
Makary’s Grip Slips at FDA, Triggering Speculation on Successor
White House leaks indicate President Donald Trump is increasingly dissatisfied with FDA Commissioner Marty Makary, shifting speculation from if to when he might be removed. The immediate catalyst appears to be Makary’s slow approval of flavored vaping products, a decision the administration deems politically critical ahead of the 2024 midterm elections. Potential successors – including Graham, Zeta and Diamantas – have emerged as frontrunners for an acting commissioner role. The situation underscores mounting political pressure on the agency’s leadership.

Odyssey Completes Journey to NASDAQ: Public Equity Report
Odyssey Therapeutics completed its long‑awaited IPO and debuted on Nasdaq, after withdrawing a previous filing in June 2025. The immunology‑focused company priced its shares in early May 2026, marking a successful market entry. At the same time, three biotech firms—Cytokinetics,...

Tr1X: Inducing Long-Term Immune Tolerance with Allogeneic Tr1 Cells
BioCentury has updated its website privacy framework, detailing the use of strict, functional, marketing, advertising, and analytics cookies. The policy explains that essential cookies enable authentication, registration, and navigation, while functional cookies enhance personalization. Marketing and advertising cookies are used...
Two Mechanisms Vie to Deliver First Hypoxic Ischemic Encephalopathy Drug
The article is BioCentury’s cookie policy, outlining five cookie categories—strictly necessary, functional, marketing, advertising, and analytics—and describing their purposes for site operation, personalization, outreach, and data collection. It explains how each type works, the data it handles, and the impact...

Science Spotlight: New Prime Editing Tools for Large DNA Insertions
BioCentury’s website employs a tiered cookie framework that classifies cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Strictly necessary cookies power core services such as authentication, registration, and user‑preference management, while functional cookies enhance site personalization. Marketing and...

Cytokinetics Scores Late-Stage Win in Non-Obstructive HCM as BMS Plans Camzyos Restart
Cytokinetics announced that its experimental drug Myqorzo (aficamten) achieved both primary endpoints in the Phase III ACACIA‑HCM trial, marking the first late‑stage success for a therapy targeting non‑obstructive hypertrophic cardiomyopathy (HCM). The result propelled the company’s stock up 17%, pushing its...

FDA, up Close and Personal: A Banker’s Recalibration
Former FDA senior advisor Peter Reikes argues that the agency’s perceived slowness stems from its core principles of independence, caution, and process discipline rather than dysfunction. He notes that despite recent leadership turnover and political pressure, a deep bench of...

Tiny Biotech’s Experience Raises Questions About FDA’s Rare Disease Policies
BioCentury’s website now publishes a detailed cookie policy that separates cookies into five categories: strictly necessary, functional, marketing, advertising, and analytics. Strictly necessary cookies are always active and essential for authentication and navigation, while functional cookies enable personalization of the...

In Chiesi’s Biggest M&A Deal yet, Pharma Buys HAE Company KalVista
Italian pharmaceutical group Chiesi announced the acquisition of U.S. biotech KalVista for approximately $1.5 billion in cash, marking its largest M&A transaction to date. KalVista, a specialist in hereditary angioedema (HAE), brings a late‑stage monoclonal‑antibody program and a pipeline of complement‑targeted...

