Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Implantable Cytokine Factories Show Promise Against Advanced Ovarian Cancer
Researchers at Rice University and MD Anderson reported first‑in‑human results for AVB‑001, an implantable cell capsule that continuously secretes interleukin‑2 within the peritoneal cavity of patients with high‑grade serous ovarian cancer. In a Phase I dose‑escalation study of 14 platinum‑resistant patients, the therapy was well tolerated, with no life‑threatening adverse events and half of the participants achieving disease stabilization. Immune profiling showed activation of CD8⁺ T cells and NK cells without expanding regulatory T cells, while dose‑dependent CTLA‑4 up‑regulation points to future combination with checkpoint inhibitors. The findings lay groundwork for repeat‑dosing and combination strategies to improve efficacy.

FDA Approves Ocrevus for Children 10+ with Relapsing‑Remitting MS
The U.S. Food and Drug Administration has approved Genentech's Ocrevus (ocrelizumab) for pediatric patients aged 10 and up with relapsing‑remitting multiple sclerosis. The decision follows the phase‑3 OPERETTA 2 trial, which showed a 48% lower annualized relapse rate versus fingolimod. The...

Isomorphic Labs Secures $2.1 B Series B to Scale AI Drug Design Engine
Isomorphic Labs announced a $2.1 billion Series B funding round led by Thrive Capital, with participation from Alphabet, GV, Temasek, CapitalG and the UK Sovereign AI Fund. The capital will expand its IsoDDE AI engine, accelerate its therapeutic pipeline and fund...

Talphera Inc (TLPH) Q1 2026 Earnings Call Transcript
Talphera reported a $21.1 million cash balance and closed a $4.1 million financing tranche, with two additional conditional tranches of $16 million tied to study milestones. The nephro CRRT trial has surpassed the 50% enrollment target and is on track for full enrollment...
Allogene Therapeutics Inc (ALLO) Q1 2026 Earnings Call Transcript
Allogene Therapeutics reported interim data from its pivotal ALPHA-3 trial, showing SemiCell achieved a 58.3% MRD clearance rate versus 16.7% in the observation arm—a 41.6% absolute improvement—alongside a near‑98% median ctDNA reduction. The safety profile was clean, with no cases...
Beyondspring Inc (BYSI) Q1 2026 Earnings Call Transcript
BeyondSpring Inc. reported that its lead asset plinabulin received FDA priority review with a PDUFA decision deadline of November 30, 2021. The company completed enrollment of 559 patients in the global Phase 3 DUBLIN‑3 trial for second‑line non‑small cell lung cancer...

Genprex Inc (GNPX) Q1 2026 Earnings Call Transcript
Genprex (GNPX) reported Papzimias generated $21.6 million in net product revenue for Q1 2026, up from $3.4 million in the prior quarter, driving total revenue to $23.3 million. The therapy now enjoys coverage for more than 90 % of insured U.S. lives and a permanent...

Humacyte Inc (HUMA) Q1 2026 Earnings Call Transcript
Humacyte reported a sharp rise in Symvess product revenue to $703,000 in Q1 2026, driven by expanding hospital approvals and its first sale to U.S. military facilities after ECAT listing. The company secured 25 Value Analysis Committee approvals covering 92...

PolyPid Ltd (PYPD) Q1 2026 Earnings Call Transcript
PolyPid announced that its D‑PLEX100 product achieved primary and all key secondary endpoints in the SHIELD II Phase III trial, confirming a meaningful reduction in surgical site infections. The FDA provided written feedback endorsing a rolling new drug application, with...

