Weekly Reads: Support Brain Tumor Work, Prasad Is Out (Again), Genetic Conditions, Texas AG, Immunotherapy Paper, SCBEM
•March 8, 2026
0
Key Takeaways
- •Funding gaps threaten brain tumor research progress
- •FDA's Prasad exit may stabilize biologics approvals
- •Texas AG sues unproven cord‑blood bank
- •Human embryo models raise ethical dilemmas
Summary
The newsletter urges donations to support a lab studying lethal childhood glioma, noting NIH grant success rates of only 4‑5%. It reports FDA biologics chief Vinay Prasad’s second departure, a rare leadership turnover that could affect approval stability. Additional highlights include the Texas Attorney General’s lawsuit against a cord‑blood banking firm, ethical concerns surrounding human embryo models, and growing hype around health‑boosting peptides. The piece also flags a contested study on morning‑time cancer immunotherapy.
Pulse Analysis
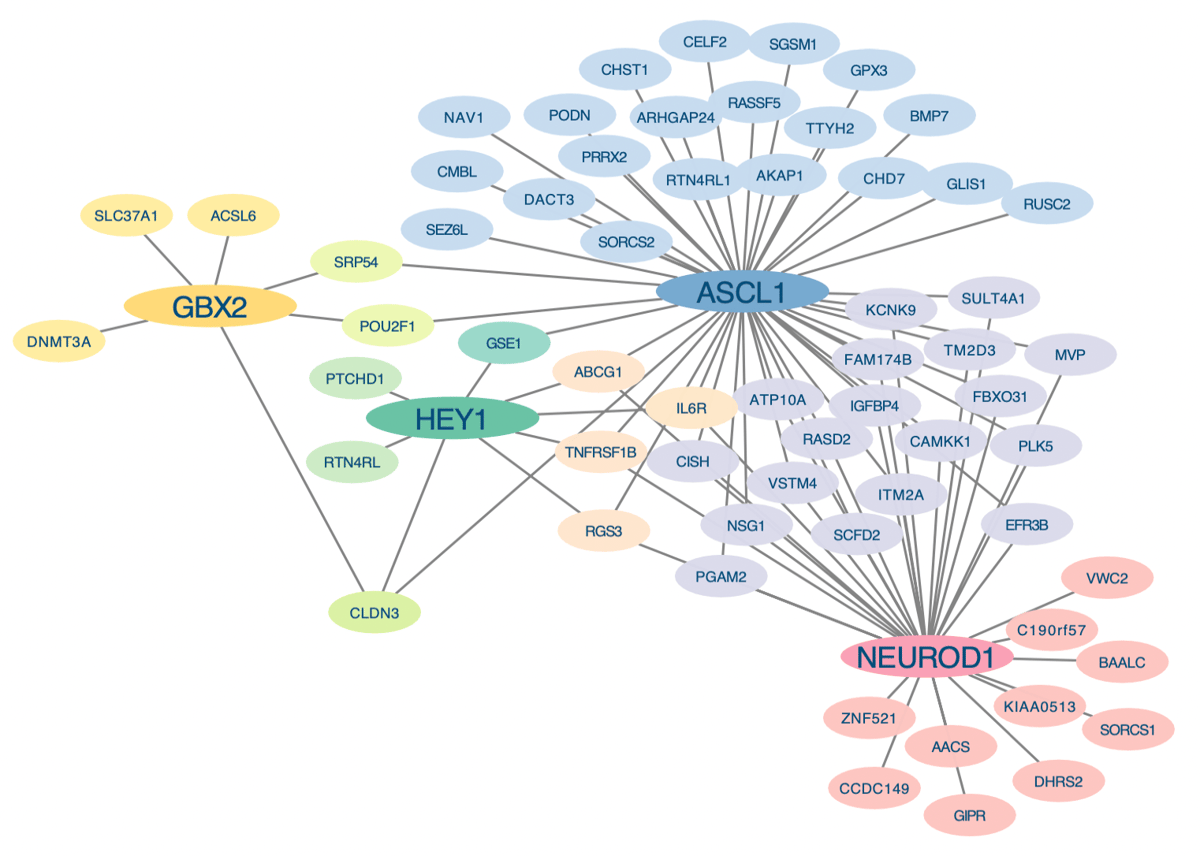
The financing landscape for brain tumor research has grown increasingly precarious, with NIH institutes approving merely 4‑5% of applications. This scarcity forces investigators to lean on private philanthropy, especially for pediatric glioma projects where mortality approaches 100% despite aggressive treatment. Leveraging donor contributions can accelerate discovery of novel targets such as the developmental transcription factor ASCL1, which recent work suggests drives tumor aggressiveness. Sustained funding is essential to translate these molecular insights into clinical trials that could finally offer hope to affected families.
Vinay Prasad’s announced exit from the FDA’s Center for Biologics Evaluation and Research marks a second, unprecedented turnover in a short span. His controversial tenure has been linked to heightened scrutiny of vaccine and rare‑disease approvals, creating uncertainty for biotech firms seeking market entry. A more predictable leadership could restore confidence among developers of cell‑based therapies and gene‑editing products, potentially smoothing the pathway for innovative biologics that address unmet medical needs. Stakeholders are watching closely to gauge whether regulatory stability will improve under new stewardship.
Beyond funding and regulation, the newsletter spotlights several emerging controversies. The Texas Attorney General’s lawsuit against a cord‑blood banking company underscores growing legal pressure on unproven stem‑cell clinics, while ethical debates intensify around human embryo models (SCBEM) that increasingly mimic natural development. Simultaneously, the peptide supplement market is exploding, prompting calls for rigorous scientific evaluation to curb misinformation. Finally, doubts about a study linking immunotherapy timing to outcomes highlight the need for reproducible research before clinical practice changes. Together, these issues illustrate the complex interplay of science, policy, and public perception shaping the biotech ecosystem.
Comments
Want to join the conversation?