Today's Pharma Pulse
CMS to launch Medicare GLP‑1 Bridge program with $50 monthly copay for obesity drugs
The Centers for Medicare & Medicaid Services will start a short‑term demonstration on July 1 2026 offering eligible Part D beneficiaries a flat $50 monthly copay for three branded GLP‑1 obesity treatments. The program runs through December 31 2027 and aims to gather utilization data ahead of potential broader policy decisions.

BMS, Hengrui Ink $15.2B Oncology Deal Covering 13 Programs
Bristol Myers Squibb and Hengrui Pharma have signed a global strategic collaboration that could be worth up to $15.2 billion, covering 13 early‑stage programs in oncology, hematology and immunology. The agreement gives BMS exclusive rights outside China and Hengrui exclusive rights inside China for each other's assets, with a $600 million upfront payment and multiple milestone payments.

Contributor: Fuel Drug Development, Not Big Pharma's Profits
The author, a 65‑year‑old ALS patient, urges faster U.S. drug development for amyotrophic lateral sclerosis, highlighting the pending ACT for ALS legislation. He notes more than 200 ALS drug candidates are stalled by a system that favors large pharmaceutical firms...

Halozyme Q1 Revenue Jumps 42% to $377 M on ENHANZE Therapy Surge
Halozyme Therapeutics posted first‑quarter 2026 revenue of $377 million, a 42% year‑over‑year increase, as ENHANZE‑enabled products gained market share. The company also unveiled a $1 billion share‑repurchase program and reaffirmed full‑year guidance of $1.71‑$1.81 billion in revenue.

US Government Studies Into Vaccine Safety Are Being Suppressed | Robert B Shpiner
The FDA withdrew two peer‑reviewed COVID‑19 vaccine safety studies—one analyzing 7.5 million Medicare beneficiaries and another covering 4.2 million recipients—after political appointees refused to sign off, despite journal acceptance. A separate Shingrix safety abstract was also blocked, even though it confirmed a...

AI-Designed Drug Reduces Fentanyl Consumption in Animal Models by Targeting Serotonin Receptors
Researchers at UC Irvine used an artificial‑intelligence platform to design a novel serotonin‑receptor drug, GATC‑1021, that dramatically lowers fentanyl self‑administration in rats. In dose‑response studies the compound cut fentanyl intake by more than 60% and maintained efficacy without developing tolerance....
How an ‘Impossible’ Idea Led to a Pancreatic Cancer Breakthrough
Daraxonrasib, a KRAS‑targeting drug, is on the brink of regulatory approval and has shown the first meaningful survival benefit for pancreatic cancer patients. The molecule binds a mutated KRAS protein long deemed “undruggable,” a breakthrough that also appears effective against...

SMC Backs First-Line Metastatic Bladder Cancer Combination After Trial Results
Scotland’s Medicines Consortium (SMC) has approved reimbursement for the first‑line combination of enfortumab vedotin and pembrolizumab for adults with unresectable or metastatic urothelial cancer. The decision follows the phase 3 EV‑302 trial, which showed median overall survival of 33.8 months versus...

Inhibrx Announces Positive Interim Results From HexAgon Trial
Inhibrx Biosciences reported positive interim Phase II data from its HexAgon trial of INBRX‑106 combined with pembrolizumab in PD‑L1‑positive metastatic head and neck squamous cell carcinoma. In the evaluable cohort, the combination achieved a 44% objective response rate versus 21% with...
EktaH Links Novel Obesity Drug to Fat Loss, Muscle Retention in Early-Phase Trial
EktaH disclosed early‑phase data for two oral candidates that activate fat‑taste receptors CD36 and GPR120, aiming to treat obesity by restoring lipid sensing. In a four‑week dose escalation study, the CD36/GPR120 agonist NKS‑5 reduced fat mass by 4.30% and modestly...

The Real Reason CGT Programs Struggle to Scale
Cell and gene therapy (CGT) programs often start with fresh leukapheresis material because it speeds early feasibility work. As patient cohorts and sites expand, the tight timing and manual handling of fresh workflows create operational variability and strain manufacturing capacity....