VTv Therapeutics Inc (VTVT) Q1 2026 Earnings Call Transcript
Travere Therapeutics announced the first full FDA approval of its kidney drug Filspari for focal segmental glomerulosclerosis (FSGS) without nephrotic syndrome, expanding the product’s label beyond IgA nephropathy. In Q1 2026, Filspari net sales jumped 88% year‑over‑year to $105.2 million, driving...
Weight Loss Maintained Seven Times More Effectively with Continued Maximum Dose of Tirzepatide, Study Finds
A Phase 3b trial presented at ECO 2026 showed that continuing tirzepatide at the maximum tolerated dose (10 mg or 15 mg) after an initial 60‑week weight‑loss phase preserved a 21.9% reduction from baseline after a further 52 weeks, compared with only 9.9% in the...
Oral Pill
Two new randomized studies just published @NatureMedicine for weaning from injectable GLP-1 drugs to maintain weight loss —Pill, orforglipron https://t.co/r9Nx9J9vJg —Bacterial supplement for gut microbiome https://t.co/km2V3NkIOX

Daily Pill Can Help People Maintain Weight Loss After They Come Off Jabs, Trial Shows
Scientists reported that the oral GLP‑1 receptor agonist orforglipron helps patients retain most of the weight lost with injectable tirzepatide or semaglutide. In a 12‑month randomized trial of 376 U.S. participants, those who switched to the daily pill kept about...
Physionic Podcast Videos and Summaries / Transcripts
A 2023 meta‑analysis of 29,913 patients shows that pure EPA, administered as icosapent ethyl, significantly reduces myocardial infarction, cardiovascular death and all‑cause mortality. The REDUCE‑IT trial, using 4 g daily of prescription EPA, delivered a 25 % relative risk reduction and a...
Personalized DNA Vaccine Doubles Glioblastoma Survival Rates
A phase‑1 trial of Geneos Therapeutics' personalized DNA vaccine GNOS‑PV01 showed it was safe and generated robust immune responses in newly diagnosed glioblastoma patients. The vaccine, which encodes up to 40 patient‑specific neoantigens, more than doubled 12‑month overall survival to...

BMS Just Signed a $15.2 Billion Drug Deal with China’s Biggest Pharma Company. The Patent Cliff Left It No Choice.
Bristol Myers Squibb (BMS) has struck a licensing deal with China’s Jiangsu Hengrui Medicine worth up to $15.2 billion for 13 early‑stage drug programmes in oncology, haematology and immunology. The agreement includes $950 million in near‑term payments and $14.25 billion tied to development,...
Slowing Parkinson’s by Blocking a Key Protein
Researchers at the University of Pennsylvania have pinpointed glycoprotein nonmetastatic melanoma B (GPNMB) as a key driver of alpha‑synuclein spread in Parkinson’s disease. In pre‑clinical models, monoclonal antibodies that block GPNMB halted the neuron‑to‑neuron transmission of toxic protein clumps. Analysis...

Semaglutide Cuts 15% Body Weight in Seniors, Study Shows
Researchers analyzing the STEP trials reported that adults 65 and older taking semaglutide lost an average of 15.4% of body weight over 68 weeks, far outpacing the 5.1% loss seen with placebo. The findings suggest the GLP‑1 agonist is both...

Marty Makary: The Worst FDA Commissioner in 25 Years
And here's @matthewherper on why Marty Makary was the worst FDA commissioner in 25 years https://t.co/a44yjB5sZn
FDA Issues New Guidance on Post‑Approval Pregnancy Safety Data Collection
The U.S. Food and Drug Administration released final industry guidance on how drug sponsors should collect post‑approval safety data for pregnant patients, detailing registry design, study methods and recruitment strategies. The framework aims to fill long‑standing data gaps and improve...
Medicare Spending on New Alzheimer’s Drugs Falls Short of Billion‑Dollar Forecasts
Medicare officials told STAT that spending on the newly approved Alzheimer’s therapies Leqembi and Kisunla will be far lower than earlier projections, with no significant outlays expected for 2026 or 2027. The shortfall reflects muted uptake driven by complex administration,...

AAVs in Focus: Practical Approaches to Capsid Analytics and Plasmid DNA Control
The United States Pharmacopeia (USP) is hosting a three-part webinar series on adeno‑associated virus (AAV) platforms, scheduled for May 19, June 16 and July 8, 2026. Each session moves from an overview of the current AAV landscape to deep dives...

VCs Eye Clinical‑Stage Assets as Biotech Podcast Highlights Investment Playbook
A fresh podcast series hosted by Jeffrey Snyder features Dr. Andrew Snyder of Monash University, who outlines the criteria VCs use to evaluate biotech startups. The discussion flags a surge in IPOs, M&A and a premium on clinical‑stage assets, while...

