Recent Posts

News•Feb 27, 2026
Multicenter Study Shows High Local Tumor Control After Resection with GammaTile in Newly Diagnosed Brain Metastases
GT Medical Technologies released Phase IV registry data showing that GammaTile brachytherapy, implanted at the time of surgical resection, achieved 92.3% local tumor control in newly diagnosed brain metastases after a median 12.4‑month follow‑up. The study of 51 patients reported a 7.8% cumulative incidence of leptomeningeal disease and a 9.8% rate of Grade 3 toxicity, with no radiation necrosis observed. Researchers highlighted the advantage of immediate, localized radiation in closing the treatment gap between surgery and conventional radiotherapy. Ongoing randomized trials will further define GammaTile’s comparative efficacy.
By Imaging Technology News (ITN)
News•Feb 26, 2026
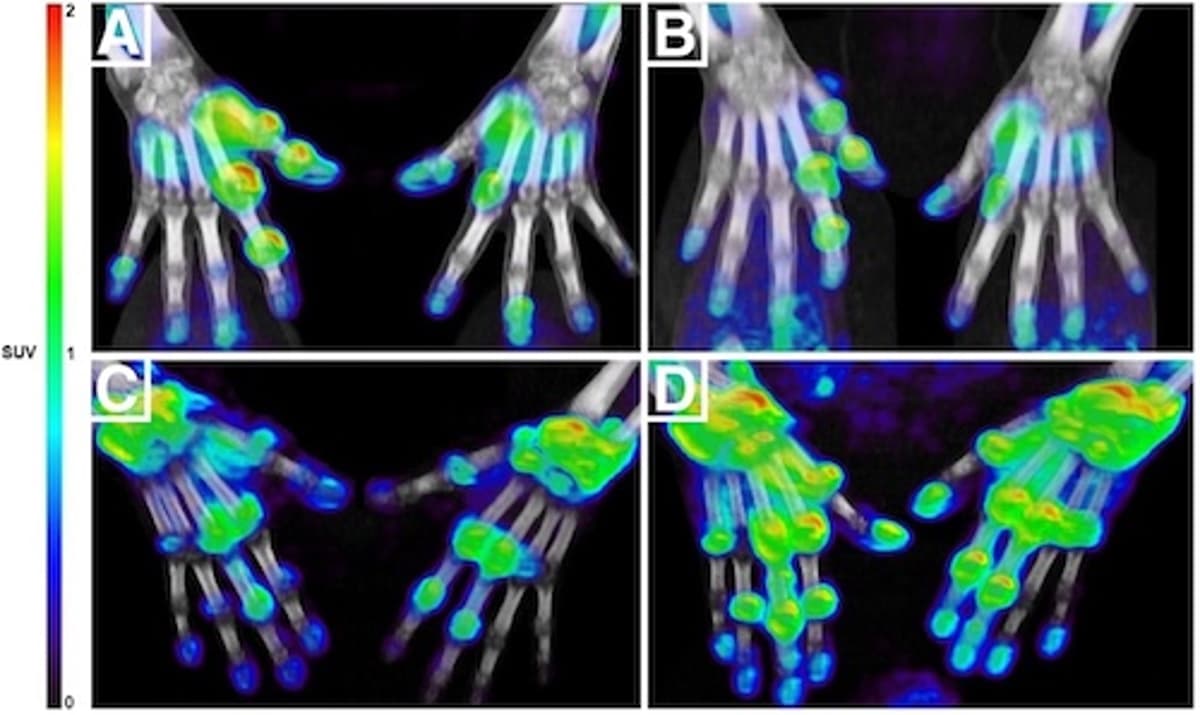
Targeted PET/CT Tracer Helps Rheumatoid Arthritis Patients
A new macrophage‑targeted PET/CT tracer (11C‑DPA‑713) can identify rheumatoid arthritis patients who will respond to anti‑TNF therapy within four weeks, far earlier than the traditional three‑to‑six‑month window. The study of 20 patients showed that standardized uptake values (SUVs) at baseline...