RESEARCH: TOCOTRIENOLS in COLORECTAL CANCER - 2024 Review Paper From Malaysia
A 2024 review from Malaysia examined 38 peer‑reviewed articles on tocotrienols, a subclass of vitamin E, and their effects on colorectal cancer. The analysis highlighted two isoforms, gamma‑ and delta‑tocotrienol, which consistently inhibited tumor cell proliferation, induced apoptosis, and reduced metastatic...

Roche Gets Second European Approval for Alzheimer’s Test
Roche, in partnership with Eli Lilly, secured a CE mark for its second Elecsys blood test, the pTau217 assay, enabling sales across the European Economic Area. The new test can both confirm and exclude Alzheimer’s disease, expanding on the earlier pTau181...

BMS Partners with Hengrui Pharma in a Potential ~$15.2B Deal to Advance 13 Early-Stage Programs Across Oncology, Hematology, and Immunology
Bristol‑Myers Squibb (BMS) has struck a strategic partnership with China’s Hengrui Pharma to advance 13 early‑stage programs covering oncology, hematology and immunology. Hengrui will lead early clinical development while BMS receives exclusive ex‑China rights to the assets, and Hengrui secures...

Swish Club Rebrands To SwishX, Pivots To AI For Pharma
Swish Club has rebranded as SwishX and shifted from device leasing to an agentic AI platform serving pharmaceutical and med‑tech firms. The new suite includes Tender IQ, Contract IQ, Marketing IQ and Channel IQ, aimed at automating sales, marketing, distribution...
FDA Issues CMC Flexibility Guidance to Accelerate Cell and Gene Therapy Development
The U.S. Food and Drug Administration published a new guidance outlining chemistry, manufacturing and controls (CMC) flexibilities for human cellular and gene therapy (CGT) products. The draft lists phase‑appropriate CGMP exemptions, permissive release criteria and risk‑based comparability pathways intended to...
FDA Grants First NRG1‑Fusion Targeted Therapy Approval for Cholangiocarcinoma
The FDA approved Partner Therapeutics' BIZENGRI (zenocutuzumab‑zbco) for adults with advanced NRG1‑fusion cholangiocarcinoma, the first targeted therapy for this rare subtype. The decision, accelerated by a National Priority Voucher, rests on a Phase 2 trial showing a 36.8% overall response rate...
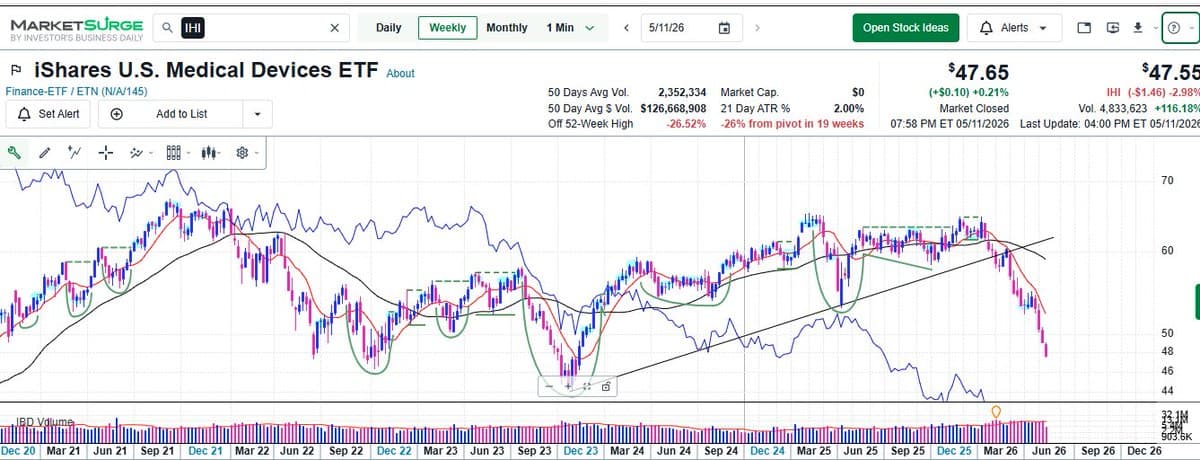
GLP‑1 Disruption Drives Healthcare to 13‑year Low
GLP-1 disruption has caused the concentrated Med-Tech ETF $IHI to weigh sharply on Healthcare's performance this yr, resulting in what used to be the 2nd largest sector in SPX slipping to just 8% @IBDinvestors @marketsurge Selectivity crucial until this Sub-industry...