Makary Out at FDA. Independent Doctors Urge the Next Commissioner to Confront America’s Prescription Drug Crisis — and End Big...
The Independent Medical Alliance (IMA) is urging the White House to appoint a new FDA commissioner who will prioritize patient safety over pharmaceutical profits following Marty Makary's departure. IMA’s president, Dr. Joseph Varon, highlighted that 69% of American adults take...
Acute Myeloid Leukemia Therapy Improved by CRISPR Stem Cell Transplant
A Phase I/II multicenter trial led by Washington University used CRISPR‑Cas9 to delete CD33 from donor hematopoietic stem cells in 30 high‑risk AML or MDS patients. The edited cells engrafted on schedule, with platelet recovery by day 16 and overall...

Multi-Attribute Methods Advance Analytics for Complex Therapeutic Development
At the 2026 AAPS National Biotechnology Conference, Agilent’s Dr. Ganesh Bala highlighted how multi‑attribute LC‑MS methods are reshaping analytics for complex bioconjugates such as antibody‑drug conjugates. He explained that peptide‑mapping‑based MAMs can simultaneously quantify critical quality attributes—including site‑specific conjugation and...

LSD Triggers Region‑Specific White Matter Plastic
Curious. Opposite DTI/FA findings to those we recently published on. Region specific? Neuroplastic white matter changes in patients with major depression following lysergic acid diethylamide treatment: Cell Reports Medicine https://t.co/sIXRAZumie

From Gimmick to Gold Standard
Clinical wearables have transitioned from wellness gadgets to FDA‑recognized trial endpoints, with more than 1,000 interventional studies between 2001 and 2025 already incorporating sensor data. Advances in sensor fidelity, AI‑driven analytics, and regulatory acceptance are enabling continuous cardiac, metabolic, and...
HealthcareWATCH
ICON and Advarra have formed a strategic partnership to create a connected, research‑ready site network that integrates ICON’s global trial services with Advarra’s CTMS, eISF and eSource platforms, promising faster study start‑up and reduced administrative burden. Eli Lilly reported that its...
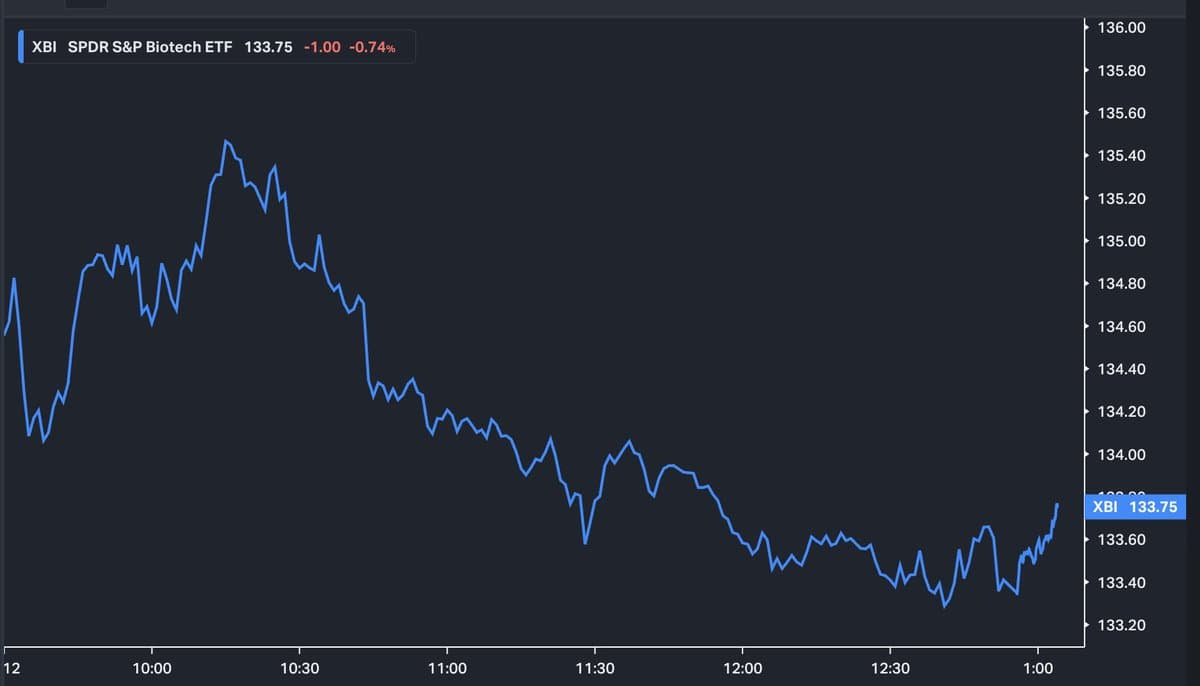
XBI Sees Modest Boost After Anticlimactic FDA Decision
$XBI getting small lift (so far) from the Un-Martying of the FDA. It's all very anticlimactic. https://t.co/q4ta0bkJh4

