
Ovarian Cancer Clinical Trials: How Patient-Centered Design Can Improve Late-Line Study Enrollment
Genelux is advancing Olvi-Vec, an oncolytic immunotherapy, in late‑line ovarian cancer with a Phase III OnPrime trial that compares Olvi-Vec plus chemotherapy to physician’s choice. The Phase II VIRO‑15 study reported a 54% objective response rate, 11‑month median progression‑free survival and 15.7‑month overall survival in patients who had a median of four prior therapies. Zindrick emphasizes patient‑centered trial design—simplifying visits, using telehealth, and removing caps on prior treatments—to reduce enrollment friction. The FDA’s evolving guidance on using a single robust trial plus confirmatory evidence could accelerate approvals for such high‑unmet‑need indications.

What It Takes to Run Rare Pediatric Disease Trials Across Borders: Insights From Polaryx Therapeutics
Polaryx Therapeutics is gearing up to test its oral candidate PLX-200 in a Phase II SOTERIA basket trial for four ultra‑rare lysosomal storage disorders—CLN2, CLN3, Krabbe and Sandhoff. The FDA has granted Fast Track designation for all four indications, promising closer...

What Psychedelic Clinical Trials Could Teach Psychiatry
Treatment‑resistant depression (TRD) affects roughly one‑third of U.S. adults on antidepressants and remains a high‑need, low‑treatment area. Compass Pathways’ synthetic psilocybin, COMP360, recently achieved its primary endpoints in two Phase III trials—258 patients in COMP005 and 581 in COMP006—showing rapid, durable...
World AIDS Vaccine Day 2026: What HIV Vaccine Research Is Testing Now
World AIDS Vaccine Day 2026, themed “Rethink. Rebuild. Rise,” highlighted a shifting HIV prevention landscape. In Europe, funding for vaccine R&D fell from about $16.5 million in 2009 to roughly $9.9 million in 2020, jeopardizing expertise. New data show individual broadly neutralizing...
Clinical Trial Failures of 2026
Clinical trial success rates remain low, hovering around 5% overall, with Phase II proving especially challenging. Industry‑sponsored Phase II and III terminations have roughly doubled from 2013 to 2023, often driven by commercial strategy shifts. In 2026, several high‑profile late‑stage...

Top 20 Fastest Growing Pharma & Biotech Companies in the Americas for 2026
The Financial Times‑Statista 2026 ranking spotlights the 20 fastest‑growing pharma and biotech firms in the Americas, measuring compound annual growth rates from 2021‑2024. Companies such as Capricor Therapeutics, BioAtla, Ardelyx, Legend Biotech and Repare Therapeutics posted explosive revenue gains, driven...

Marty Makary Steps Down as FDA Commissioner After Complaints, Adding to Regulatory Uncertainties
Marty Makary resigned as FDA commissioner after a turbulent 13‑month tenure marked by rapid regulatory experiments and political controversy. President Trump named deputy commissioner for food, Kyle Diamantas, a lawyer, as acting commissioner, placing a non‑physician at the agency’s helm. Makary’s...

World Clinical Trials Day 2026 Looks at the Science, Setbacks and Systems Behind New Medicines
World Clinical Trials Day 2026, observed on May 20, highlighted the “Research Rising” theme to honor the clinical‑research workforce and patients. Phase II studies now comprise 43% of the 584,000 active global trials, while oncology still drives 38% of Phase I‑III starts despite...

Top 7 Fastest Growing Pharma & Biotech Companies in Asia for 2026
The Financial Times’ 2026 ranking spotlights the seven fastest‑growing Asian pharma and biotech firms, each posting double‑digit revenue surges and landmark milestones. SillaJen earned an FDA IND for its oncolytic virus BAL0891, while Oscotec leveraged lazertinib royalties to boost 2024...

The Case for Smarter Neuroinflammation Clinical Trials
INmune Bio, led by CEO David Moss, is developing non‑immunosuppressive therapies that target innate immune pathways, including the XPro1595 program for neurodegenerative disease and the CORDStrom mesenchymal stromal cell platform for recessive dystrophic epidermolysis bullosa (RDEB). The company argues that...

New Blood Test for Early Alzheimer’s Detection with FNIH’s Dr. Alessio Travaglia — Episode 255
A new blood test developed by the Foundation for the National Institutes of Health (FNIH) can predict Alzheimer’s symptom onset three to four years in advance, according to a study published in Nature Medicine. The test leverages a clock‑model biomarker...

Sanofi Bets $294M on Toronto AI Hub
Sanofi is committing roughly US$218 million (CAD 294 million) to expand its Toronto Artificial Intelligence Centre of Excellence, adding 50 machine‑learning positions and bringing its digital workforce in Ontario to more than 150 by 2028. The province is contributing a conditional grant of...

