Targeted PET/CT Tracer Helps Rheumatoid Arthritis Patients
•February 26, 2026
0
Why It Matters
Early detection of treatment response shortens ineffective therapy periods, reducing joint damage risk and healthcare costs while enabling precision medicine in rheumatoid arthritis.
Key Takeaways
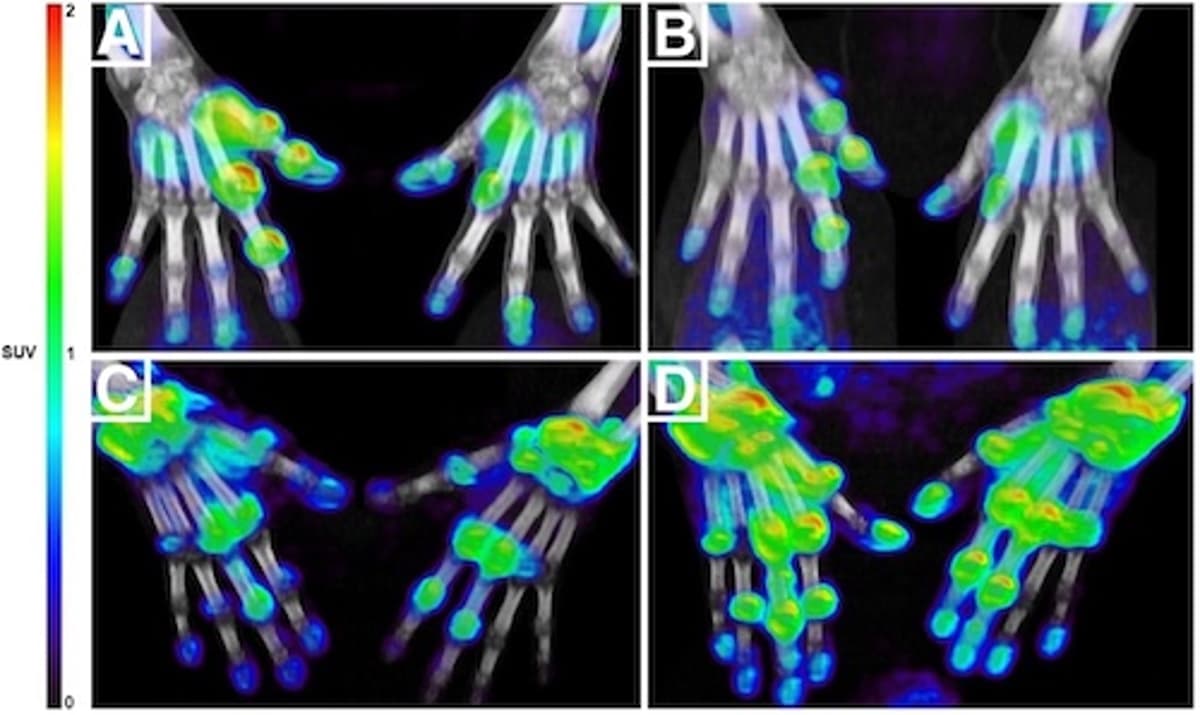
- •11C‑DPA‑713 PET/CT quantifies macrophage activity.
- •Detects anti‑TNF response as early as four weeks.
- •Baseline SUV predicts 26‑week clinical outcomes.
- •Combines imaging with clinical data for better prediction.
- •Enables personalized rheumatoid arthritis treatment strategies.
Pulse Analysis
Rheumatoid arthritis management has long been hampered by a lag between treatment initiation and measurable clinical response. Conventional assessment relies on symptom scores and imaging that may not reveal therapeutic efficacy until six months have passed, during which irreversible joint damage can accrue. The industry therefore seeks biomarkers that reflect underlying disease biology in real time, allowing clinicians to pivot quickly when a regimen fails. Molecular imaging, particularly PET/CT, offers a window into cellular activity that conventional radiography cannot provide.
The 11C‑DPA‑713 tracer targets macrophages, the immune cells that drive synovial inflammation. By quantifying tracer uptake (SUV) across affected joints, researchers captured a snapshot of inflammatory burden at baseline and after four weeks of anti‑TNF therapy. Statistical models demonstrated that higher early SUVs reliably forecasted disease activity at 26 weeks, and that integrating select clinical variables sharpened predictive power. This early signal transforms the decision‑making timeline, turning a months‑long guesswork process into a data‑driven, week‑level assessment.
If adopted broadly, this imaging approach could reshape rheumatology practice. Payers would benefit from reduced expenditures on ineffective biologics, while patients experience fewer flares and preserved joint function. Moreover, the methodology sets a precedent for using immune‑cell‑specific tracers across autoimmune conditions, accelerating the shift toward personalized medicine. Ongoing trials will need to validate cost‑effectiveness and scalability, but the current evidence positions macrophage‑targeted PET/CT as a pivotal tool in the next generation of rheumatoid arthritis care.
Targeted PET/CT Tracer Helps Rheumatoid Arthritis Patients
0
Comments
Want to join the conversation?
Loading comments...