Arvinas, Pfizer Secure New Partner for PROTAC Breast Cancer Therapy
Arvinas, Pfizer find new partner for ‘Protac’ breast cancer drug https://t.co/IKMKNP1Zfk by @Lilah_Alvarado $ARVN $PFE $RIGL + 7%
Lenz Therapeutics Q1 2026 Revenue Hits $1.9M as COO Olsson Charts Operational Push
Lenz Therapeutics reported $1.9 million net revenue for Q1 2026, driven by $1.7 million in product sales and a 19% rise in filled prescriptions. President and COO Shawn Olsson used the earnings call to detail field‑sales expansion, direct‑to‑consumer initiatives, and manufacturing upgrades, while acknowledging...

Programmable Drug Targets Cancer Cells via DNA Recognition
FinalDose is building the first programmable drug platform - a single smart drug molecule that finds diseased cells by their DNA and destroys them. They're starting with all cancers. Congrats on the launch, @Jeffliu6068Liu, @sklin_lite, and @liyaohuang2! https://t.co/uKJgl7lpmR https://t.co/l4b1hS2mn7

Dual-Ligase Strategy Adds New Layer of Control to Targeted Protein Degradation
Researchers at CeMM, AITHYRA and the University of Dundee have identified a small‑molecule degrader that simultaneously engages two distinct E3 ligases to eliminate SMARCA2/4, key subunits of the BAF chromatin‑remodeling complex. The dual‑ligase mechanism acts as a molecular backup: degradation...
Personalized Neoantigen Vaccine Shows Promise Against Glioblastoma
Personalized neoantigen vaccines vs cancer: we've seen benefits for pancreatic, renal and melanoma. Today initial, encouraging results vs glioblastoma in 9 patients with this deadly brain cancer @NatureCancer https://t.co/ACdCjiXnjJ

Small Cell Lung Cancer Research Moves Toward a More Precision-Driven Era
Small cell lung cancer (SCLC) is transitioning toward precision oncology as researchers uncover distinct molecular subtypes and high‑frequency targets such as DLL3. Amgen’s DLL3‑directed T‑cell engager tarlatamab and emerging antibody‑drug conjugates illustrate a shift from conventional chemotherapy to targeted immunotherapies....

Alkermes' Narcolepsy Phase 3 Success; Boehringer's Antibody Deal
Alkermes announced that its narcolepsy candidate Lumryz achieved its primary and key secondary endpoints in a pivotal Phase 3 trial, demonstrating statistically significant reductions in cataplexy attacks and excessive daytime sleepiness. The study enrolled 500 patients across North America and Europe...

Scarlet Therapeutics Secures $4 Million Seed Round After Lab‑Grown Blood Mirrors Donor Survival
Scarlet Therapeutics announced a $4 million seed funding round alongside data showing its lab‑grown universal red blood cells survive in the bloodstream as long as donor‑derived cells. The result validates a key performance metric for engineered blood products and positions the...

