FDA Clears Six Indications for Qure.ai's Chest X-Ray Reporting Tool
•February 26, 2026
0
Why It Matters
The clearance gives U.S. hospitals a regulated AI solution that can improve early detection and workflow efficiency, potentially reducing diagnostic delays and costs. It also sets a regulatory benchmark for AI adaptability through the Predetermined Change Control Plan.
Key Takeaways
- •FDA grants 510(k) clearance for six chest X-ray indications
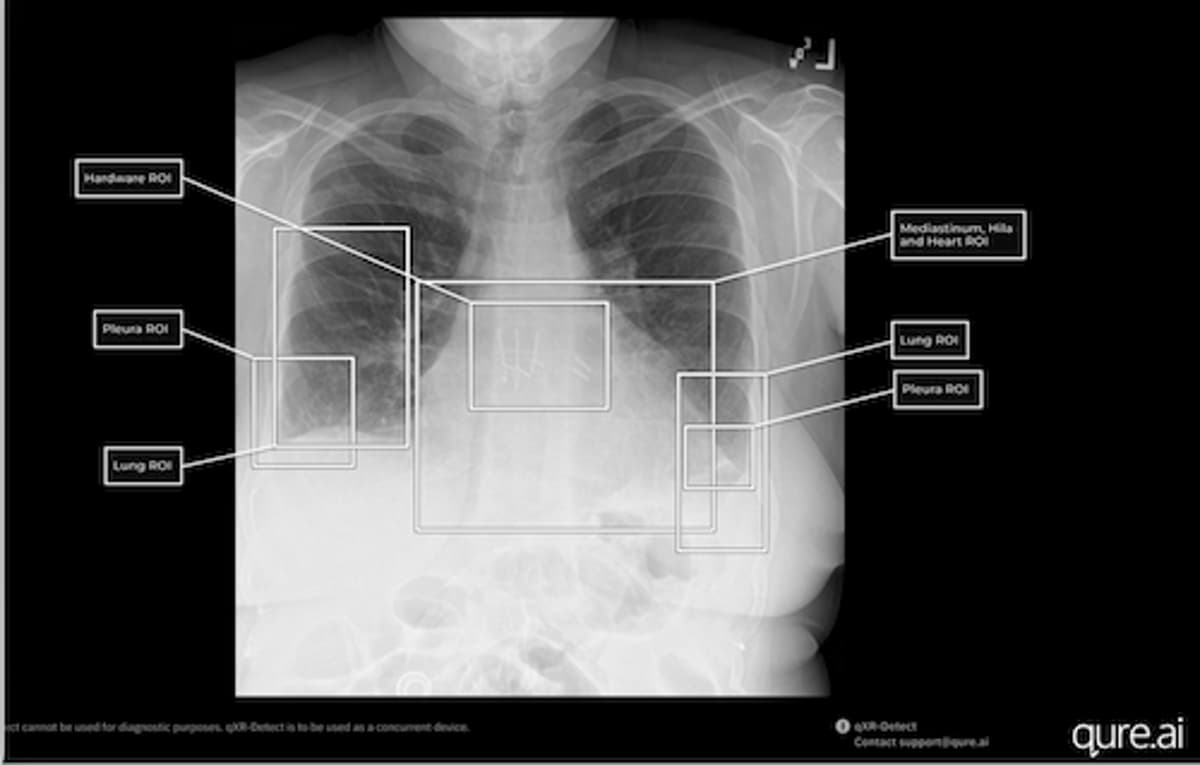
- •qXR-Detect covers lung, pleura, mediastinum, bone, hardware
- •AI provides localization with bounding boxes and ROI labels
- •Only chest CADe device with FDA Predetermined Change Control Plan
- •Qure.ai now holds 26 FDA clearances across X-ray and CT
Pulse Analysis
Chest radiography remains the most common imaging exam in the United States, with roughly 70 million studies performed annually across emergency departments, outpatient clinics, and routine health checks. As demand for rapid, accurate interpretation grows, AI‑driven computer‑assisted detection (CADe) tools have emerged as a practical solution to augment radiologists and frontline clinicians. By automatically highlighting abnormalities, these systems help prioritize cases, reduce oversight, and support earlier clinical interventions, especially in high‑volume settings where human fatigue can affect diagnostic accuracy.
Qure.ai’s recent FDA clearance of qXR-Detect marks a significant regulatory milestone. The six‑indication clearance expands the algorithm’s scope to six anatomical regions, delivering visual localization through bounding boxes and region‑of‑interest labels. Uniquely, qXR-Detect is the only chest‑X‑ray CADe device cleared with a Predetermined Change Control Plan, a framework that allows the manufacturer to roll out algorithmic updates without filing new submissions. This flexibility ensures the tool can evolve with emerging data, maintaining state‑of‑the‑art performance while preserving compliance—a competitive edge in a market where rapid innovation is essential.
For health systems, the cleared product promises tangible workflow benefits. Emergency physicians can receive instant triage alerts for critical findings such as pneumothorax or lung nodules, while radiologists gain a second set of eyes that highlights subtle pathologies often missed in busy practices. The integration of explainable AI—visual cues that clarify why an alert was generated—helps maintain clinician trust and facilitates faster decision‑making. As reimbursement models increasingly reward value‑based care, tools like qXR-Detect that improve diagnostic speed and accuracy are likely to see accelerated adoption, shaping the next generation of AI‑enhanced radiology services.
FDA Clears Six Indications for Qure.ai's Chest X-Ray Reporting Tool
0
Comments
Want to join the conversation?
Loading comments...