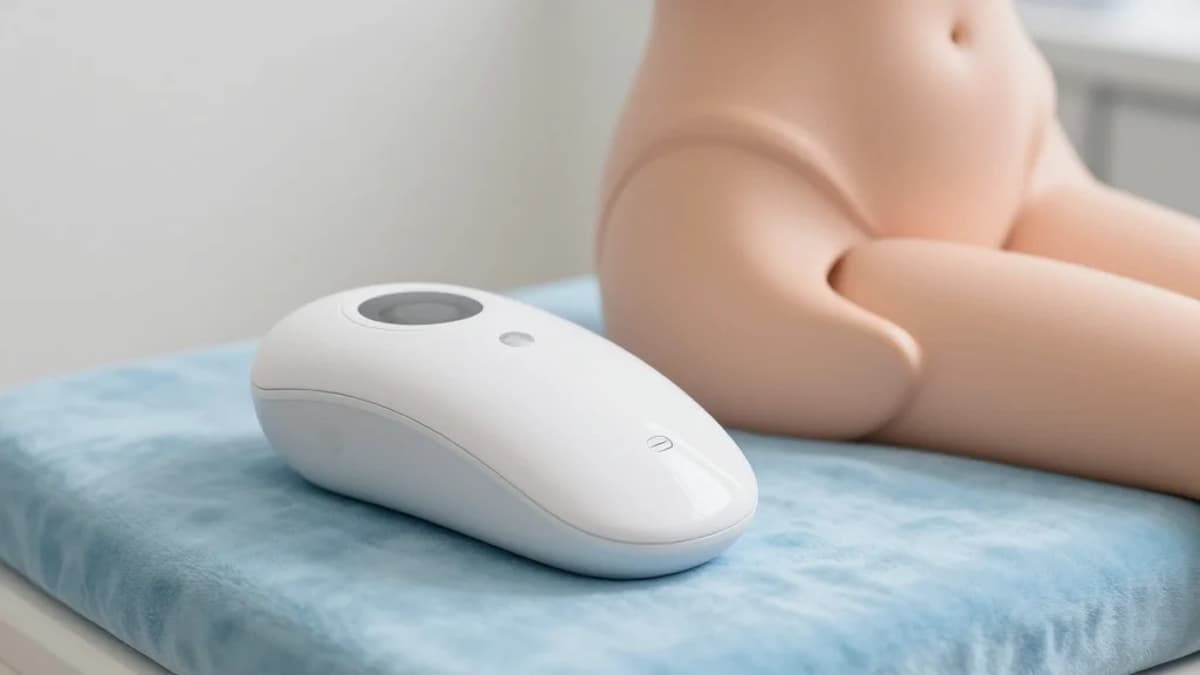
New Real-World Evidence Study Shows Leva® Pelvic Health System Significantly Improves Fecal Incontinence Symptoms in Women
•March 9, 2026
0
Why It Matters
The results validate a non‑surgical, technology‑driven option for a condition that affects millions of women, potentially reshaping treatment standards and payer coverage decisions.
Key Takeaways
- •Leva reduced FI episodes by 45% on average
- •Pelvic floor strength scores improved significantly
- •Patient‑reported quality‑of‑life increased 30%
- •Study confirms safety across diverse clinical settings
- •Digital therapeutic offers drug‑free alternative
Pulse Analysis
The new real‑world evidence study adds a critical layer of credibility to Axena Health’s Leva Pelvic Health System, a digital therapeutic that combines biofeedback, sensor‑guided exercises, and remote clinician support. By publishing in a peer‑reviewed journal, Axena demonstrates that its technology can deliver measurable outcomes outside the controlled environment of randomized trials, a factor increasingly demanded by insurers and health systems seeking cost‑effective solutions for chronic pelvic floor disorders.
Fecal incontinence remains an under‑treated condition, with prevalence estimates ranging from 5 to 15 percent among women over 50. Traditional management often relies on medication, invasive procedures, or long‑term pelvic floor therapy that suffers from low adherence. Leva’s platform addresses these gaps by providing personalized, data‑driven exercise regimens that patients can perform at home, while clinicians monitor progress through a secure dashboard. The study’s reported 45 percent reduction in incontinence episodes and a 30 percent boost in quality‑of‑life scores suggest that digital therapeutics can achieve outcomes comparable to, or better than, conventional interventions.
From a market perspective, the success of Leva signals a broader shift toward evidence‑based digital health solutions in women’s pelvic health. Payers are beginning to reimburse for remote therapeutic programs that demonstrate real‑world efficacy, and manufacturers are investing heavily in sensor technology and AI‑enhanced coaching. As more data emerge, competitors will need to match Axena’s clinical rigor and patient engagement models to capture market share, while regulators may tighten standards for digital therapeutic validation. The study thus not only validates Leva’s clinical promise but also accelerates the adoption trajectory for non‑invasive pelvic health technologies.
New Real-World Evidence Study Shows Leva® Pelvic Health System Significantly Improves Fecal Incontinence Symptoms in Women
0
Comments
Want to join the conversation?
Loading comments...