DOSE Trial Reveals Best Heart Failure Infusion Strategy
Bolus or drip? What the DOSE trial actually showed about heart failure [PODCAST] http://dlvr.it/TSV5l4 Podcast #Cardiology

Postbiotics Use Reduces Gingival Inflammation - Japan Study
A double‑blind, placebo‑controlled trial in Japan found that a gummy containing 500 mg of heat‑inactivated Lactiplantibacillus pentosus ONRICb0240 significantly reduced gingival inflammation over six weeks. Participants taking the postbiotic showed bleeding on probing fall from 17.6% to 12.3% and a Gingival...
Tomatidine Boosts Memory and Cuts Cellular Aging in Mice
Tomatidine is a senotherapeutic compound that improves cognitive function and reduces cellular senescence in aged mice https://t.co/jVfshXgzxQ https://t.co/6l86CBdoBC

EnGene's Phase II Data Triggers 83% Sales Forecast Plunge
11May: How much have peak sales forecasts nosedived after $ENGN reported updated Phase II data for detalimogene? Read about enGene Therapeutics' 83% plunge plus an IPO roundup and updates on $ATRA, $TRDA, $MRNA, & $VRDN in my latest StockWatch for...
RNA Therapy Slows Harmful Heart Remodeling After Heart Attack in Clinical Trial
A Phase II international trial (HF‑REVERT) tested CDR132L, an antisense inhibitor of microRNA‑132, in 294 patients who suffered a heart attack. The drug was administered in three intravenous doses alongside standard heart‑failure therapy and proved safe, with no liver, kidney or...
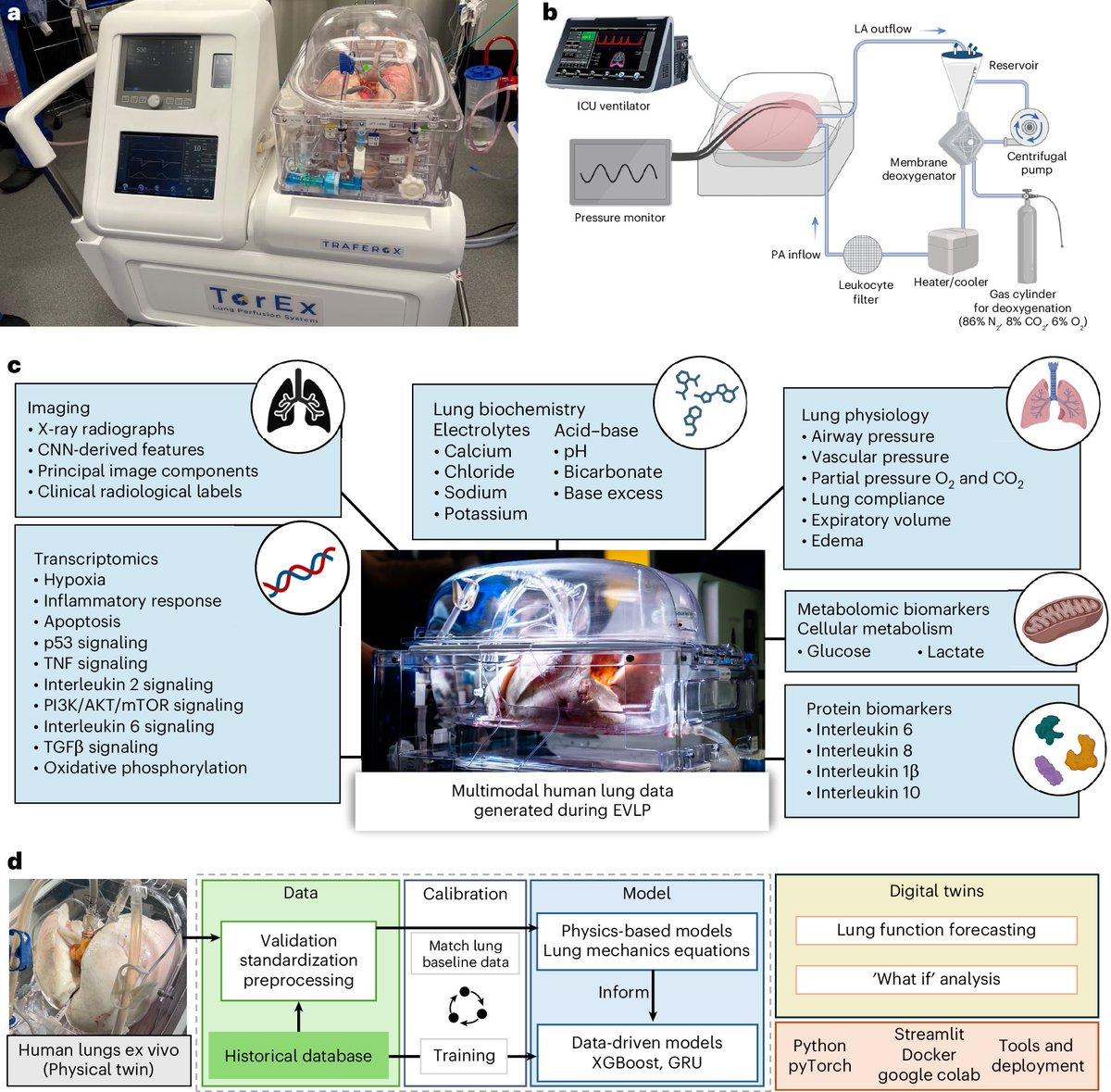
Digital Twins of Human Lungs Personalize Therapy Assessment