Medisca and Dsm–Firmenich Partner to Expand Access to Pharmaceutical-Grade Vitamin APIs for U.S. Compounding Market
Medisca has struck a strategic partnership with dsm‑firmenich to bring a portfolio of pharmaceutical‑grade vitamin active pharmaceutical ingredients (APIs) to U.S. compounding pharmacies and 503B outsourcing facilities. The vitamins will be sourced from dsm‑firmenich’s European cGMP manufacturing sites, ensuring compliance...

Mayo Clinic Starts Clinical Use of New Phase’s Magnetic Nanoparticle System to Ablate Tumors
Mayo Clinic has begun treating patients with New Phase’s magnetic nanoparticle‑mediated hyperthermia system, marking the first U.S. clinical use of the technology. Six stage‑4 metastatic cancer patients have already received the investigational therapy under an FDA‑granted IDE, highlighting a new...
Pancreatic Cancer Patient Gains Years with New Targeted Drug Daraxonrasib
Patient Vicky Stinson, diagnosed with stage‑III pancreatic cancer, is now living beyond expectations after receiving daraxonrasib, a mutation‑targeting therapy that recent trial data say triples survival compared with standard chemotherapy. The story underscores a wave of biotech advances that could...

20/20 BioLabs Exclusively Licenses PSA Velocity Algorithm From the University of South Carolina
20/20 BioLabs announced an exclusive worldwide licensing deal with the University of South Carolina to commercialize a patented PSA velocity algorithm that flags rapidly rising prostate‑specific antigen levels. The tool identifies aggressive prostate tumors before PSA crosses the conventional 4.0 ng/mL...

Lexicon Pharmaceuticals to Present at the 4th Annual H.C. Wainwright BioConnect Investor Conference
Lexicon Pharmaceuticals (NASDAQ: LXRX) announced that its management will join a fireside chat at the 4th Annual H.C. Wainwright BioConnect Investor Conference on May 19, 2026, held at Nasdaq World Headquarters in New York. The session will be streamed live...

Rigel Signs ~$445M Licensing Pact with Arvinas and Pfizer for Veppanu
Rigel has signed an exclusive, global licensing agreement with Arvinas and Pfizer to develop, manufacture and commercialize the FDA‑approved oral PROTAC drug Veppanu (vepdegestrant) for ER+/HER2‑, ESR1‑mutated advanced or metastatic breast cancer. The deal includes a $70 million upfront payment, a...

EU Agrees Plan to Tackle Essential Medicine Shortages
The European Union has reached a provisional agreement on the Critical Medicines Act, a legislative package aimed at strengthening the supply chain for essential drugs. The act requires member states to diversify procurement, prioritize EU‑based manufacturing, and launch strategic projects...
STAT+: Capsida Says It Still Doesn’t Know What Caused Gene Therapy Death
Capsida Biotherapeutics announced it still cannot determine the cause of a child’s death that occurred during the September 2025 CAP-002 gene‑therapy trial, the first brain‑penetrating treatment of its kind. The investigation is stalled because the hospital conducting the study has...

U.S. Lawmakers Push FDA to Fast‑Track Psychedelic Therapies for Mental Health
A bipartisan group of 32 House members, led by Reps. Jack Bergman and Lou Correa, sent a May 1 letter to FDA Commissioner Martin Makary urging the agency to speed up evaluation of psychedelic‑based treatments for PTSD, depression and substance‑use disorders. The...
RIGL to Sell Approved Veppanu for ARVN, PFE
$RIGL will sell the newly approved Veppanu for $ARVN and $PFE. Modest financial terms https://t.co/9RsbSMzxBf
Novo Says High-Dose Wegovy Can Cause 28% Weight Loss
Novo Nordisk reported that its high‑dose 7.2 mg Wegovy formulation enabled early responders to lose nearly 28 % of body weight in the 72‑week STEP UP trial. Across the 1,400‑patient study, average weight loss reached 21 %, with 27 % of participants qualifying as early...

Scientists Question $1,000‑Plus NAD+ Supplements Amid Booming Longevity Market
Leading researchers say the surge in NAD+ supplements and infusions outpaces the scientific evidence supporting them. While clinics charge hundreds to thousands of dollars per treatment, human trials remain small and inconclusive, fueling a debate between biohackers and the scientific...