
Standing Ovation for Daraxonrasib Survival Benefit at ASCO26
Incredible #ASCO26 moment. Dr. Brian Wolpin, presenter of the daraxonrasib study, received a standing ovation DURING his talk after he stated the survival benefit for PDAC patients. It was sustained. Cheering. I have never see anything like it in the middle of a talk. $RVMD https://t.co/I2wQfDqsvh
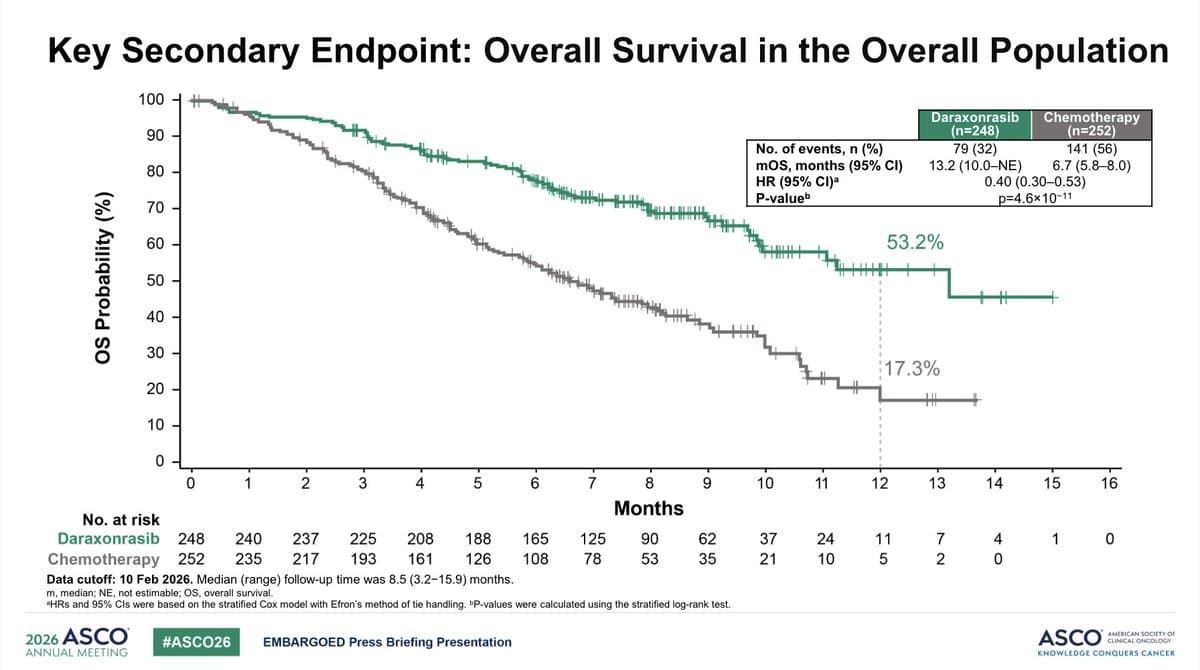
New OS Data Reveal Daraxonrasib
A warm #ASCO26 welcome to daraxonrasib and RASolute-302 study in 2nd line metPDAC from $RVMD My story in the post below. Here are the OS curves you haven't seen yet. https://t.co/VoNSu8BH3f

ASCO Day 1: Positive Myeloma and Lung Cancer Data
#ASCO26 Day 1. We're ASCOmaxxing. Positive data for Bristol $BMY in multiple myeloma and Pfizer $PFE in lung cancer. The FDA will surely convene an adcomm for $REPL, right? And Dizal's good day/bad day. https://t.co/NiENYMHswv via @statnews

FDA Cancer Office, Not CBER, Blocked RP1 Approval
Whether or not you believe $REPL RP1 deserves to be approved or not, let's please be truthful about what has transpired so far: Richard Pazdur and others in the FDA's cancer office were responsible for RP1's initial rejection. They objected...

Lilly's Verve Gene Editor Cuts Cholesterol, but Delays Loom
Eli Lilly $LLY says Verve’s gene editor lowers cholesterol levels in early, but development timelines have slipped. https://t.co/fuJOPyDwQb via @Jasonmmast

Kelun's MRK ADC Shows Promise Over Akeso in Lung Cancer
The #ASCO26 abstracts are live. I wrote a story about the Kelun $MRK ADC sac-TMT, which looks very good in lung cancer, potentially better than Akeso / $SMMT 's ivonescimab https://t.co/exfmgabTLa

ASCO Abstracts Posted Tomorrow, Full Data Saved for Meeting
The #ASCO26 abstracts will be posted tomorrow at 5 pm EDT. What this means, and what is doesn't. -- No late-breaker abstracts tomorrow. You will not see the plenary session data from $RVMD, Akeso / $SMMT, $JNJ or $LLY....

