
Why Breakthrough Cell and Gene Therapies Still Fail at the Finish Line
Nearly half of cell and gene therapy submissions now receive FDA complete response letters, with 42% flagged for deficiencies. The analysis shows that most of these CRLs arise from manufacturing, chemistry, and controls (CMC) issues rather than safety concerns. Shifts in assay methods, late‑stage process changes, and overly broad quality specifications create regulatory gaps that are hard to bridge after pivotal trials. Addressing manufacturing discipline early—by generating bridging data, finalizing processes before Phase II, and aligning specifications with clinical batch performance—can reduce delays and costs.

Photosynthes-Eyes: Spinach-Based Therapy Offers Hope for Dry Eyes
Scientists at the National University of Singapore have engineered a nanosized eye‑drop called LEAF, derived from spinach thylakoid membranes, that generates NADPH when exposed to ambient light. The 400 nm particles boost antioxidant production, rapidly normalising reactive oxygen species in cultured...

ASCO26 HARMONi6 Updated – Plaudits, Pitfalls, and Pratfalls
Akeso Bio presented updated interim results from its HARMONi‑6 trial at ASCO 2026, eight months after the initial ESMO data. The new dataset, with longer follow‑up, revealed a modest overall response rate and emerging safety signals that fell short of...

Tempus Unveils the Next-Generation of Lens, Expanding Its Agentic AI Platform for Oncology Drug Development
Tempus AI launched the next‑generation Lens, an agentic AI platform that unifies its multimodal real‑world oncology data, high‑performance computing and validated AI agents. The cloud‑based system lets drug developers create research plans in plain language, execute analyses on more than...
Pfizer’s BRAFTOVI Regimen Nearly Doubles Median Progression-Free Survival in Metastatic Colorectal Cancer
Pfizer reported that its BRAFTOVI (encorafenib) regimen combined with cetuximab and FOLFIRI halved the risk of disease progression or death in patients with BRAF V600E‑mutant metastatic colorectal cancer. Median progression‑free survival more than doubled to 15.2 months versus 8.3 months for...

Kelonia Therapeutics Presents Updated First-in-Human Data From Phase 1 inMMyCAR Study of KLN-1010 in Vivo BCMA CAR-T Therapy at the...
Kelonia Therapeutics reported updated Phase 1 inMMyCAR data for its in‑vivo BCMA CAR‑T candidate KLN‑1010 at ASCO 2026. All 18 patients dosed showed a 100% overall response rate with MRD‑negative bone marrow, and the earliest‑treated patient remains disease‑free beyond ten months....

On KRAS Inhibitors and Why Potency Doesn’t Equal Durability
The latest ASCO data on second‑generation KRAS inhibitors reveal that higher potency does not automatically translate into longer patient responses. While first‑gen G12C agents such as sotorasib showed modest durability, new G12D compounds demonstrate improved binding but still face resistance...

