Simple At-Home Tests Strips Can Detect Cat and Dog Viruses
•February 9, 2026
0
Why It Matters
Early, accurate detection of FPV and CPV can curb mortality and reduce veterinary visits, while empowering owners with rapid, home‑based testing. The technology also opens a pathway for broader at‑home pet disease diagnostics.
Key Takeaways
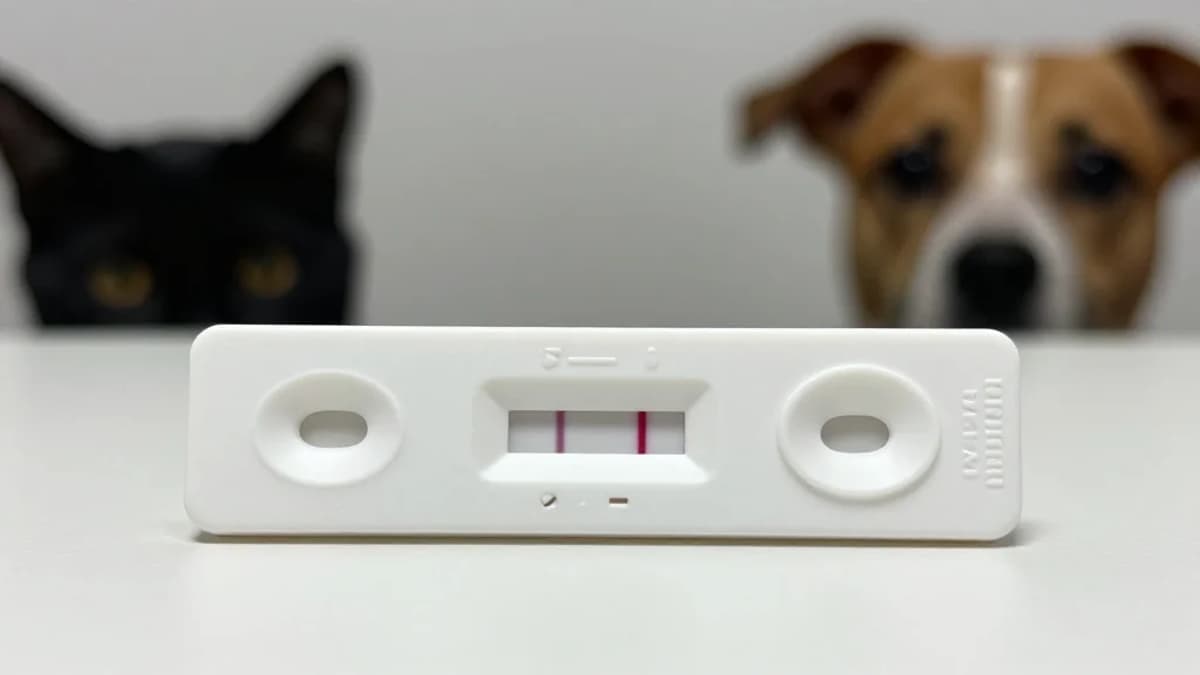
- •100% sensitivity on clinic samples for FPV and CPV
- •Test completes in ~35 minutes using hand‑warm vial
- •Dual strip detects FPV and feline herpesvirus simultaneously
- •Outperforms commercial immunoassays missing positive cases
- •DNA‑based strip works at body temperature, suitable for home
Pulse Analysis
Pet health monitoring has long relied on veterinary labs, which can delay treatment for fast‑acting viruses like feline and canine parvovirus. Traditional at‑home kits focus on protein detection and often suffer from low sensitivity, leading to false negatives that jeopardize animal welfare. As pet ownership rises, the market demands faster, more reliable diagnostics that can be administered without professional equipment. The emergence of nucleic‑acid‑based lateral‑flow tests addresses this gap, offering laboratory‑grade accuracy in a format familiar to consumers from COVID‑19 home tests.
The new assay leverages a two‑step DNA amplification of the VP2 gene, a region unique to FPV and CPV, within a vial warmed by a human hand. After roughly 35 minutes, the amplified product is applied to a strip that produces a visible red line when viral DNA is present. Validation on 14 feline and 38 canine clinical samples showed perfect concordance with laboratory PCR, while a competing commercial immunoassay missed several positives. A dual‑strip variant further demonstrated the capacity to detect feline herpesvirus, highlighting the platform's flexibility for multiplexed testing.
Beyond immediate clinical benefits, this technology could reshape the pet care ecosystem. Veterinarians may adopt a triage model where owners perform preliminary screening at home, reserving in‑clinic visits for confirmed cases. Manufacturers can expand the platform to cover other common pathogens, creating a suite of at‑home diagnostics akin to human health kits. As regulatory pathways for veterinary point‑of‑care devices evolve, the market potential for accurate, affordable home testing is poised for rapid growth, promising better outcomes for pets and peace of mind for owners.
Simple at-home tests strips can detect cat and dog viruses
0
Comments
Want to join the conversation?
Loading comments...