Budget; Novartis to Exit Oral Drug Factory in Germany
Amgen announced an additional $300 million investment to expand its manufacturing footprint in Puerto Rico, bringing its total U.S. capital outlay to nearly $2 billion over the past year. The funding will support new bioprocessing lines and increase the island’s capacity for...

Breaking Down Frontline BTK Inhibitor Selection in CLL: Kerry Rogers, MD
When choosing a frontline Bruton tyrosine kinase (BTK) inhibitor for chronic lymphocytic leukemia, clinicians currently rely on FDA‑approved covalent agents, with acalabrutinib and zanubrutinib favored for their superior cardiovascular safety over ibrutinib. Emerging data from the 2025 ASH meeting show...

STAT+: Pharmalittle: We’re Reading About Sanofi and an FDA Voucher, FDA Rethinking a Rejection, and More
Sanofi asked the FDA to withdraw its type 1 diabetes antibody teplizumab from the new fast‑track voucher program after Center for Drug Evaluation and Research director Tracy Beth Høeg publicly challenged a staff decision to approve the drug. The agency missed...

Entrada Crashes as Duchenne Therapy Comes in ‘Below Expectations’ in Early Study
Entrada Therapeutics reported that its investigational oligonucleotide ENTR‑601‑44 raised dystrophin levels by 2.36% in the first cohort of its Phase 1/2 ELEVATE‑44‑201 trial, far below the company’s double‑digit target. The modest protein increase triggered a 50% plunge in the Boston‑based biotech’s...

SGLT2 Inhibitors Cut Cardiovascular Death by 14%
As a medical school professor: when I trained, "diabetes drugs" meant blood-sugar drugs. That ceiling has been broken. Meta-analysis in Am J Med (Jaiswal et al, U Chicago, Mar 26 2026) pooled 18 RCTs, n=95,913 patients with diabetes, heart failure, or...
International Team Launches Hantavirus Vaccine Development
JUST IN: Scientists from the UK, US, and South Africa formed a team to develop a hantavirus vaccine, per BBC

Podcast: Autonomous Labs Redefine the Role of Biopharma Researchers
Autonomous laboratories, integrating robotic hardware with AI-driven decision making, are emerging as a transformative force in biopharma R&D. In a GlobalData Media podcast, Frankie Fattorini interviewed Jason Kelly, CEO of Ginkgo Bioworks, who described how these labs can conduct experiments with unprecedented precision...

Preprints Safeguard Vaccine Safety Data From FDA Suppression
"The Paper That Didn't Disappear" FDA reportedly held back several vaccine safety papers headed to publication. Fortunately, one was on medRxiv and remains available. Why preprints matter; my latest Substack: https://t.co/YfTbkEPBU4 Also discussed in #healthandveritas podcast. https://t.co/k24zPtoGA2
CRISPR Selectively Eliminates KRAS‑mutant Cancer Cells
The CRISPR Killer as in KRAS mutated cancer cells https://t.co/y1zrmYbIAQ Explainer thread @N8Krah co-author https://t.co/CIweqZMUqY

STAT+: FDA Revisits a Rare Cancer Treatment It Rejected a Few Months Ago
The FDA has announced it will re‑evaluate a rare‑cancer therapy it dismissed just months earlier, citing new data submitted by the drug’s sponsor. The treatment, aimed at a subtype of metastatic sarcoma, originally failed to meet the agency’s efficacy benchmarks...

Amylyx Pharmaceuticals Q1 Earnings Call Highlights
Amylyx Pharmaceuticals reported that the Phase 3 LUCIDITY trial of its GLP‑1 antagonist avexitide completed dosing and is on track for a top‑line readout in Q3 2026. The company launched a U.S. expanded access program to treat up to 250 adults with...

FDA Launches Real‑Time Clinical Trials to Accelerate Drug Development
The U.S. Food and Drug Administration announced the start of two real‑time clinical trials—one for mantle‑cell lymphoma and another for small‑cell lung cancer—and issued a request for information on a broader pilot program slated for summer 2026. The move aims...

Atara, Pierre Fabre's Cell Therapy to Get Another Shot at FDA Approval
Atara Biotherapeutics and Pierre Fabre Pharmaceuticals are reviving a T‑cell therapy that was rejected twice by the FDA. Regulators have signaled willingness to base a new approval decision on data from a Phase 3 trial, a departure from the earlier requirement...
UK NHS Rolls Out One‑minute Injectable Cancer Therapy, Slashing Visits for 14,000 Patients
The UK National Health Service is replacing two‑hour intravenous pembrolizumab infusions with a one‑minute subcutaneous injection for roughly 14,000 cancer patients each year. The change cuts appointment time by up to 90 % and promises to free clinical capacity while improving...

Wolfe Research Says Market Reaction on Eli Lilly (LLY) Foundayo Report Appears Overdone
Wolfe Research says the market overreacted to an FDA adverse‑event report on Eli Lilly’s GLP‑1 drug Foundayo. Lilly determined the serious hepatic failure case was not reasonably related to the product. Shares fell about 3% in pre‑market trading, yet analysts kept...

Vertex Secures German Reimbursement for CASGEVY Gene Therapy
Vertex Pharmaceuticals announced a reimbursement agreement with Germany's GKV‑Spitzenverband, granting sustainable access to its CRISPR/Cas9 therapy CASGEVY for patients 12 years and older with sickle cell disease or transfusion‑dependent beta thalassemia. The deal makes Germany the latest market to fund...