Digital twins of ex vivo human lungs enable accurate and personalized evaluation of therapeutic efficacy https://t.co/0FMBqmiMyM https://t.co/3MrdTqK82w

Urolithin A Trial Shows 12% Muscle Strength Gain and Better Endurance in Middle‑Aged Adults
Researchers at EPFL and the NIH reported that a clinical trial of Urolithin A supplementation increased muscle strength by 12% and enhanced aerobic endurance in middle‑aged participants. The results highlight the compound’s potential as a mitochondrial‑targeted anti‑aging supplement, while underscoring the...
FDA Expands Vyvgart Approval to All Adult Myasthenia Gravis Patients
The U.S. FDA has broadened the label for Argenx's FcRn antagonist efgartigimod (Vyvgart) to include every adult with generalized myasthenia gravis, regardless of antibody status. The decision rests on positive Phase 3 data from the ADAPT SERON trial, which showed meaningful...

CMS Launches $50‑a‑Month Medicare GLP‑1 Bridge Covering Foundayo, Wegovy and Zepbound
The Centers for Medicare & Medicaid Services announced that, beginning July 1, 2026, eligible Medicare beneficiaries will receive covered access to specific GLP‑1 medications at a flat $50 per month under a new GLP‑1 Bridge program that runs through Dec 31, 2027. The rollout...
Is Biogen Inc. (BIIB) One of the Best Biotech Stocks to Invest In According to Billionaire Steve Cohen?
Biogen Inc. agreed to pay up to $850 million for exclusive Greater China rights to felzartamab, an experimental immune‑therapy from TJ Biopharma. The deal includes a $100 million upfront payment and up to $750 million in milestones and royalties. Felzartamab is in late‑stage...

