The Dosing Problem That Precision Medicine Has Yet to Solve
Why It Matters
Accurate dosing directly impacts treatment efficacy and toxicity, making exposure monitoring a missing yet critical layer of precision oncology. Scaling a convenient sampling solution could unlock real‑world pharmacokinetic data that informs clinicians, pharma and payers alike.
Key Takeaways
- •Only 45% of cancer patients receive optimal drug dose.
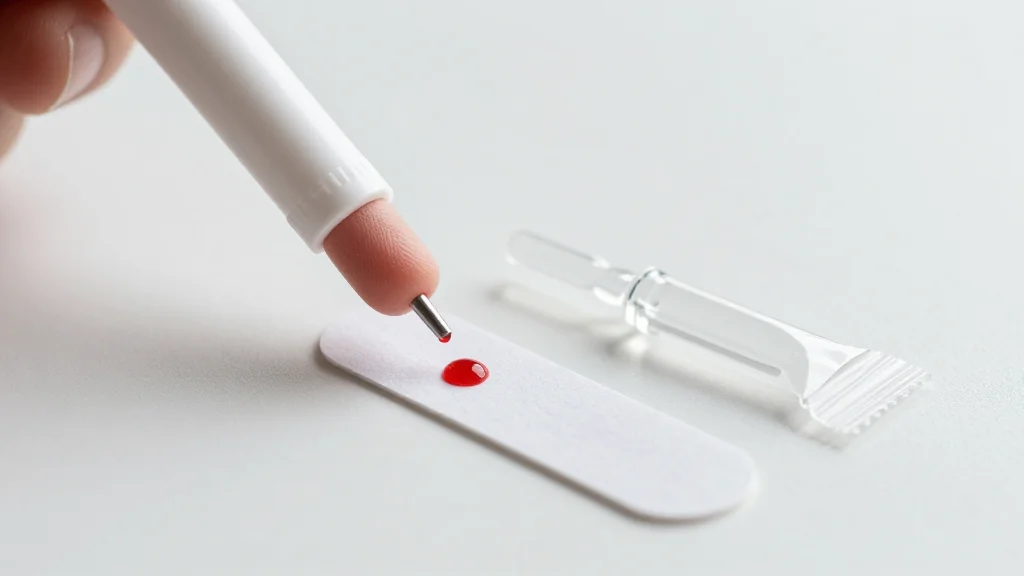
- •True Dose offers at‑home fingerprick sampling for oncology drugs.
- •Early studies show capillary samples match venous draws.
- •Adoption needed for data layer and predictive dosing.
- •Reimbursement and workflow integration remain major hurdles.
Pulse Analysis
The persistent dosing gap in oncology highlights a blind spot in precision medicine. While genomic profiling and targeted agents have transformed treatment selection, they ignore the pharmacokinetic variability that determines whether a drug reaches its intended exposure. Under‑dosing can lead to disease progression, whereas over‑dosing fuels severe toxicity, both eroding patient outcomes and inflating healthcare costs. Addressing this gap requires routine therapeutic drug monitoring, yet traditional venous draws impose logistical burdens that many patients and clinics cannot sustain.
True Dose’s at‑home dried blood‑spot system seeks to lower that barrier. By enabling a simple fingerprick, stabilising the sample at room temperature, and mailing it to a central lab, the company converts a complex clinical workflow into a consumer‑friendly process. Validation studies published in 2025 demonstrate comparable accuracy to venous sampling for drugs such as tamoxifen, epirubicin, taxanes, CDK4/6 and ALK inhibitors. With CE marking secured for the European market, the technology is poised for early commercial rollout, but its success hinges on clinician acceptance, insurance reimbursement and seamless integration into electronic health records.
If widely adopted, the platform could generate a longitudinal, multi‑drug exposure dataset—True Dose Insights—that fills a critical data void. Pharma could leverage this for post‑marketing pharmacokinetic analyses, while payers might use it to refine value‑based contracts. Moreover, the aggregated data could fuel predictive dosing algorithms, moving oncology from reactive dose adjustments to proactive, individualized regimens. Realizing these benefits will require scaling the service, ensuring data quality, and navigating regulatory and reimbursement landscapes, but the potential to enhance treatment efficacy and reduce toxicity makes the endeavor compelling for the entire oncology ecosystem.
The Dosing Problem That Precision Medicine Has Yet to Solve
Comments
Want to join the conversation?
Loading comments...