Inventiva Reloads C-Suite as It Looks to Readout
Inventiva S.A. announced three senior appointments as it prepares for a Phase III readout of lanifibranor in metabolic dysfunction‑associated steatohepatitis (MASH) in Q4 2026. Axel‑Sven Malkomes, former CureVac CFO, will become CFO, with incumbent Jean Volatier moving to EVP finance & corporate...
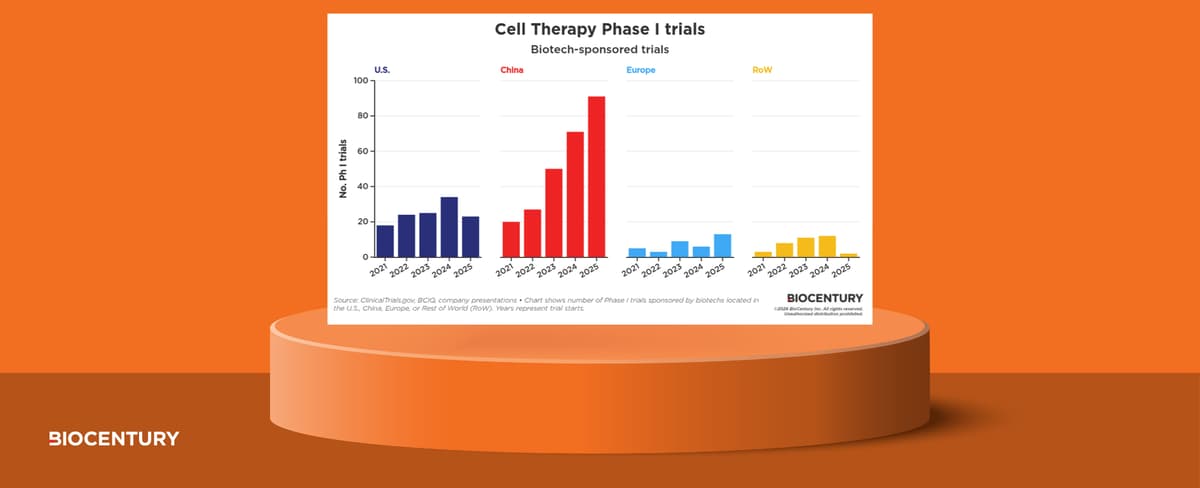
Cell Therapy Phase I Activity Accelerates on China Surge
BioCentury’s website now features a detailed Privacy Preference Center that categorizes cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Users can toggle consent for each category, though disabling essential cookies may impair login and navigation. The policy clarifies...

Pan-RAS Inhibitor ADCs Emerge at AACR 2026
At the 2026 American Association for Cancer Research meeting, four biotech companies unveiled antibody‑drug conjugates that deliver pan‑RAS inhibitors directly to tumor cells. Pan‑RAS inhibition has recently delivered unprecedented survival improvements in pancreatic and other RAS‑mutant cancers, but its clinical...

Multi-Regional Trials Have yet to Become the Norm
BioCentury’s website outlines a comprehensive cookie policy that categorises cookies into strictly necessary, functional, marketing, advertising, and analytics types. Strictly necessary cookies are always active and essential for authentication and site navigation, while functional cookies enable enhanced personalization. Marketing and...

Trump’s Psychedelic Order Tests Review Norms
BioCentury’s website uses a tiered cookie system to support essential functions, personalization, marketing, advertising, and analytics. Strictly necessary cookies are always active, enabling authentication, registration, and navigation, while functional cookies enhance site features. Marketing and advertising cookies help tailor product...

Nektar’s Phase II Alopecia Extension Reveals Late Responders to IL-2 Therapy
Nektar Therapeutics reported results from a Phase II extension study of its interleukin‑2 (IL‑2) therapy for alopecia areata. The data revealed a subset of patients who did not respond initially but achieved meaningful hair regrowth after prolonged exposure, raising the...

A 32-Year Look at Biotech’s Capital Expansion
Over the past 32 years biotechs have amassed nearly $1.2 trillion in equity financing, according to BioCentury’s BCIQ database. Remarkably, half of that capital was raised in the eight‑year window from 2018 to 2025, underscoring an accelerating funding wave. The surge,...

Remembering Sofinnova’s Denis Lucquin, Father of French Biotech
Denis Lucquin, a founding partner of Sofinnova Partners and a pivotal architect of France’s biotech sector, died on April 3 at age 69 from pancreatic cancer. Over three decades he invested in 31 European biotech companies, including Ablynx, Nicox and Novexel, and...