Is Regeneron Pharmaceuticals (REGN) Among the Best Biotech Stocks to Invest In According to Billionaire Steve Cohen?
Regeneron Pharmaceuticals received FDA approval for Otarmeni, the first gene therapy targeting genetic hearing loss caused by OTOF gene mutations. The treatment delivers a functional copy of the OTOF gene via a viral vector directly into the cochlea and will...
AstraZeneca PLC (AZN) Stock Falls as FDA Panel Votes Against New Cancer Drug
AstraZeneca’s oral breast‑cancer candidate camizestrant faced a setback when an FDA advisory panel voted 6‑3 against its approval on May 1. The panel cited the SERENA‑6 trial’s inability to demonstrate a long‑term survival advantage for early switching, though it raised no...

Immunome, Inc. (IMNM) Reports Submission of an NDA for Varegacestat in Desmoid Tumors
Immunome, Inc. filed a new drug application with the FDA for varegacestat, a gamma‑secretase inhibitor aimed at treating desmoid tumors. The filing relies on Phase 3 RINGSIDE trial data that met its primary endpoint, showing an 84% reduction in the risk...

Ensoma to Unveil First-in-Human Safety Data for In Vivo HSC Gene Therapy at ASGCT
Ensoma announced it will present initial safety data from the first participant dosed with EN-374, an in vivo hematopoietic stem cell (HSC) gene‑editing therapy for X‑linked chronic granulomatous disease, at the American Society of Gene & Cell Therapy (ASGCT) Annual...

Arvinas Inc (ARVN) Q1 2026 Earnings Call Transcript
Arvinas reported a steep revenue drop to $9.5 million in Q4, primarily due to the loss of a $40 million Novartis license, while cash fell to just over $85 million. The company cut G&A and R&D expenses, extending its cash runway into 2028,...

Animal-Testing Alternatives Will Require a Cultural Change in Research Institutions
New‑approach methodologies (NAMs) are gaining traction as alternatives to animal testing, backed by recent commitments from the US, UK and Canadian governments to fund their development and regulatory integration. While the science is advancing, the authors argue that institutional culture—routines,...

Imunon Inc (IMNN) Q1 2026 Earnings Call Transcript
Imunon reported $5.3 million cash on hand, extending its runway to mid‑Q1 2026 after a $4.5 million equity raise. Operating expenses fell 31% YoY, with R&D down 44% and CMC down 52%, cutting monthly burn to $1.25‑$1.5 million. The pivotal OVATION 3 phase III ovarian‑cancer trial...
Acurx Pharmaceuticals Inc (ACXP) Q1 2026 Earnings Call Transcript
AcelRx announced that its sufentanil product DSUVIA received Milestone C approval from the Department of Defense, allowing inclusion in U.S. Army kits. The company forecasts roughly $30 million in initial military orders over the next three years and expects broader adoption...

Testosterone Treatment Found to Improve Sexual and Physical Function for Men After Prostate Cancer Surgery
A randomized SPIRIT trial led by Shalender Bhasin showed that three months of testosterone replacement therapy (TRT) markedly improved sexual activity, desire, physical function, and aerobic performance in men who had undergone radical prostatectomy. The study enrolled 136 men with...

News Wrap: Supreme Court Temporarily Extends Access to Mifepristone
The U.S. Supreme Court issued an administrative stay on Friday, extending access to the abortion pill mifepristone through at least Thursday. The stay preserves telemedicine prescribing and mail delivery while justices consider an emergency request to block a lower‑court ruling...

The BioPharm Brief: Precision Medicine Expansion Accelerates Autoimmune and Targeted Oncology Development
The FDA broadened Vyvgart’s label to cover all adult patients with generalized myasthenia gravis, removing the previous antibody‑status restriction. Zai Lab received fast‑track designation for its DLL3‑targeting antibody‑drug conjugate aimed at extrapulmonary neuroendocrine carcinomas, a disease with limited options. The...