Retatrutide Is Bringing a Hard Look at Health Outcomes
Eli Lilly’s phase‑3 TRIUMPH‑1 trial showed retatrutide delivering unprecedented weight loss, with participants on the top dose shedding an average of 28.3% of body weight after 80 weeks and 45.3% achieving at least a 30% reduction. In a two‑year extension...
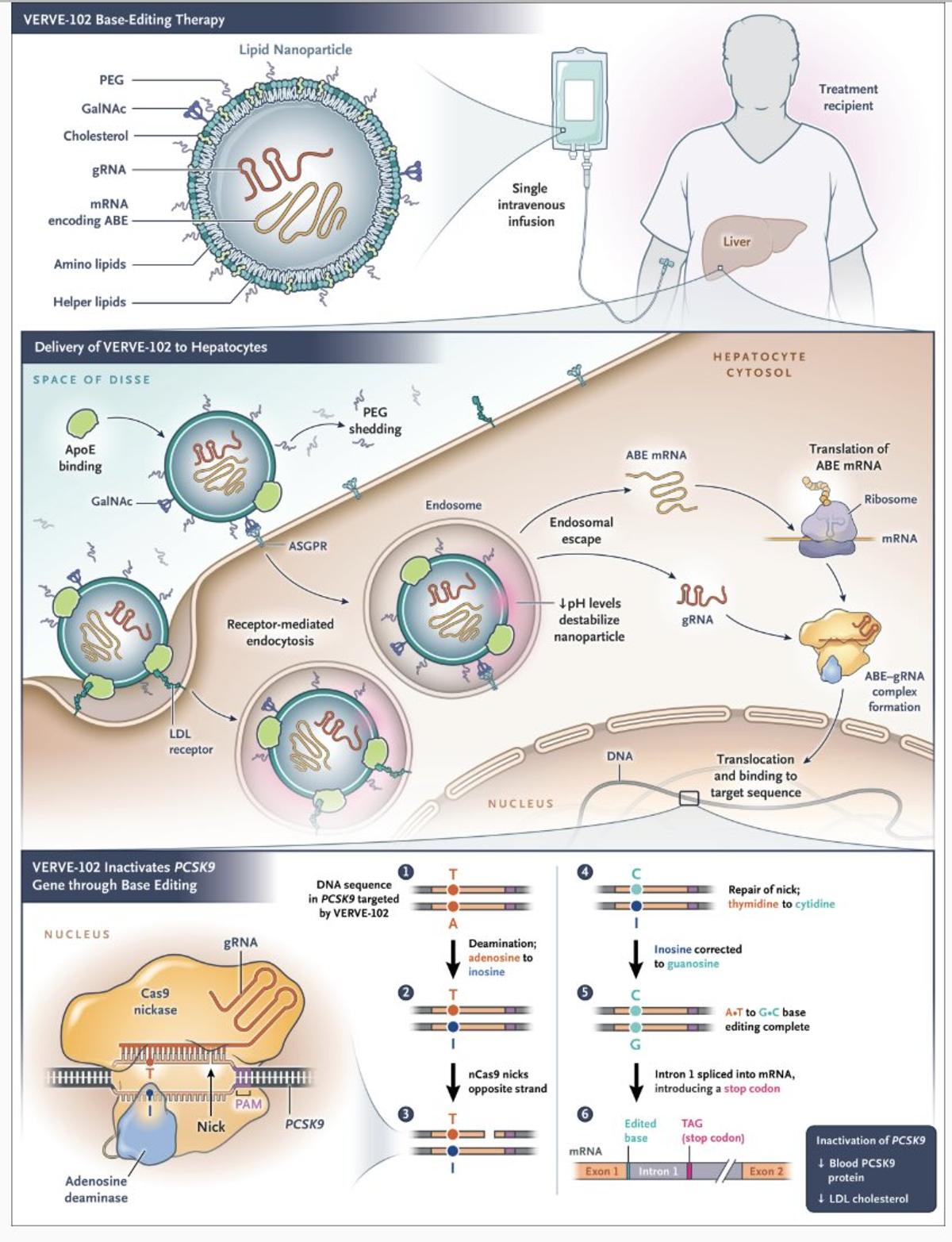
Weekly Reads: Effective LDL Gene Editing, Chinese Genetics Guidelines, Human Embryo Models in Space
A single‑infusion base‑editing drug (VERVE‑102) lowered LDL cholesterol in a small human study, sparking talk of a potential one‑time cure for high cholesterol. Parallel research unveiled universal transcriptomic aging clocks that can forecast mortality, advancing precision longevity metrics. Meanwhile, China...
ASCO 2026: Bayer Shares New Findings for NUBEQA® (Darolutamide) Regarding Cognitive Decline in Men with Advanced Prostate Cancer Compared to...
Bayer presented Phase II ARACOG trial data at ASCO 2026 showing that NUBEQA® (darolutamide) significantly limited cognitive decline in men with advanced prostate cancer compared with enzalutamide. Over 24 weeks, the median maximal cognitive domain change was –15.8% for darolutamide versus...
Salubris Biotherapeutics Announces Updated Phase 1/2 Data for JK06, a 5T4-Targeted Antibody Drug Conjugate, at the 2026 American Society of...
Salubris Biotherapeutics presented updated Phase 1/2 data for JK06, a 5T4‑targeted antibody‑drug conjugate, at the 2026 ASCO meeting. The study treated 173 patients across dose‑escalation and expansion cohorts, reporting a 50% objective response rate (ORR) in squamous non‑small cell lung cancer...