Psoriasis Paves the Way for Next-Generation TYK2 Inhibitors in Autoimmunity
Takeda and Alumis reported Phase III data for their next‑generation TYK2 inhibitors, zasocitinib and envudeucitinib, showing more than half of patients achieve PASI 90 in plaque psoriasis. The results close the efficacy gap between oral agents and injectable biologics, surpassing the 28%...

RevMed’s Pancreatic Cancer Win Strengthens the Case for Targeting RAS(ON)
RevMed reported a positive Phase 2 trial of its RAS(ON) inhibitor in patients with advanced pancreatic ductal adenocarcinoma, showing a 23% objective response rate and a median progression‑free survival of 5.8 months. The study enrolled 45 heavily pre‑treated patients and...

Biotech Executive, Combative Conservative Hemmati May Head CBER
The FDA is nearing a leadership transition at its Center for Biologics Evaluation and Research (CBER) as Vinay Prasad prepares to depart at month‑end. Houman David Hemmati, a biotech executive known for his combative conservative views, tops Commissioner Marty Makary’s shortlist. Several other candidates...
Fibrosis Company Avalyn Seeks NASDAQ Listing Amid Volatility: Finance Report
Avalyn Pharma, a Boston biotech focused on inhaled treatments for pulmonary fibrosis, filed a prospectus to list on NASDAQ despite ongoing market volatility. The company’s pipeline includes two Phase II programs: AP01, an inhaled pirfenidone in the Phase IIb MIST trial, and...

Blackstone’s Galakatos on Scaling Deals with $6.3B Fund: ‘Broader, Further’
Blackstone’s Galakatos team announced a new $6.3 billion private‑equity fund aimed at scaling the number and size of its transactions. The fund will pursue a broader geographic footprint and deeper sector coverage, focusing on later‑stage growth and buy‑and‑build opportunities. Galakatos plans...

Tackling Translation: The Parker Institute Model
The Parker Institute for Cancer Immunotherapy marks its tenth anniversary by spotlighting a deliberate approach to translating scientific discoveries into medicines. Founder Karen Knudsen notes that while U.S. discovery remains strong, the nation struggles with moving breakthroughs through development pipelines....
2Q Markets Preview, Tariffs and Biotech Takeouts — a BioCentury Podcast
Biotech remains resilient as markets wobble, with M&A activity persisting. The BioCentury podcast highlighted two major pending deals: Eli Lilly’s $6.3 billion acquisition of Centessa Pharmaceuticals and Biogen’s proposed $5.6 billion purchase of Apellis. Analysts also examined the lingering effects of Trump-era tariffs...

Alzheimer’s Company Korsana Heads to NASDAQ via Merger: Deals Report
Korsana Biosciences, a neuroscience startup backed by the Fairmount Funds‑backed Paragon incubator, announced a reverse‑merger with Cyclerion Therapeutics to go public on Nasdaq under the ticker KRSA. The all‑stock deal gives Korsana shareholders roughly 98.5% of the combined equity, leaving...
Liver Specialist Amit Singal Joining Curve Biosciences
Amit Singal, a leading hepatology expert, has joined Curve Biosciences as chief medical officer. Singal currently serves as chief of hepatology at UT Southwestern Medical Center and authored the latest AASLD practice guidance on liver cirrhosis monitoring. Curve, based in San Mateo, leverages its...

Pharma Tariffs Pressuring Smaller Companies to Do MFN Deals
BioCentury’s website now outlines a comprehensive cookie framework that classifies cookies into five distinct groups: strictly necessary, functional, marketing, advertising, and analytics. Each category serves a specific purpose, from enabling authentication and core site functions to gathering anonymous usage metrics...

Resiliency in the Face of Volatility
Biotech markets are poised to sustain their outperformance as they navigate heightened geopolitical uncertainty. BioCentury’s Q2 2026 preview highlights strong follow‑on equity offerings and a robust M&A pipeline as primary catalysts. The sector’s resilience is underscored by more than $1 billion in...

