Recent Posts
News•Feb 25, 2026
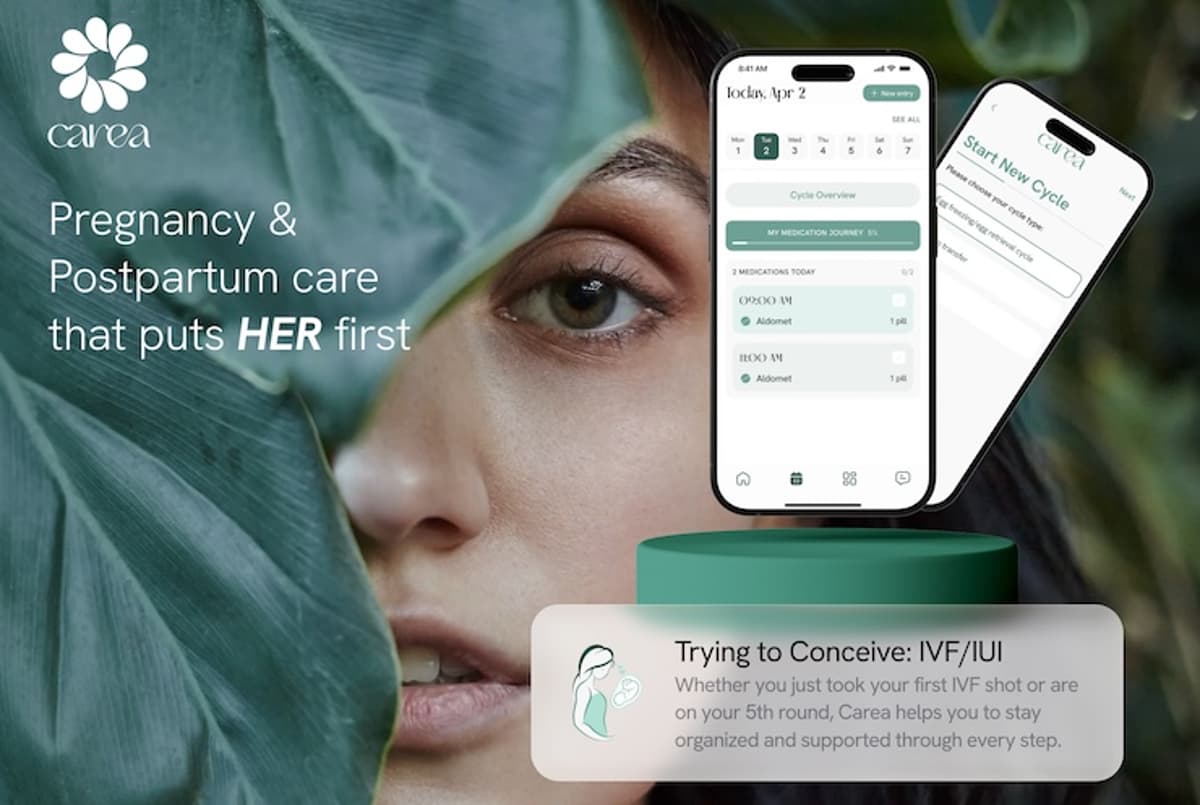
Carea Launches Support Platform for Complex Fertility Treatment
Carea has introduced a new "Trying to Conceive: IVF/IUI" mode within its pregnancy and postnatal wellbeing app, offering a day‑to‑day support system for women undergoing fertility treatment. The feature includes a personalised medication tracker, video guides, reminders, and a visual progress bar to keep users on schedule. Expert‑led articles, podcasts and affirmations provide emotional reassurance throughout IVF, IUI or egg‑freezing cycles. The mode launches free of charge, expanding Carea’s suite that already covers pregnancy, postpartum and, soon, loss recovery.
By Med-Tech Insights