Why Attend the 2026 Canadian Pharma & Biotech PPM Summit Toronto
The Canadian Pharma & Biotech Project, Program and Portfolio Management Summit will convene senior life‑science leaders in Toronto on May 27‑28, 2026. Attendees will explore how to strengthen portfolio performance amid pricing pressure, evolving regulations, and intensified competition for global...

Small Cell Lung Cancer Research Moves Toward a More Precision-Driven Era
Small cell lung cancer (SCLC) is transitioning toward precision oncology as researchers uncover distinct molecular subtypes and high‑frequency targets such as DLL3. Amgen’s DLL3‑directed T‑cell engager tarlatamab and emerging antibody‑drug conjugates illustrate a shift from conventional chemotherapy to targeted immunotherapies....
Not Just Arthritis: A Joint Effort to Change Arthritis Care in Canada
Arthritis Society Canada released its 2026 State of Arthritis Report Card, revealing that one in five Canadians—more than those with diabetes, heart disease, cancer, stroke and dementia combined—live with arthritis, and half are under 65, including 25,000 children. Despite a...
Targeting Ischemic Disease with DiaMedica CEO Rick Pauls — Episode 254
DiaMedica Therapeutics, led by CEO Rick Pauls, is advancing a recombinant KLK1 protein to treat ischemia‑driven diseases such as preeclampsia, fetal growth restriction, and acute ischemic stroke. The podcast episode highlights the company’s focus on restoring vascular blood flow and...

World Ovarian Cancer Day 2026: New Diagnostics and ADCs
World Ovarian Cancer Day 2026 highlighted the #NoWomanLeftBehind campaign as ovarian cancer, though only 1% of U.S. cases, claims an estimated 12,450 deaths. Recent FDA approvals—including Corcept’s Lifyorli for platinum‑resistant disease and Agilent’s PD‑L1 IHC 22C3 companion diagnostic—expand treatment options....

AstraZeneca’s Camizestrant Hit by FDA Advisory Committee Vote While Truqap Moves Ahead
FDA’s Oncologic Drugs Advisory Committee issued a split verdict on AstraZeneca’s oncology pipeline this week. The committee voted 3‑to‑6 against the benefit‑risk profile of camizestrant combined with a CDK4/6 inhibitor for ESR1‑mutated advanced breast cancer, despite a 56% progression‑free survival...
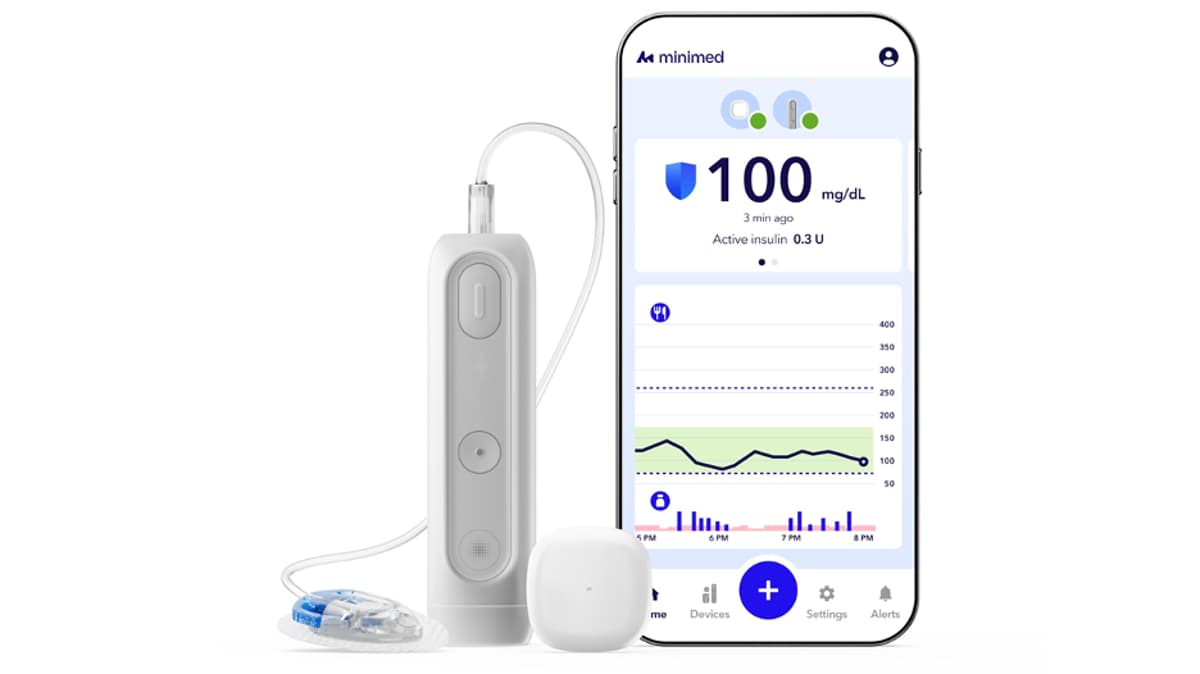
MiniMed IPO Points to Smarter Diabetes Devices
Medtronic spun off its diabetes technology unit MiniMed in a March IPO, offering 28 million shares at $20 each and raising roughly $538 million. Post‑offering Medtronic retains about 90 % of MiniMed’s common stock, preserving strategic control. Shortly after the listing, MiniMed secured...

