Mansoor Amiji: Rare Gem and Modern-Day Polymath
•January 21, 2026
0
Why It Matters
The juxtaposition of high‑profile honors with persistent research‑integrity issues threatens trust in translational nanomedicine and could influence funding, partnership decisions, and regulatory oversight.
Key Takeaways
- •Amiji elected National Academy of Inventors Fellow, Dec 2025.
- •31 of his papers flagged on PubPeer for image issues.
- •Animal studies exceeded NIH humane tumor size guidelines.
- •Over 25 pharma collaborations and multiple biotech co‑founder roles.
- •Corrections issued, yet data integrity concerns remain unresolved.
Pulse Analysis
The National Academy of Inventors typically celebrates scientists whose work translates into tangible health solutions, and Amiji’s election underscores his prolific output in vaccine delivery and oncology nanocarriers. His portfolio spans oil‑droplet emulsions, polymeric nanoparticles, and targeted gene‑therapy platforms, earning multiple patents and industry partnerships. However, the accolade arrives amid a growing chorus of post‑publication scrutiny, highlighting how prestigious recognitions can sometimes outpace rigorous validation of underlying data.
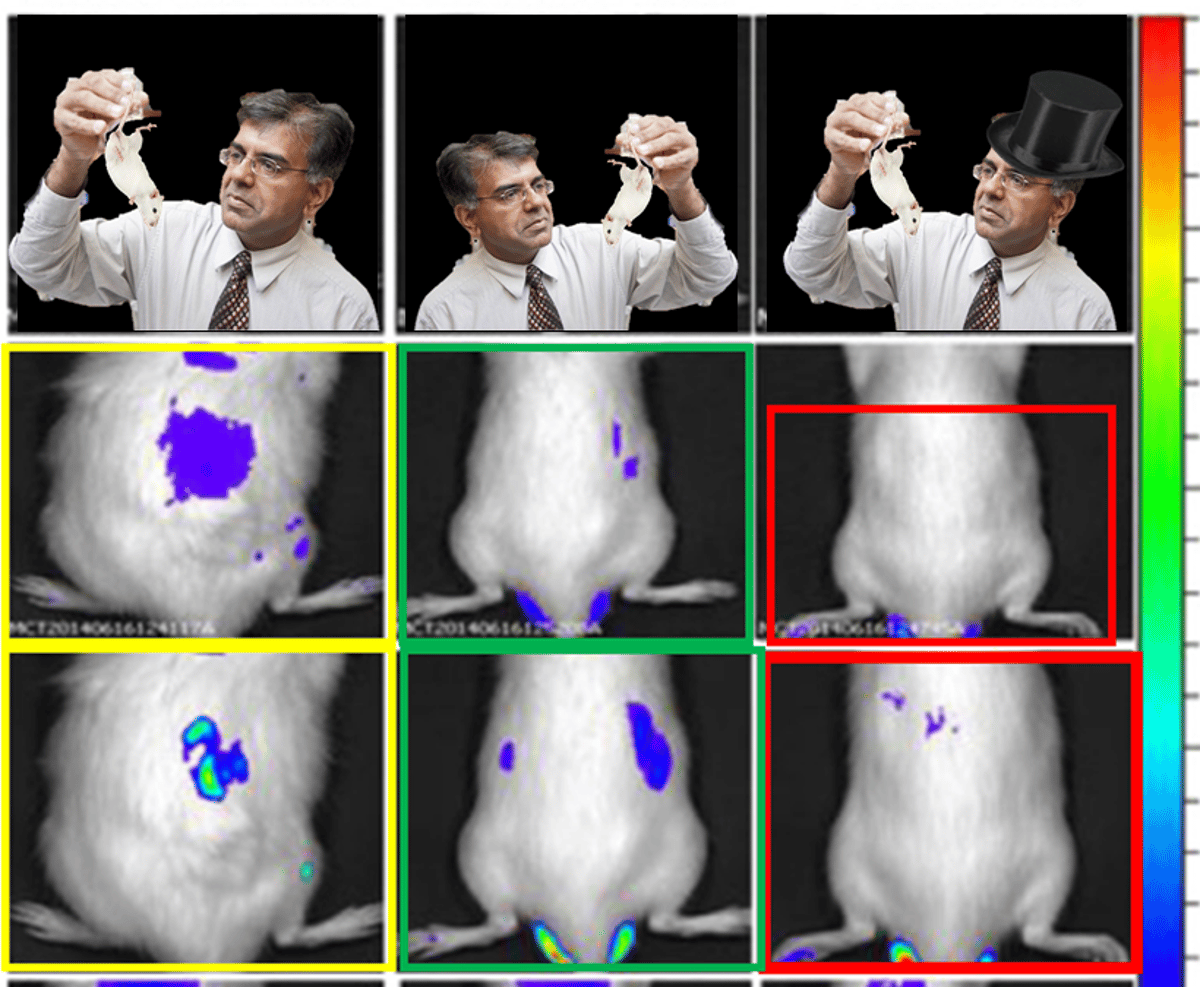
A systematic audit of Amiji’s record uncovers 31 PubPeer entries flagging duplicated or potentially manipulated images across high‑impact journals. Critics also point to animal‑ethics lapses, with tumor dimensions in mouse models surpassing NIH humane endpoints, suggesting possible non‑compliance with IACUC standards. Coupled with undisclosed advisory board positions and co‑founder roles at firms like Nemucore Medical Innovations, these issues raise red flags about conflict‑of‑interest management and the reproducibility of his preclinical claims.
The broader lesson for the biotech ecosystem is clear: accolades alone cannot substitute for transparent, reproducible science. Universities, funding agencies, and industry collaborators must enforce stricter oversight, demand timely corrigenda, and require full disclosure of financial ties. For investors and regulators, a balanced assessment of both scientific merit and integrity safeguards is essential to ensure that promising nanomedicine platforms advance without compromising public trust.
Mansoor Amiji: rare gem and modern-day polymath
0
Comments
Want to join the conversation?
Loading comments...