New AI Target Discovery Paper Sets Benchmark
New Paper Alert 🚨: A collaboration with the leading target ID experts in Nature Reviews Drug Discovery. If you are looking to teach your AI target discovery - this is a good place to start. Thanks Peter Kirkpatrick for giving...
FDA Seeks Input On Chronic Diseases To Prioritize For Drug Repurposing
The FDA announced a public‑comment period to identify chronic diseases that should be prioritized for drug‑repurposing. The agency seeks stakeholder input on which conditions lack commercial interest and which approved drugs could be redirected to new therapeutic uses. This effort...

Justice Alito Extends Administrative Stay of Mifepristone Order
Justice Samuel Alito extended the administrative stay on the Fifth Circuit's order halting telemedicine prescriptions of the abortion medication mifepristone, setting a new deadline for Thursday. The extension maintains the status quo while the Supreme Court considers whether to issue...

Eli Lilly’s Orforglipron Cuts Weight up to 13% and HbA1c 1.7% in Seniors, Study Shows
At the European Congress on Obesity, Eli Lilly presented a post‑hoc analysis of its oral GLP‑1 agonist orforglipron showing seniors lost up to 13% of body weight and cut HbA1c by 1.7% over 72 weeks. The findings, drawn from 616 participants...
Windward Bio Secures $165 Million to Accelerate Late‑Stage Asthma and COPD Trials
Windward Bio announced a $165 million crossover financing round led by OrbiMed, giving the Basel‑based biotech the runway to push its WIN378 antibody into Phase 3 trials for asthma and COPD. The capital injection underscores strong investor confidence in long‑acting respiratory...

Supreme Court Allows Abortion Pill Access for Three Days
JUST IN: The Supreme Court grants a widely accessed abortion pill temporary availability for at least three days.

UnitedHealth Ends Spread Pricing, Signals PBM Reform
Optum Rx/UnitedHealth says its ending "spread pricing" in its PBM, after years of criticism, @ShelbyJLiv reports for @endpts This is a big change in PBM policy, as PBMs face pressure to reform some of the practices they've faced scrutiny over: https://t.co/jq4FmvrO9C

Supreme Court Continues Access to Abortion Pill by Mail, for a Few Days
The U.S. Supreme Court, through Justice Samuel A. Alito Jr., extended a temporary stay that keeps the abortion pill mifepristone available by mail nationwide until 5 p.m. on May 14. The pause follows an emergency petition by the drug’s manufacturers to block...
Atsena Advances XLRS and LCA1 Gene Therapies to Pivotal Phase 3 Trials
Atsena announced that its XLRS (ATSN-201) and LCA1 (ATSN-101) gene‑therapy programs are entering pivotal Phase 3 studies. The move follows 12‑month foveal schisis closure in 7 of 9 treated eyes and a sustained 20‑decibel improvement in dark‑adapted vision across 15 LCA1...

DOJ, DEA Reschedule Medical Cannabis Products
The U.S. Justice Department and DEA have reclassified FDA‑approved and state‑licensed medical cannabis products from Schedule I to Schedule III under the Controlled Substances Act. The change follows a 2025 Trump executive order and a 2024 DEA proposal that attracted 43,000 public...

Pharmaceutical Executive Daily: GSK Enters Collaboration to Support Launch of Bepirovirsen
GlaxoSmithKline (GSK) has signed a 5.5‑year exclusive partnership with Chia Tai Tianqing Pharmaceutical Group to launch bepirovirsen, a chronic hepatitis B candidate, across more than 5,000 Chinese hospitals. The drug is currently in priority regulatory review in mainland China, while GSK...

Spotlight On: Biosimilar Litigations - May 2026
The May 2026 Venable LLP update delineates which disputes qualify as biosimilar litigations. It includes lawsuits between biosimilar applicants or manufacturers and reference‑product sponsors, as well as conflicts among biosimilar firms themselves. It expressly excludes disputes solely between reference‑product sponsors, university‑sponsor...