Science Spotlight: Three Teams Converge on RNU2‑2 as Targetable for Neurodevelopmental Epilepsies
Three independent research teams reported in Nature Genetics that variants in the non‑coding RNA RNU2‑2 cause both a dominant neurodevelopmental epilepsy syndrome and a prevalent recessive childhood disorder with epilepsy. By analyzing overlapping international cohorts and shared genomic datasets, they...

Shreehas Tambe Becomes First CEO of Newly Combined Biocon
Shreehas Tambe has been appointed the first chief executive officer of the newly combined Biocon entity, formed after Biocon’s merger with contract‑research firm Syngene. The merged company is valued at roughly $5.5 billion and aims to become India’s largest biotech services...

Excelsior Sciences: Automating Small Molecule Chemistry
Excelsior Sciences, backed by Deerfield, unveiled an automated platform for small‑molecule discovery that leverages modular "smart blocs" and generative AI. The system integrates iterative carbon‑carbon bond formation, robotic synthesis, and in‑vitro assays into a continuous make‑test‑learn loop. By translating chemical...

HHS Unveils MANA Plan
On April 1, 2026, the U.S. Department of Health and Human Services (HHS) unveiled a new initiative dubbed the MANA plan, short for “Make America Normal Again.” The announcement was delivered by HHS Secretary Robert F. Kennedy Jr. during a briefing at the...
Zag Bio: Rewiring the Thymus for Antigen-Specific Tolerance
Zag Bio, a Cambridge‑based biotech, announced an $80 million Series A round to advance its lead program that rewires the thymus for antigen‑specific tolerance. The company’s approach employs bispecific antibodies to ferry self‑antigens into thymic cells, aiming to re‑educate T‑cells and induce...

RFK Jr.’s Peptide Deregulation Threatens the Foundations of Drug Safety
HHS Secretary Robert F. Kennedy Jr announced on a podcast that the federal government will lift restrictions on 14 injectable peptides, allowing compounding pharmacies to produce and sell them without the usual FDA review. The move bypasses the scientific advisory...

Going ‘Golden’: K-Biotech May Be Prepping for Its Moment on the Global Stage
Korean biotech is gaining global visibility after the East‑West Biopharma Summit highlighted its first‑in‑class pipelines. The Korean Ministry of Health and Welfare announced that Eli Lilly will invest $500 million over the next five years to collaborate with domestic firms. The deal...

MHRA’s Tallon on the Tall Order of Boosting the U.K. as a Destination for Clinical Trials
Lawrence Tallon, the MHRA chief, is set to oversee the launch of the Medicines for Human Use (Clinical Trials) (Amendment) Regulations 2025 on April 28, 2026. The amendment represents the most extensive overhaul of U.K. clinical‑trial rules in two decades, targeting faster approval,...

AL-S Pharma Tests How Far SOD1 Biology Extends Into Sporadic ALS
The article outlines BioCentury’s cookie policy, detailing the categories of cookies used on its website—strictly necessary, functional, marketing, advertising, and analytics. Each type is described in terms of purpose, activation status, and impact on user experience. The policy emphasizes that...

Recursion’s Khan: AI Will Be Judged by the Medicines It Makes
Recursion Pharmaceuticals CEO Najat Khan says AI’s worth will be judged by the medicines it helps create, not by the sophistication of the tools. She emphasizes deploying AI selectively in chemistry, trial execution, and out‑of‑domain predictions to generate clinical proof...

