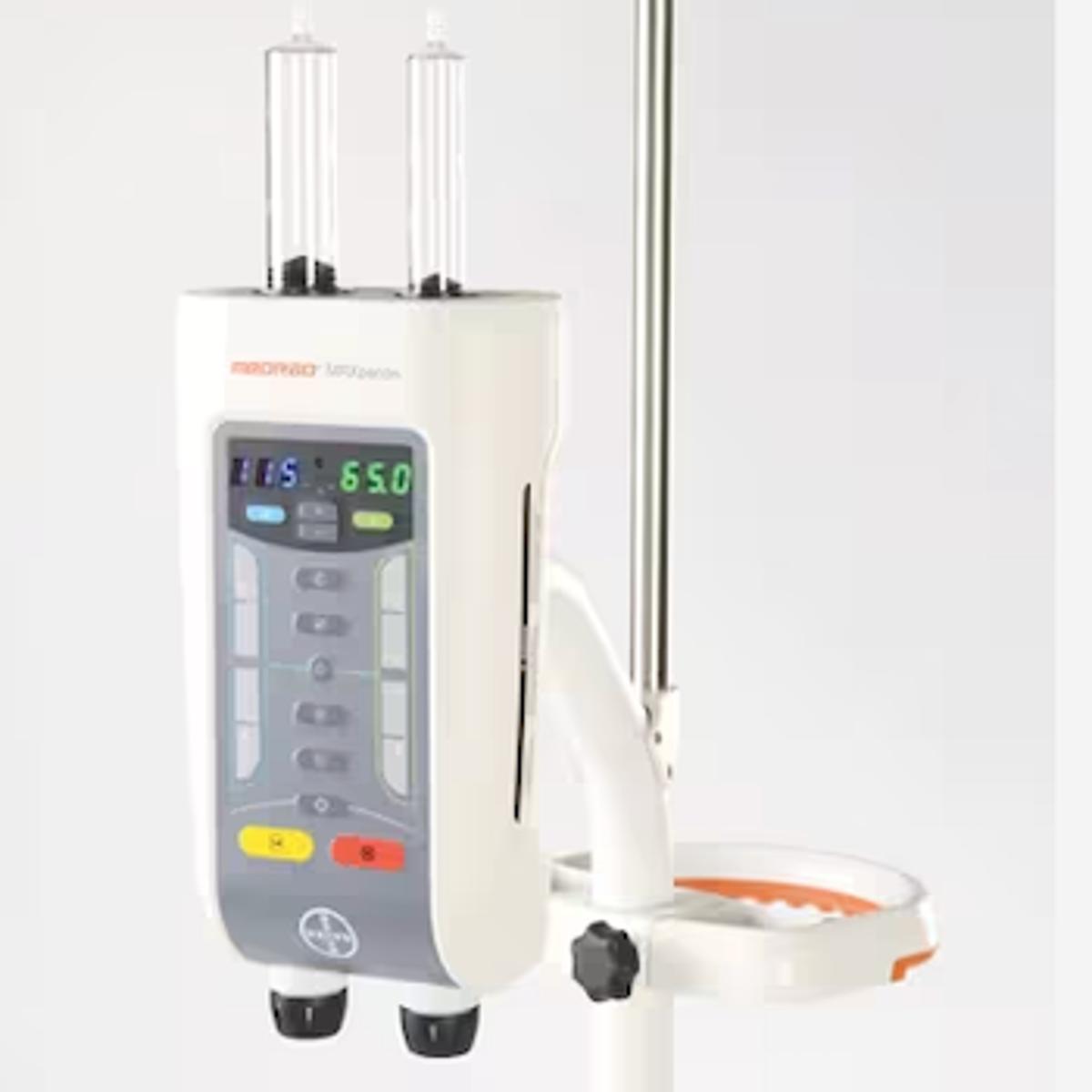
FDA Grants 510(k) Clearance for Expanded Capabilities of MR Injection System
•March 4, 2026
0
Why It Matters
The clearance positions Bayer to capture growing demand for ultra‑high‑field MRI, improving efficiency and expanding its market share in advanced imaging workflows.
Key Takeaways
- •FDA cleared MRXperion for up to 7‑Tesla MRI scanners
- •New ISI2 interface enables injector‑scanner communication
- •Supports both routine clinical and advanced research imaging
- •Enhances Bayer’s integrated MR contrast and workflow portfolio
- •Offers radiology departments greater flexibility across MR suite configurations
Pulse Analysis
The FDA’s 510(k) approval of Bayer’s MEDRAD MRXperion marks a pivotal step in the evolution of magnetic resonance technology. Ultra‑high‑field MRI systems, especially those operating at 7 Tesla, are gaining traction in academic hospitals and research centers for their superior signal‑to‑noise ratio and detailed tissue characterization. By extending MRXperion’s compatibility to this field strength, Bayer removes a key barrier that previously limited power injector use to lower‑field scanners, aligning hardware capabilities with the latest imaging hardware trends.
Beyond field strength, the introduction of Imaging Scanner Interface 2 (ISI2) creates a seamless data link between the injector and the MRI console. This integration automates contrast‑delivery timing, reduces manual entry errors, and synchronizes scan protocols, which translates into shorter exam times and higher patient throughput. For radiology departments juggling diverse scanner models, the ISI2 capability simplifies workflow orchestration, supporting both high‑volume clinical schedules and the precise timing required for advanced research studies such as functional MRI and spectroscopy.
Strategically, the clearance strengthens Bayer’s end‑to‑end MR offering, complementing its contrast agents and workflow software. Competitors must now address the combined demand for ultra‑high‑field compatibility and intelligent injector‑scanner communication to stay competitive. As hospitals invest in 7 T platforms to differentiate their diagnostic services, Bayer’s expanded MRXperion is poised to become a preferred choice, potentially driving higher market penetration and reinforcing the company’s leadership in the radiology solutions space.
FDA Grants 510(k) Clearance for Expanded Capabilities of MR Injection System
0
Comments
Want to join the conversation?
Loading comments...