Most Sustainable Pharma and Biotech Companies in 2026, According to Corporate Knights
Corporate Knights’ 2026 Global 100 ranking spotlights sustainability leaders in pharma and biotech, with Novonesis A/S, Eisai Co Ltd and Novo Nordisk A/S earning top spots. The list introduces a new "sustainable revenue momentum" metric, now weighing one‑third of each company’s score....

PharmaLogic Expands Radiopharmaceutical Footprint With New Atlanta Facility
PharmaLogic announced the opening of a new PET radiopharmaceutical manufacturing facility in Atlanta, Georgia, as part of its $250 million US expansion. The state‑of‑the‑art plant will increase production capacity, accelerate delivery to Southeast hospitals, and serve as an innovation hub for...

Where Did Your Web Traffic Go? How to Reclaim Leads From AI Answer Engines
In 2026 AI answer engines are delivering zero‑click answers, siphoning traffic from life‑science marketers’ websites. The AI scans public pages in real time, providing scientists with quick answers and leaving the source unvisited. Marketers are urged to adopt a dual‑layer...

Pharmaville Positions Life Science Locations on the Global Investment Map
Pharmaville, an Xtalks‑backed life‑science location intelligence initiative, launches its Smart Locator platform to centralize fragmented regional data for manufacturers, R&D teams, CROs and professionals. The rollout comes as U.S. pharma giants announce major investments—AbbVie’s $1.4 billion Durham campus and Eli Lilly’s $3.5 billion...

New Report Finds 75% of Women Skip Healthcare Due to Scheduling Constraints
Teal Health’s State of Her Health 2026 report reveals that 75% of U.S. women have likely skipped a medical appointment because it didn’t fit into their schedules, and 55% say they definitely have. The survey of more than 500 women...

FDA Clears Custom Titanium Cranial Implant for Skull Repair
The U.S. FDA granted 510(k) clearance to CGBIO’s EASYMADE TI, a patient‑specific titanium cranial and craniofacial implant made via laser powder‑bed fusion. The device is designed from each patient’s CT scan, manufactured in South Korea and shipped to U.S. hospitals within...

World Asthma Day 2026: New Anti-Inflammatory Drugs and More for Asthma Control
World Asthma Day on May 5 highlighted the persistent gap in access to anti‑inflammatory inhalers, a core theme of the Global Initiative for Asthma. The FDA approved AstraZeneca’s Breztri Aerosphere, the first single‑inhaler triple therapy that combines an inhaled corticosteroid, a...
Should Regulators Rely More on AI to Accelerate Drug Approvals?
Regulators, led by the FDA, are testing AI tools to speed drug approval processes. The agency’s pilot projects include AI‑driven real‑time monitoring of clinical trials and automated data review. Proponents argue AI can cut analysis time and uncover hidden patterns,...

FDA Moves to Speed Review Support for Psychedelic-Related Treatments
The FDA announced a new fast‑track mechanism for psychedelic‑based therapies, issuing Commissioner’s National Priority Vouchers to three programs targeting depression and PTSD with psilocybin or methylone. It also granted an IND for DemeRx NB to study noribogaine hydrochloride as a...
Xtalks Featured Member: Ravindra Kumar, Medical Technologist, PGIMER, Chandigarh
Ravindra Kumar, a medical technologist at PGIMER in Chandigarh, has been highlighted by Xtalks for his work in MRI optimization and radiation dose reduction. He recently completed a phantom‑based chest CT study that improved image quality while lowering patient exposure,...

Xtalks Featured Member: Parminder Singh Bhasin, Senior Director, Head of Analytical Research and Project Management, Intech Biopharm. Ltd
Parminder Singh Bhasin, Senior Director at Intech Biopharm Ltd., has been highlighted as an Xtalks Featured Community Member. With more than 26 years in pharmaceutical R&D, he oversees analytical research, method validation, and project management for complex generics, pMDI, nasal...

