New Antibody Drugs Target Disease From Within
Why It Matters
Enabling antibodies to function inside cells opens a new therapeutic class for neurodegenerative and other intracellular diseases, potentially accelerating drug pipelines and repurposing existing biologics.
Key Takeaways
- •AI redesign stabilizes over 600 intracellular antibody fragments.
- •Intracellular antibodies directly bind neurodegenerative disease proteins.
- •Charge engineering prevents aggregation inside cells.
- •Platform rapidly converts existing antibodies for new intracellular targets.
- •Gene‑delivery lets cells produce intracellular antibody fragments.
Pulse Analysis
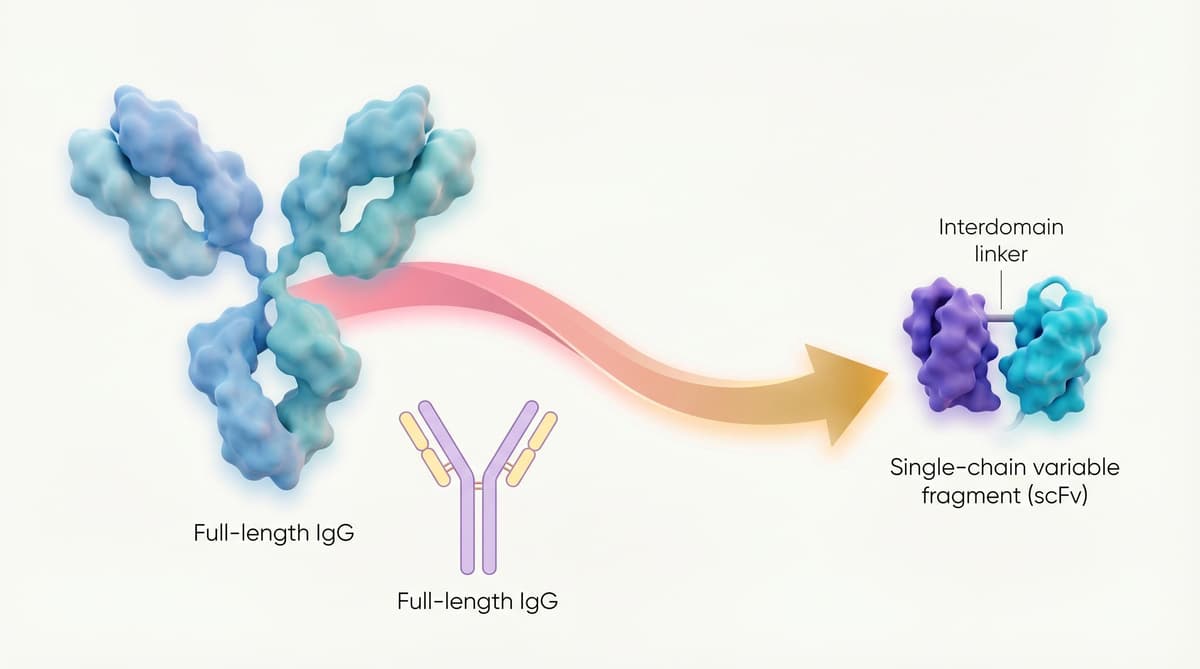
Antibody therapeutics have dominated the biopharma landscape, yet their activity has been confined to extracellular targets because full‑size immunoglobulins and even fragment formats quickly lose stability inside the cytosol. A recent study leverages artificial‑intelligence‑driven protein engineering to rewrite the charge profile of the antigen‑binding (Fab) region, producing more than 600 intracellular‑compatible fragments. By preserving the original paratope while eliminating aggregation‑prone residues, the redesigned molecules stay soluble and functional within the reducing, high‑ionic‑strength environment of living cells. This technical leap turns a long‑standing biological barrier into a designable space.
The most immediate beneficiaries are neurodegenerative disorders, where pathogenic protein aggregates such as tau, α‑synuclein and huntingtin accumulate inside neurons. Intracellular antibody fragments can now recognize these misfolded species with the same precision that conventional antibodies achieve on cell surfaces, offering a way to neutralize toxic proteins at their source. Early data suggest selective binding to disease‑specific conformations without disturbing normal isoforms, a critical safety consideration. Beyond Alzheimer’s and Parkinson’s, the approach is adaptable to cancer‑intracellular oncogenes, inflammatory signaling molecules, and rare genetic mutations that have eluded extracellular targeting.
From a commercial perspective, the platform creates a reusable pipeline: any approved or pipeline antibody can be retrofitted for intracellular delivery, dramatically shortening development timelines and reducing R&D costs. Coupled with viral or mRNA‑based gene‑delivery systems, manufacturers could program patients’ own cells to manufacture the therapeutic fragment, echoing the emerging cell‑free biologics trend. Investors are likely to view this as a high‑margin, multi‑indication asset, especially as the biotech sector seeks novel modalities to address unmet disease areas. As AI‑guided redesign matures, the line between extracellular biologics and intracellular gene‑editing tools may continue to blur.
New Antibody Drugs Target Disease From Within
Comments
Want to join the conversation?
Loading comments...