FDA Review Highlights Single‑Arm Trial Approval Risks
$IBRX -- The FDA accepted the sBLA to expand Anktiva to include the treatment of MNBIC papillary. The PDUFA date is Jan. 6, 2027. Standard review. A positive development for ImmunityBio, but like all things, details matter. Approval...

AI Designs Entirely New Therapeutic Molecules, Says Ex‑Stanford President
Marc Tessier-Lavigne, CEO of Xaira Therapeutics, talking about using AI to make entirely novel (potentially) therapeutic molecules that do not exist in nature. Wow type stuff @matthewherper also asking about his tenure as Stanford president that was cut short (he resigned),after accusations...

FDA CDER Acting Director Tracy Hoeg Set to Leave
FDA Center for Drug Evaluation & Research (CDER) Acting Director Tracy Beth Hoeg expected to depart from agency - Reuters
Seeking Must-See ASCO26 Abstracts for May 21
Hey BioXers who pay attention to cancer: Are there data coming out on May 21 in #ASCO26 abstracts (not late breakers) that you deem must see? I'm trying to figure out what needs to be covered on May 21 when...

RGNX Shows 71% Microdystrophin, Eyes 2027 Approval
$RGNX Duchenne gene therapy study results are out: microdystrophin expression reached an average of 71% of normal across all 31 boys at 12 wks. Functional improvements in a subset of boys followed for one year also seen. The big question...

Marty Makary: The Worst FDA Commissioner in 25 Years
And here's @matthewherper on why Marty Makary was the worst FDA commissioner in 25 years https://t.co/a44yjB5sZn
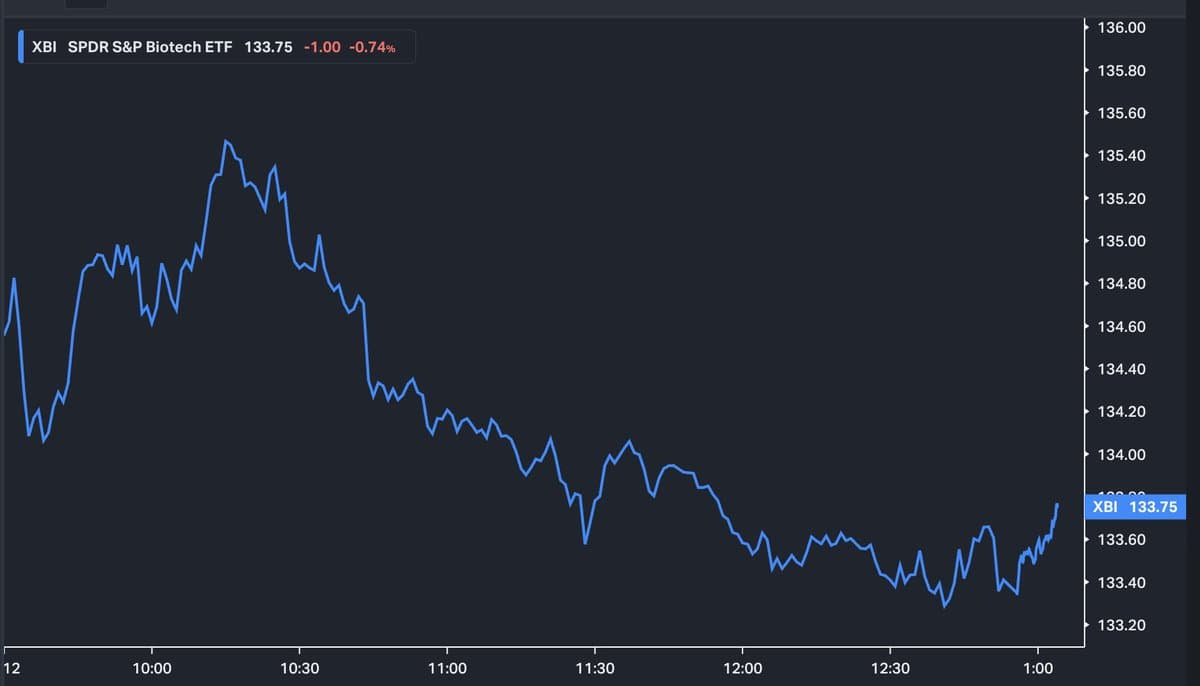
XBI Sees Modest Boost After Anticlimactic FDA Decision
$XBI getting small lift (so far) from the Un-Martying of the FDA. It's all very anticlimactic. https://t.co/q4ta0bkJh4
RIGL to Sell Approved Veppanu for ARVN, PFE
$RIGL will sell the newly approved Veppanu for $ARVN and $PFE. Modest financial terms https://t.co/9RsbSMzxBf