Xtalks Featured Member: Laura Saltonstall, VP, Medical Affairs, Immunology, Sobi
Xtalks spotlighted Dr. Laura Saltonstall, Vice President of Medical Affairs for Immunology at biotech firm Sobi. Drawing on her clinical background and personal experience as a parent of a child with a rare genetic disorder, she drives patient‑centered strategies that...

Why Copyright Matters in Life Sciences in the Age of AI: Featuring Neal Dunkinson of CCC (Copyright Clearance Center) —...
In episode 253 of the Xtalks Life Science Podcast, Neal Dunkinson, senior director of solution sales at the Copyright Clearance Center (CCC), explains why copyright and licensing have become mission‑critical for life‑science organizations, especially as AI tools infiltrate research workflows....

Alamar Biosciences IPO Raises $219.9M for Blood-Based Biomarker Tools
Alamar Biosciences closed an upsized IPO, raising $219.9 million by pricing 12.9 million shares at $17 each, and began trading on Nasdaq under the ticker ALMR. The California‑based firm offers the NULISA platform, which detects low‑level protein biomarkers in blood, and its...

Kailera Therapeutics IPO Raises $718.8M for Obesity Drug Pipeline
Kailera Therapeutics completed a $718.8 million IPO, pricing 44.9 million shares at $16 each and listing on Nasdaq under KLRA. The company’s lead candidate, ribupatide, is a once‑weekly injectable GLP‑1/GIP dual agonist currently in three global Phase III trials targeting obesity and type 2...

500,000 UK Biobank Records Leaked and Listed for Sale on Alibaba
On April 23, 2026, listings for the health records of 500,000 UK Biobank participants appeared on the Chinese e‑commerce platform Alibaba. The data, while stripped of direct identifiers, includes age, sex, socioeconomic status and detailed health measurements, raising re‑identification concerns....

Can We Truly Deliver Gene Therapies to Patients Without Solving the Cost and Scale Challenges of AAV Production?
Gene‑therapy developers are confronting a manufacturing bottleneck as adeno‑associated virus (AAV) vectors must be produced at commercial scale. FUJIFILM Biotechnologies proposes 2,000‑liter single‑use bioreactors combined with an integrated CDMO model to boost yield, cut cost per dose, and preserve product...

World Maternal Mental Health Day 2026: Advances in Clinical Research
World Maternal Mental Health Day on May 6, 2026 spotlights the growing urgency of perinatal mood and anxiety disorders. Studies show 70% of affected women hide symptoms, while the U.S. National Maternal Mental Health Hotline has fielded over 89,426 calls and...
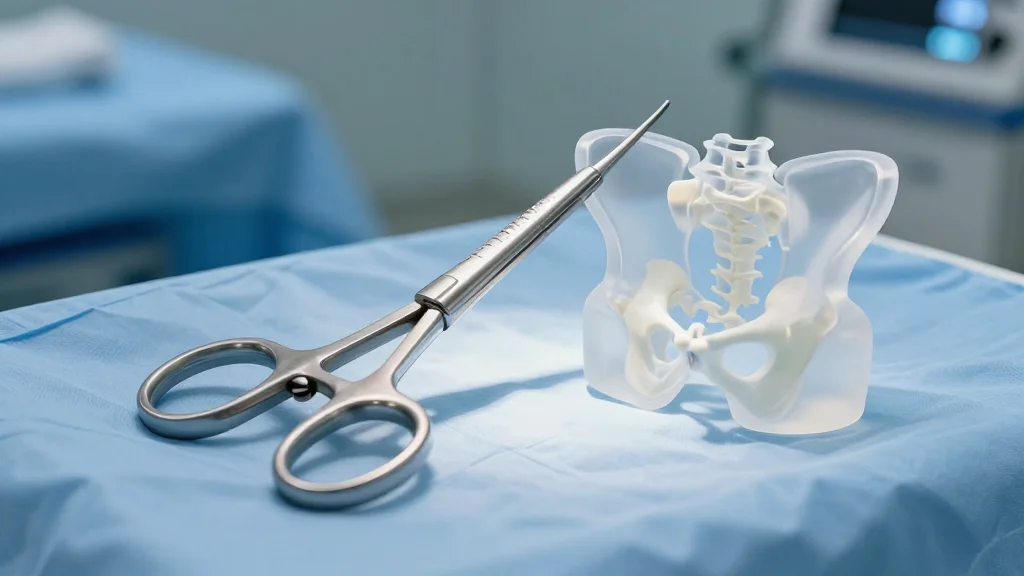
SurGenTec Adds Navigation Option to FDA-Cleared SI Joint Fusion System
SurGenTec received FDA 510(k) clearance for its TiLink navigation instruments, which integrate with Medtronic’s StealthStation platform to provide real‑time guidance during minimally invasive sacroiliac (SI) joint fusion. The new tools are designed to help surgeons locate, access, and prepare the...