News•Feb 24, 2026
Medi-Globe Launches mAI Companion AI Assistant for Pancreatic EUS
Medical device maker Medi‑Globe has launched mAI Companion, a CE‑marked real‑time AI assistant for pancreatic endoscopic ultrasound (EUS). Developed with IHU Strasbourg, the system analyses the pancreas during procedures to highlight solid and cystic lesions, acting as a second set...
By Med-Tech Insights

News•Feb 24, 2026
MiraDx Launches New PROSTOX Genetic Test for Prostate Cancer Patients
MiraDx has launched PROSTOX Standard, a clinically validated genetic test that predicts long‑term urinary toxicity for prostate cancer patients receiving conventional or moderately hypofractionated radiation therapy. The test uses a cheek swab and returns results in five to seven days,...
By Med-Tech Insights

News•Feb 24, 2026
FDA Grants Breakthrough Designation to Orthonika’s Synthetic Total Meniscus Implant
Orthonika, an Imperial College London spin‑out, received FDA Breakthrough Device Designation for its synthetic Total Meniscus Replacement (TMR) implant and an invitation to the Total Product Life Cycle Advisory Program. The designation promises prioritized regulatory engagement, speeding the path to...
By Med-Tech Insights

News•Feb 23, 2026
Mental Health Patients in Wrexham First to Benefit From New Digital Solution
Mental health wards at Wrexham Maelor Hospital have gone live with electronic Prescribing and Medicines Administration (ePMA), the first in Wales to also record discharge medicines in the Shared Medicines Record (SMR). The early‑adopter rollout, led by Betsi Cadwaladr University...
By Med-Tech Insights

News•Feb 23, 2026
BVI Completes First U.S. Implantations of FDA-Approved FINEVISION HP Trifocal IOL
BVI announced the first successful U.S. implantations of its FDA‑approved FINEVISION HP hydrophobic trifocal intraocular lens. Five leading cataract and refractive surgeons performed the procedures across multiple surgical centers. The lens, cleared by the FDA in October 2025, incorporates BVI’s proprietary POD...
By Med-Tech Insights

News•Feb 19, 2026
FDA Clearance Sets Stage for U.S. Commercialisation of Eyonis LCS
Median Technologies has named veteran imaging executive Oran Muduroglu as President of its U.S. subsidiary, Median eyonis Inc., to spearhead the commercial launch of eyonis LCS, an AI‑powered lung‑cancer‑screening SaMD that recently received FDA 510(k) clearance. The rollout will leverage a defined Medicare reimbursement...
By Med-Tech Insights
News•Feb 19, 2026
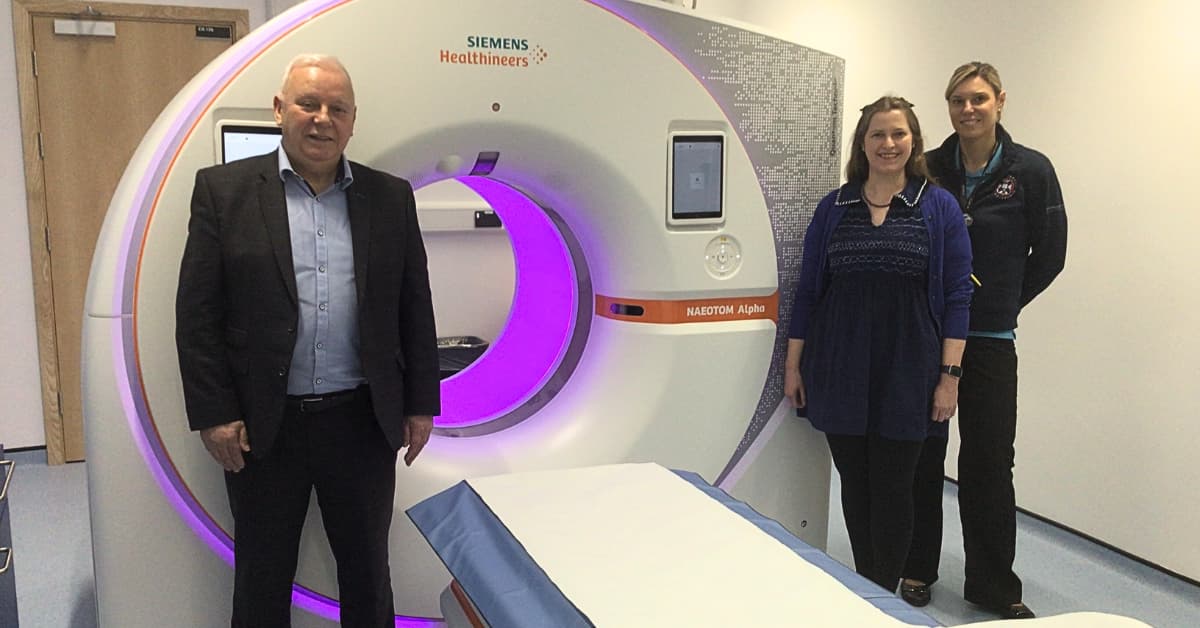
Scotland’s First Photon-Counting CT Scanner Set to Advance Multi-Organ Research and Enhance Patient Diagnosis & Care
The University of Edinburgh has installed Scotland’s first photon‑counting CT scanner, the Siemens Healthineers NAEOTOM Alpha, funded jointly with the British Heart Foundation. This technology captures each X‑ray photon, delivering ultra‑high‑resolution, spectral images that surpass conventional CT capabilities. It will...
By Med-Tech Insights