Rapamycin Signalling Profile on THP-1
A 24‑hour, 10 µM rapamycin treatment of the THP‑1 monocytic leukemia cell line triggers a complex transcriptional rewiring. Negative feedback loops in the PI3K/AKT/mTOR axis are released, leading to up‑regulation of PIK3R1, AKT isoforms and mTORC1 scaffolds while FOXO3 mRNA is...
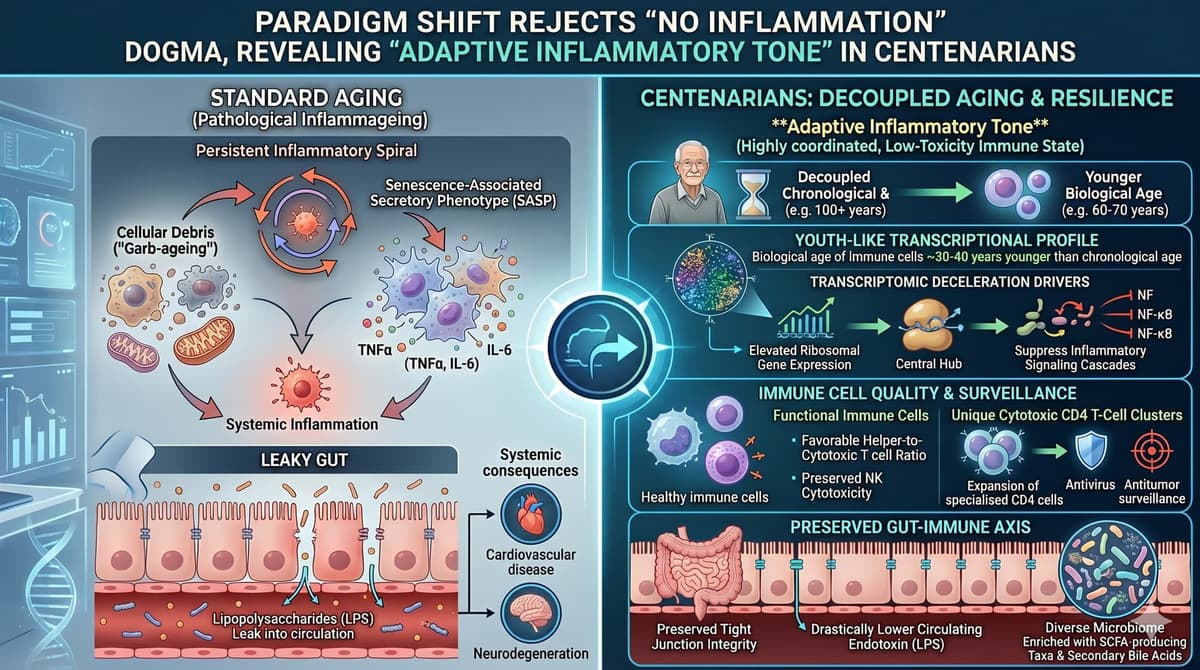
The Century-Old Immunome: Learning From the Adaptive Shield of Human Centenarians
The article outlines translational strategies to mimic centenarians’ elevated RNASEH2C activity, which clears cytoplasmic RNA:DNA hybrids and dampens chronic inflammation. It proposes four therapeutic levers: epigenetic maintenance to prevent RNASEH2C hyper‑methylation, delivery of centenarian‑derived extracellular vesicles, upstream protection of mitochondrial...
Exelixis Announces Results From Subgroup Analysis of Phase 3 CABINET Pivotal Trial Evaluating CABOMETYX® (Cabozantinib) in Non-Functional and Functional Neuroendocrine...
Exelixis presented a subgroup analysis of the phase 3 CABINET trial at ASCO 2026, showing that CABOMETYX (cabozantinib) significantly extends progression‑free survival (PFS) in advanced neuroendocrine tumors (NET) regardless of functional status. In non‑functional NET, median PFS was 9.4 months versus 3.1 months on...

Massive Bio Announces Next-Generation Reticulum Nexus™ Product Suite at ASCO 2026
Massive Bio unveiled the next‑generation Reticulum Nexus™ suite at ASCO 2026, integrating six AI‑driven agents—Patient Connect™, TrialRelay™, NexusPulse™ calculators, Sentinel Agents™, DrArturo AI™ and Phoebe AI™—into a real‑time orchestration layer for precision oncology. The platform moves patients from fragmented trial...