Only 9% of Americans Know How to Maintain Brain Health, Alzheimer’s Association Finds
The Alzheimer’s Association’s 2026 Brain Health in America report reveals a stark knowledge gap: while 88% of U.S. adults aged 40+ consider brain health very important, only 9% say they know “a lot” about how to protect it. Respondents recognize...
Neuromodulation and Nerve Ablation for Chronic Pain with NeuroOne CEO Dave Rosa — Episode 252
NeuroOne Medical Technologies, led by veteran CEO Dave Rosa, is advancing minimally invasive electrodes that combine nerve ablation, neuromodulation, and direct drug delivery for conditions like epilepsy and chronic pain. Rosa, who has spent three decades at firms such as...
Acorys System Gains FDA Clearance for Real-Time 4D Cardiac Mapping
Corify Care’s Acorys system received FDA clearance, offering clinicians a real‑time, four‑chamber view of cardiac electrical activity without the need for CT or MRI scans. The platform merges 3D anatomical modeling with live electrical signals, creating what the company calls...

1 in 5 US Blood Donors Show Sign of Prediabetes or Diabetes, Study Finds
A new analysis by the American Red Cross of more than 920,000 U.S. blood donors found that one in five exhibits hemoglobin A1C levels indicating prediabetes or diabetes. Roughly 80% of those elevated readings fall in the prediabetes range, while...

Top 10 Fastest-Growing Healthcare Companies in 2026, From Financial Times’ List
The Financial Times’ 2026 Americas Fastest‑Growing Companies list spotlights ten U.S. healthcare firms that are scaling rapidly amid a broader surge in health‑service deals and AI‑driven consumer behavior. PwC expects deal volume to climb as buyers chase technology‑enabled businesses, while...

Erica Schwartz CDC Nomination: What You Need to Know
President Donald Trump nominated Dr. Erica Schwartz, MD, JD, MPH, as the next CDC Director, pending Senate confirmation. Schwartz, a former Deputy Surgeon General and rear admiral in the Coast Guard, brings two decades of public‑health service and a blend...
New HPV Test Kit for At-Home Use Secures FDA Clearance
The FDA has cleared Waters Corporation’s Onclarity HPV Self-Collection Kit, allowing individuals to collect a vaginal sample at home and send it to a lab for analysis with the BD Onclarity HPV Assay. The kit detects high‑risk HPV strains and...

Anti-Amyloid Alzheimer’s Drugs Show Limited Benefit in Cochrane Review
A Cochrane review of 17 randomized trials involving 20,342 participants found that anti‑amyloid antibodies deliver little to no clinically meaningful improvement in memory, cognition, or dementia severity after roughly 18 months of treatment. The analysis also identified a markedly higher...
Top 10 Most Innovative Medical Device Companies in 2026, Per Fast Company
Fast Company’s 2026 list spotlights ten medical‑device innovators reshaping a market that reached $584 billion in 2025. The companies span connected wearables, AI‑driven diagnostics, focused‑ultrasound therapy and minimally invasive tools, reflecting a broader industry shift toward at‑home and less invasive care....
FDA Clears Endomina EZFuse System for GI Suturing
The FDA granted 510(k) clearance to Endo Tools Therapeutics’ Endomina EZFuse system, enabling U.S. commercialization of a next‑generation GI suturing platform. The device uses a single‑movement mechanism that can cut procedure time by up to 50 percent, simplifying internal stitching...
World Immunization Week 2026: What’s Driving Immunization Progress?
World Immunization Week 2026, themed “For every generation, vaccines work,” underscores vaccines’ role across the life span. The WHO’s Immunization Agenda 2030 Mid‑Term Review reports that vaccines have averted more than 150 million deaths in five decades and save over 4 million...

Retatrutide Vs. Zepbound: How Does Lilly’s Next-Gen Obesity Drug Compare to Tirzepatide?
Eli Lilly’s investigational triple‑agonist retatrutide is delivering weight‑loss results that eclipse its approved dual‑agonist Zepbound (tirzepatide), with Phase III trials reporting up to 28.7% body‑weight reduction over 68 weeks and HbA1c drops of about 2.0%. Zepbound, already a blockbuster generating roughly...