By Imaging Technology News (ITN)

News•Feb 26, 2026
Positrigo Launches BrainPET Accelerator Program in U.S.
Positrigo, a Swiss developer of nuclear medical devices, has launched the BrainPET Accelerator Program in the United States, offering neurology practices a ready‑to‑operate brain PET solution. The program bundles a compact NeuroLF scanner, radiotracer logistics, regulatory assistance, reimbursement support, and...
By Imaging Technology News (ITN)
News•Feb 26, 2026
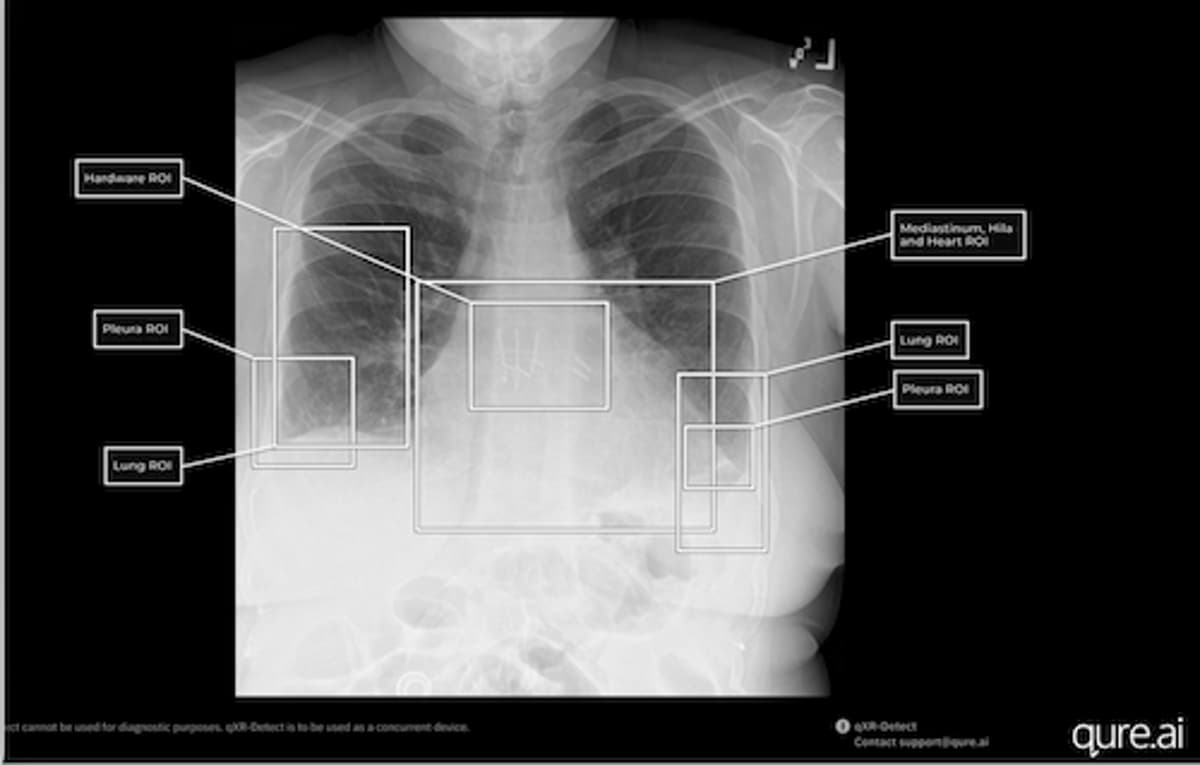
FDA Clears Six Indications for Qure.ai's Chest X-Ray Reporting Tool
The FDA granted 510(k) Class II clearance to Qure.ai’s qXR-Detect, adding six new chest‑X‑ray detection indications. The tool now flags findings across lung, pleura, mediastinum/hila & heart, bone, hardware and other regions, expanding the company’s U.S. portfolio to 26 FDA clearances. The clearance...
By Imaging Technology News (ITN)