News•Feb 19, 2026
Osaka Hospital Launches Project to Safely Utilise Generative AI
Japan Community Healthcare Organization Osaka Hospital has partnered with Fujitsu Japan, Fortience Consulting and Microsoft Japan to launch a generative‑AI programme across all medical operations. The pilot, slated for June 2026, will automate roughly 16,000 discharge summaries and support nursing...
By Med-Tech Insights

News•Feb 19, 2026
UK Medical Device Testing Hits Record High as MHRA Backs Growth in Brain and AI Technology
The UK Medicines and Healthcare products Regulatory Agency reported a 17% rise in approved clinical investigations for 2025, setting a record high. Average approval times fell to 51 days, outpacing the 60‑day target, while neurotechnology now accounts for roughly a...
By Med-Tech Insights

News•Feb 18, 2026
Making Global Market Access Practical – How Medilink North of England Supports International Growth
Medilink North of England provides a structured market‑access offering that helps MedTech and digital‑health innovators move beyond regulatory clearance to achieve commercial adoption in overseas health systems. The service is built around five pillars—market segmentation, global strategy, regulatory documentation, reimbursement...
By Med-Tech Insights

News•Feb 17, 2026
BIOTRONIK Launches of ‘World’s First’ CRT-D Systems Approved for Conduction System Pacing
BIOTRONIK has introduced the Acticor Sky and Rivacor Sky family, the world’s first CE‑approved high‑voltage devices capable of left bundle branch area pacing (LBBAP). The inaugural European implant was performed at University Hospital Frankfurt in an 87‑year‑old with ischemic cardiomyopathy and atrial...
By Med-Tech Insights

News•Feb 17, 2026
Patchwork Health Launches AI-Powered ‘Preference-Based Rostering’ for NHS Clinicians
Patchwork Health has launched an AI‑driven Preference‑Based Rostering tool for NHS Trusts, instantly converting clinicians' shift preferences and service demand into compliant, fair schedules. The platform claims to meet 98% of negative preferences, cut unfilled shifts by 97% and slash...
By Med-Tech Insights

News•Feb 17, 2026
Tanaka Establishes Total Solutions System for Contract Manufacturing of Diagnostics
Tanaka Precious Metal Technologies has built a total‑solution system for contract manufacturing of in‑vitro diagnostic test kits, adding dedicated dispensing and packaging lines for extraction buffer. The new infrastructure lets the company handle every step—from assay development to final product...
By Med-Tech Insights