R1: China-to-U.S. NewCo Planning Global Testing of New CKD Mechanism
The article outlines BioCentury’s cookie policy, detailing categories of cookies used on its website. Strictly necessary cookies are always active and enable authentication, registration, and core navigation. Functional, marketing, advertising, and analytics cookies provide personalization, targeted promotions, ad tracking, and...
Rare Disease Spotlight: Friedreich Ataxia Moves Beyond Mitochondrial Bandages
Friedreich ataxia (FA) received its first FDA‑approved therapy in 2023 when omaveloxolone, marketed as Skyclarys, earned accelerated approval. Biogen’s $7.3 billion acquisition of Reata Pharmaceuticals secured the drug and highlighted the market’s appetite for rare‑disease assets. Skyclarys works by activating the...
Kenai: Off-the-Shelf Parkinson’s Progenitors with Precision Transplant Tech
Kenai, a new allogeneic cell‑therapy company, is developing off‑the‑shelf iPSC‑derived progenitor cells for Parkinson’s disease, paired with a precision brain‑delivery platform that uses real‑time functional imaging. The approach builds on Japan’s recent approvals of iPSC‑based Parkinson’s treatments and aims to...
Solid Eyes Accelerated Approval as DMD Gene Therapy Faces FDA Uncertainty
Solid Biosciences reported Phase I/II data for its DMD gene therapy SGT‑003, showing higher microdystrophin expression and a greater proportion of positive fibers than Sarepta’s Elevidys. The results strengthen SGT‑003’s case for accelerated FDA approval, but the pathway is clouded by...
Can FDA Tolerate Cancer Risk for Rare Pediatric Disease Gene Therapies?
The FDA placed a clinical hold on Regenxbio’s RGX‑111 and RGX‑121 gene‑therapy trials after a pediatric MPS I patient developed a tumor four years post‑treatment. The case marks the first documented long‑latency cancer linked to an adeno‑associated virus (AAV) vector in...
Science Spotlight: New Ways to Attack Β-Amyloid Plaques in Alzheimer’s
Two pre‑clinical studies propose active clearance of β‑amyloid as a new Alzheimer’s strategy. Researchers at Washington University engineered astrocytes with chimeric antigen receptors (CARs) that engulf plaques, while another team designed bispecific peptides that ferry amyloid into cells for lysosomal...
Roche’s Giredestrant Miss Refines Treatment Settings for Oral SERDs
Roche’s oral selective estrogen receptor degrader (SERD) giredestrant failed to meet its primary endpoint in a late‑stage Phase III trial, but the data revealed meaningful activity in specific patient subgroups, particularly those with ESR1 mutations. The miss prompted Roche to...

RTW-Backed Prolium Emerges with $50M a Year After China Licensing Deal: Finance Report
Prolium Bioscience, a NewCo‑style biotech backed by RTW Investments, announced its public debut after securing a $50 million financing package. The capital influx follows a licensing agreement with China’s Innocare Pharma and Keymed Biosciences, granting the partners rights to develop Prolium’s...
EpiciphAI: Reading Disease States From Blood’s Epigenetic Fingerprints
EpiciphAI, a Chinese pre‑seed biotech, is building a liquid biopsy that reads histone modifications on cell‑free chromatin to pinpoint a fragment’s tissue of origin and its disease state. Unlike most epigenetic tests that focus on DNA methylation, this platform targets...
Promising Henlus Data Tripped up by Psychedelic Trial Design Dilemma
Helus Pharma reported Phase II data for its psychedelic candidate HLP004, indicating symptom improvement in generalized anxiety disorder patients for up to six months. However, the trial’s low‑dose active control arm showed similar efficacy, making it difficult to isolate the...

Promising Helus Data Tripped up by Psychedelic Trial Design Dilemma
Helus Pharma reported Phase II data for its psychedelic candidate HLP004 in generalized anxiety disorder, showing symptom improvement lasting six months. However, the trial’s active low‑dose control produced outcomes similar to the therapeutic dose, making it difficult to isolate the drug’s...

Asia Deals Aim for First-in-Class Biology, with at Least Four Deals for New Targets
Asian biotech firms outside China announced 72 deals in 2024‑25, emphasizing first‑in‑class assets over me‑better or me‑too programs. At least four transactions involve novel targets such as CNTN4, GPR52, MTARC1 and OK‑1, spanning new modalities and indications. Global pharmaceutical companies...