New Technique Lights up Where Drugs Go in the Body, Cell by Cell
•January 2, 2026
0
Why It Matters
Cell‑by‑cell drug localization transforms safety assessment, allowing developers to spot off‑target interactions before costly clinical failures. This capability could accelerate the design of more precise, lower‑risk therapeutics.
Key Takeaways
- •vCATCH visualizes covalent drug binding at single‑cell resolution
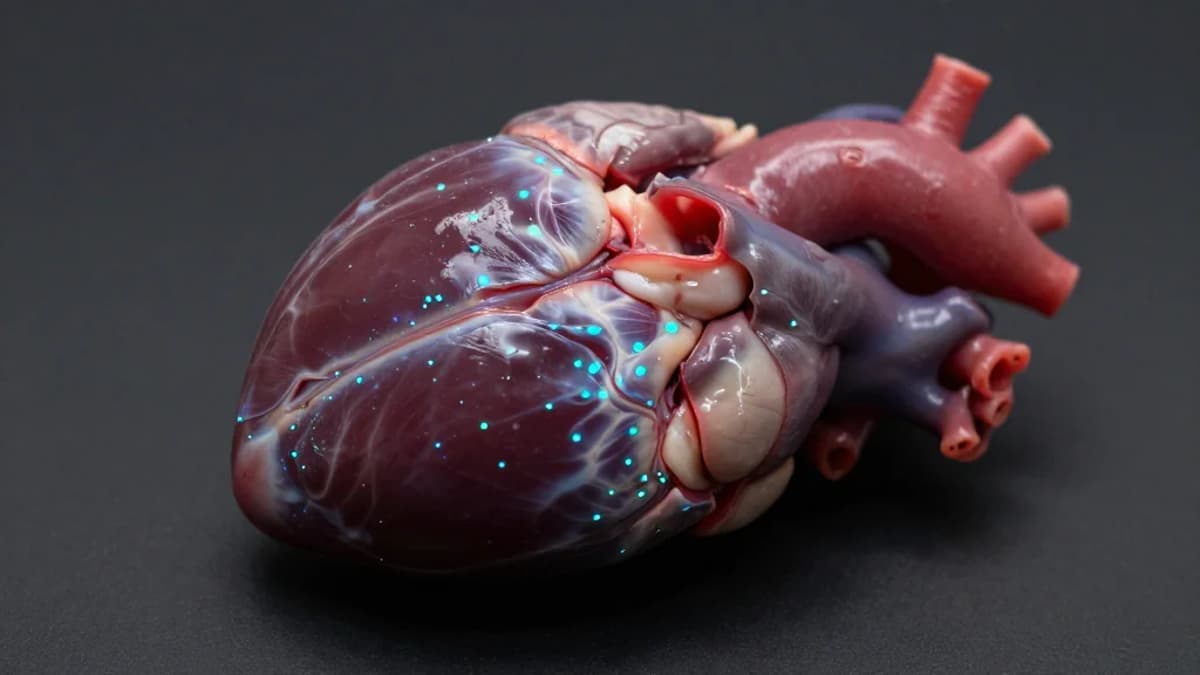
- •ibrutinib binds heart and liver immune cells, explaining side effects
- •Technique overcomes copper sequestration using pre‑treatment and multiple cycles
- •AI pipelines handle terabytes of whole‑body imaging data
- •Enables early safety screening for late‑stage drug candidates
Pulse Analysis
Traditional pharmacokinetic studies tell us which organs a drug reaches, but they lack the granularity to identify the exact cells involved. vCATCH bridges that gap by coupling covalent drug chemistry with a copper‑catalyzed click reaction that affixes a bright fluorescent tag to the drug’s molecular handle. After systemic injection, tissues are harvested, pre‑treated with excess copper to neutralize competing proteins, and cycled through multiple tagging steps, producing high‑contrast, cell‑specific signals even in deep structures like the brain and heart.
When the researchers mapped two FDA‑approved covalent cancer agents, the technology confirmed expected distribution for afatinib in lung tissue while exposing a surprising off‑target pattern for ibrutinib. The drug accumulated in cardiac muscle cells and liver immune populations, correlating with its known arrhythmia and bleeding risks. By integrating AI‑driven image analysis, the team could parse terabytes of three‑dimensional data, automatically cataloguing drug‑bound cells across the entire mouse anatomy. This level of insight, previously unattainable, equips scientists to link cellular binding profiles directly to adverse events.
Beyond oncology, vCATCH promises to reshape late‑stage drug development across therapeutic areas. Pharma companies can now screen candidate molecules for unintended tissue engagement before human trials, potentially reducing attrition rates and regulatory hurdles. The method also opens avenues for studying neuropsychiatric drugs, where cell‑type specificity in the brain is critical, and for personalizing treatments based on patient‑specific cellular landscapes. As the industry seeks more predictive preclinical tools, vCATCH stands out as a scalable, high‑resolution solution that could become a new standard in safety pharmacology.
New technique lights up where drugs go in the body, cell by cell
0
Comments
Want to join the conversation?
Loading comments...